Physicochemical Properties
| Molecular Formula | C6H12N2O4S2 |
| Molecular Weight | 240.29 |
| Exact Mass | 240.023 |
| CAS # | 56-89-3 |
| Related CAS # | L-Cystine-d4;1192736-38-1;L-Cystine-34S2;113512-08-6;(S)-L-Cystine-15N2;L-Cystine-3,3'-13C2;2483736-13-4 |
| PubChem CID | 67678 |
| Appearance | White to off-white solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 468.2±45.0 °C at 760 mmHg |
| Melting Point | 260-261ºC |
| Flash Point | 237.0±28.7 °C |
| Vapour Pressure | 0.0±2.5 mmHg at 25°C |
| Index of Refraction | 1.653 |
| LogP | 1.23 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 14 |
| Complexity | 192 |
| Defined Atom Stereocenter Count | 2 |
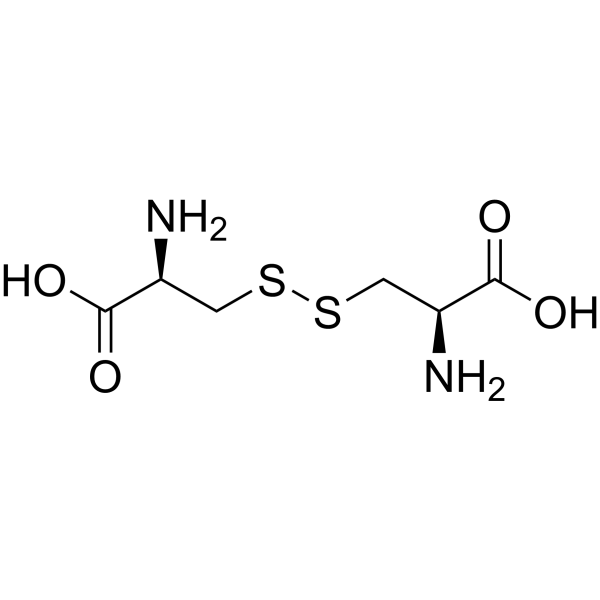
| SMILES | C([C@@H](C(=O)O)N)SSC[C@@H](C(=O)O)N |
| InChi Key | LEVWYRKDKASIDU-IMJSIDKUSA-N |
| InChi Code | InChI=1S/C6H12N2O4S2/c7-3(5(9)10)1-13-14-2-4(8)6(11)12/h3-4H,1-2,7-8H2,(H,9,10)(H,11,12)/t3-,4-/m0/s1 |
| Chemical Name | (2R)-2-amino-3-[[(2R)-2-amino-2-carboxyethyl]disulfanyl]propanoic acid |
| Synonyms | NSC-13203; NSC 13203; Cystine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. Extracellular Concentration of L-Cystine Determines the Sensitivity to System xc- Inhibitors. Biomol Ther (Seoul). 2022 Mar 1;30(2):184-190. [2]. Enhancement of antigen-specific immunoglobulin G production in mice by co-administration of L-cystine and L-theanine. J Vet Med Sci. 2007 Dec;69(12):1263-70. [3]. Cystinuria and cystinosis are usually related to L-cystine: is this really the case for cystinosis? A physicochemical investigation at micrometre and nanometre scale. Comptes Rendus. Chimie, Volume 25 (2022) no. S1, pp. 489-502. [4]. Cellular accumulation of L-cystine in rat kidney cortex in vivo. J Clin Invest. 1973 Feb;52(2):454-62. [5]. Uptake of L-cystine via an ABC transporter contributes defense of oxidative stress in the L-cystine export-dependent manner in Escherichia coli. PLoS One. 2015 Apr 2;10(3):e0120619. [6]. Fresh Medium or L-Cystine as an Effective Nrf2 Inducer for Cytoprotection in Cell Culture. Cells. 2023 Jan 12;12(2):291. |
| Additional Infomation |
L-cystine is the L-enantiomer of the sulfur-containing amino acid cystine. It has a role as a flour treatment agent, a human metabolite, a Saccharomyces cerevisiae metabolite, a mouse metabolite and an EC 1.2.1.11 (aspartate-semialdehyde dehydrogenase) inhibitor. It is a cystine, a L-cysteine derivative and a non-proteinogenic L-alpha-amino acid. It is a conjugate acid of a L-cystine anion. It is an enantiomer of a D-cystine. It is a tautomer of a L-cystine zwitterion. A covalently linked dimeric nonessential amino acid formed by the oxidation of cysteine. Two molecules of cysteine are joined together by a disulfide bridge to form cystine. L-Cystine is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). L-cystine has been reported in Salvia hispanica, Glycine max, and other organisms with data available. Cystine is not considered one of the 20 amino acids, Cystine is a sulfur-containing derivative obtained from oxidation of cysteine amino acid thiol side chains. It functions as an antioxidant and protects tissues against radiation and pollution, slowing the aging process. It also aids protein synthesis. Cystine is abundant in many proteins of skeletal tissues and skin, and found in insulin and digestive enzymes chromotrypsinogen A, papain, and trypsinogen. (NCI04) A covalently linked dimeric nonessential amino acid formed by the oxidation of CYSTEINE. Two molecules of cysteine are joined together by a disulfide bridge to form cystine. Drug Indication It has been claimed that L-cysteine has anti-inflammatory properties, that it can protect against various toxins, and that it might be helpful in osteoarthritis and rheumatoid arthritis. More research will have to be done before L-cysteine can be indicated for any of these conditions. Research to date has mostly been in animal models. Mechanism of Action Certain conditions, e.g. an acetaminophen overdose, deplete hepatic glutathione and subject the tissues to oxidative stress resulting in loss of cellular integrity. L-Cystine serves as a major precursor for synthesis of glutathione. Pharmacodynamics L-Cystine is a covalently linked dimeric nonessential amino acid formed by the oxidation of cysteine. Two molecules of cysteine are joined together by a disulfide bridge to form cystine. Cystine is a chemical substance which naturally occurs as a deposit in the urine, and can form a calculus (hard mineral formation) when deposited in the kidney. The compound produced when two cysteine molecules linked by a disulfide (S-S) bond. Cystine is required for proper vitamin B6 utilization and is also helpful in the healing of burns and wounds, breaking down mucus deposits in illnesses such as bronchitis as well as cystic fibrosis. Cysteine also assists in the supply of insulin to the pancreas, which is needed for the assimilation of sugars and starches. It increases the level of glutathione in the lungs, liver, kidneys and bone marrow, and this may have an anti-aging effect on the body by reducing age-spots etc. |
Solubility Data
| Solubility (In Vitro) |
0.1 M NaOH : ~3.33 mg/mL (~13.86 mM) 0.1 M HCL : 2.5 mg/mL (~10.40 mM) DMSO :< 1 mg/mL H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.1616 mL | 20.8082 mL | 41.6164 mL | |
| 5 mM | 0.8323 mL | 4.1616 mL | 8.3233 mL | |
| 10 mM | 0.4162 mL | 2.0808 mL | 4.1616 mL |