Physicochemical Properties
| Molecular Formula | C11H12BRNO3S |
| Molecular Weight | 318.186881065369 |
| Exact Mass | 316.972 |
| CAS # | 1947408-74-3 |
| PubChem CID | 145925689 |
| Appearance | White to off-white solid powder |
| LogP | 2.1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 17 |
| Complexity | 280 |
| Defined Atom Stereocenter Count | 1 |
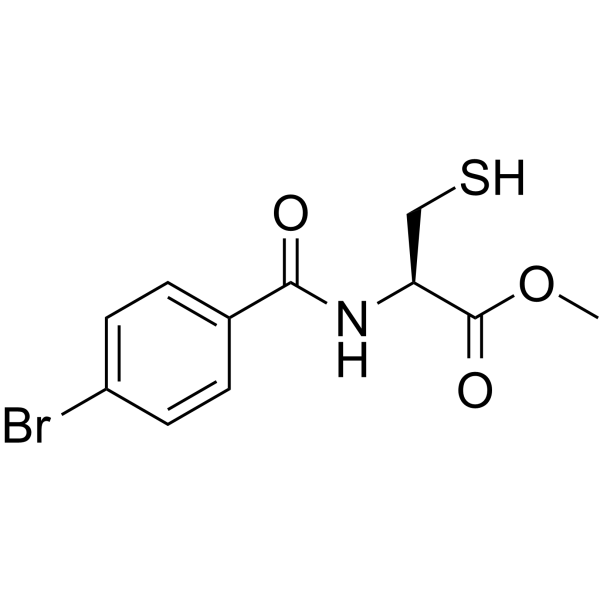
| SMILES | COC(=O)[C@H](CS)NC(=O)C1=CC=C(C=C1)Br |
| InChi Key | QLAHQHTYCQKQLI-VIFPVBQESA-N |
| InChi Code | InChI=1S/C11H12BrNO3S/c1-16-11(15)9(6-17)13-10(14)7-2-4-8(12)5-3-7/h2-5,9,17H,6H2,1H3,(H,13,14)/t9-/m0/s1 |
| Chemical Name | methyl (2R)-2-[(4-bromobenzoyl)amino]-3-sulfanylpropanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | When it comes to β-lactam and β-lactone-based electrophilic natural products, the cysteine thiol probe (probe 1) responds more easily, whereas it reacts less quickly with epoxide-based electrophilic natural products. Probe 2 reacts with natural compounds containing epoxides substantially more quickly. Their simultaneous usage in extracts is made possible by their orthogonal reactivity. The isotopic pattern of the tagged natural product indicates the structural origin of the product because probe 2 is chlorinated and the cysteine thiol probe is brominated. Competition studies with the cysteine thiol probe and 2 revealed that the chlorinated probe 2 reacts alone with the epoxide in salt amide A, while the brominated cysteine thiol probe only reacts with the β-lactam in penicillin G and the β-lactone in salinosporamide A [1]. |
| References |
[1]. Thiol Probes To Detect Electrophilic Natural Products Based on Their Mechanism of Action. ACS Chem Biol. 2016 Aug 19;11(8):2328-36. [2]. Thiol-Based Probe for Electrophilic Natural Products Reveals That Most of the Ammosamides Are Artifacts. J Nat Prod. 2017 Jan 27;80(1):126-133. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~785.69 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1428 mL | 15.7139 mL | 31.4278 mL | |
| 5 mM | 0.6286 mL | 3.1428 mL | 6.2856 mL | |
| 10 mM | 0.3143 mL | 1.5714 mL | 3.1428 mL |