Physicochemical Properties
| Molecular Formula | C21H25NO4 |
| Molecular Weight | 355.4275 |
| Exact Mass | 355.178 |
| CAS # | 3520-14-7 |
| PubChem CID | 969488 |
| Appearance | White to light yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 482.9±45.0 °C at 760 mmHg |
| Flash Point | 138.7±25.9 °C |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.609 |
| LogP | 3.7 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 26 |
| Complexity | 475 |
| Defined Atom Stereocenter Count | 1 |
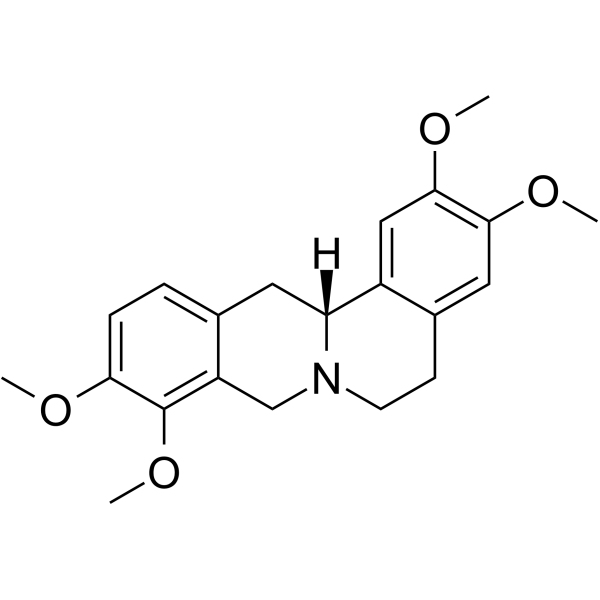
| SMILES | COC1=C(C2=C(C[C@@H]3C4=CC(=C(C=C4CCN3C2)OC)OC)C=C1)OC |
| InChi Key | AEQDJSLRWYMAQI-QGZVFWFLSA-N |
| InChi Code | InChI=1S/C21H25NO4/c1-23-18-6-5-13-9-17-15-11-20(25-3)19(24-2)10-14(15)7-8-22(17)12-16(13)21(18)26-4/h5-6,10-11,17H,7-9,12H2,1-4H3/t17-/m1/s1 |
| Chemical Name | (13aR)-2,3,9,10-tetramethoxy-6,8,13,13a-tetrahydro-5H-isoquinolino[2,1-b]isoquinoline |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Dopamine D1 receptor (Ki: 1.8 ± 0.3 μM) [2] - Dopamine D2 receptor (Ki: 0.7 ± 0.2 μM) [2] |
| ln Vitro |
1. Dopamine receptor binding activity: D-Tetrahydropalmatine exhibited specific binding to dopamine D1 and D2 receptors in rat brain membrane preparations. In competitive binding assays, it displaced [³H]-SCH23390 (D1 receptor ligand) with a Ki value of 1.8 ± 0.3 μM, and displaced [³H]-spiperone (D2 receptor ligand) with a Ki value of 0.7 ± 0.2 μM. The binding affinity for D2 receptors was approximately 2.6-fold higher than that for D1 receptors [2] 2. Inhibition of dopamine-mediated receptor activation: D-Tetrahydropalmatine inhibited dopamine-induced activation of D1 and D2 receptors. In rat striatal membrane preparations, 10 μM D-Tetrahydropalmatine reduced dopamine (1 μM)-induced D1 receptor-mediated cyclic AMP (cAMP) accumulation by 62 ± 5%, and decreased dopamine-induced D2 receptor-mediated [³H]-GTPγS binding by 78 ± 4% [2] |
| Enzyme Assay |
1. Dopamine D1 receptor binding assay: Rat cerebral cortex was homogenized in ice-cold Tris-HCl buffer (pH 7.4) containing EDTA and sucrose. The homogenate was centrifuged at 10,000 × g for 20 minutes at 4°C, and the pellet was resuspended in the same buffer to prepare crude membrane fractions. The binding reaction mixture (200 μL) contained membrane fractions (50 μg protein), [³H]-SCH23390 (0.5 nM, D1 ligand), and different concentrations of D-Tetrahydropalmatine (0.1–10 μM). The mixture was incubated at 37°C for 60 minutes, then filtered through glass fiber filters to separate bound and free ligands. The filters were washed 3 times with ice-cold buffer, and the radioactivity was measured using a liquid scintillation counter. Non-specific binding was determined in the presence of 10 μM unlabeled SCH23390, and Ki values were calculated using the Cheng-Prusoff equation [2] 2. Dopamine D2 receptor binding assay: Rat striatum was processed to obtain crude membrane fractions as described above. The reaction mixture (200 μL) included membrane fractions (40 μg protein), [³H]-spiperone (0.3 nM, D2 ligand), and D-Tetrahydropalmatine (0.05–5 μM). Incubation was performed at 25°C for 45 minutes, followed by filtration and radioactivity measurement. Non-specific binding was assessed with 10 μM unlabeled spiperone, and Ki values were derived from competitive binding curves [2] |
| Animal Protocol |
1. In vivo dopamine receptor modulation experiment (rat model): Male Sprague-Dawley rats (200–250 g) were randomly divided into three groups: D-Tetrahydropalmatine low-dose (5 mg/kg), high-dose (15 mg/kg), and vehicle control. D-Tetrahydropalmatine was dissolved in 0.9% normal saline containing 0.1% Tween 80, and administered via intraperitoneal injection once daily for 7 days. On the 8th day, rats were sacrificed, and the striatum, cerebral cortex, and hippocampus were dissected. The tissue samples were used for dopamine receptor binding assays (as described in Enzyme Assay) to evaluate the in vivo effect of D-Tetrahydropalmatine on receptor density and affinity [2] |
| References |
[1]. [Effects of different types and standard of processing vinegaron inherent constituents in rhizoma of Corydalis yanhusuo]. Zhongguo Zhong Yao Za Zhi. 2006 Mar;31(6):465-7. [2]. Effects of tetrahydroprotoberberines on dopamine receptor subtypes in brain. Zhongguo Yao Li Xue Bao. 1989 Mar;10(2):104-10. [3]. Organic cation transporter 1 mediates the uptake of monocrotaline and plays an important rolein its hepatotoxicity. Toxicology. 2013 Sep 15;311(3):225-30. |
| Additional Infomation |
D-Tetrahydropalmatine is an alkaloid. Tetrahydropalmatine is under investigation in clinical trial NCT02118610 (Treatment of Schizophrenia With L-tetrahydropalmatine (l-THP): a Novel Dopamine Antagonist With Anti-inflammatory and Antiprotozoal Activity). D-Tetrahydropalmatine has been reported in Corydalis solida, Stephania lincangensis, and other organisms with data available. - D-Tetrahydropalmatine is a tetrahydroprotoberberine alkaloid naturally present in the rhizome of Corydalis yanhusuo. Literature [1] showed that different processing vinegars (e.g., rice vinegar, sorghum vinegar) affected its content: processing with rice vinegar increased the content of D-Tetrahydropalmatine by 12–15% compared to unprocessed Corydalis yanhusuo, while sorghum vinegar processing led to a 8–10% increase [1] - The high affinity of D-Tetrahydropalmatine for dopamine D2 receptors suggests it may act as a D2 receptor antagonist or partial agonist, which is associated with its potential effects on central nervous system functions (e.g., modulation of dopamine neurotransmission) [2] - Literature [3] focuses on monocrotaline and organic cation transporter 1, with no relevant information about D-Tetrahydropalmatine [3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~140.67 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.03 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8135 mL | 14.0675 mL | 28.1349 mL | |
| 5 mM | 0.5627 mL | 2.8135 mL | 5.6270 mL | |
| 10 mM | 0.2813 mL | 1.4067 mL | 2.8135 mL |