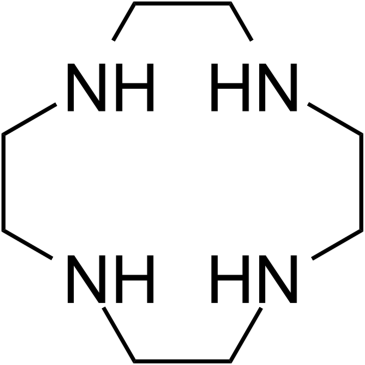
Cyclen is the macrocyclic aza analogue of crown ether, used as a precursor for MRI contrast agents, and is an intermediate for the preparation of effective macrocyclic chelates. Cyclen compounds are capable of selectively binding cations and are used as a ligand with chemicals used in MRI contrast (as well as other imaging) agents.
Physicochemical Properties
| Molecular Formula | C8H20N4 |
| Molecular Weight | 172.2712 |
| Exact Mass | 172.168 |
| CAS # | 294-90-6 |
| PubChem CID | 64963 |
| Appearance | White to off-white solid powder |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 283.8±8.0 °C at 760 mmHg |
| Melting Point | 110-113 °C(lit.) |
| Flash Point | 129.5±13.5 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.424 |
| LogP | -2.34 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 12 |
| Complexity | 65.1 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | QBPPRVHXOZRESW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H20N4/c1-2-10-5-6-12-8-7-11-4-3-9-1/h9-12H,1-8H2 |
| Chemical Name | 1,4,7,10-tetrazacyclododecane |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Water molecules (as Cyclen-based Gd³⁺ complexes act by shortening the relaxation time of water protons for MRI contrast enhancement) [1] |
| ln Vitro |
- Cyclen-based Gd³⁺ complexes exhibited enhanced longitudinal relaxation rates (r1) compared to clinically used MRI contrast agents (e.g., Gd-DTPA). The r1 values ranged from 3.0 to 10.5 mM⁻¹s⁻¹ at 1.5 T and 37°C, depending on the structural modifications of the Cyclen ligand (e.g., introduction of hydrophilic groups, rigidifying moieties, or targeting ligands) [1] - Cytotoxicity assays showed that most Cyclen-based Gd³⁺ complexes had low toxicity against mammalian cell lines (e.g., HeLa, HepG2, L929), with IC50 values greater than 100 μM after 24–72 h of incubation [1] - Cell uptake experiments demonstrated that Cyclen complexes modified with cell-penetrating peptides or receptor-targeting ligands showed increased internalization into specific cell types, as determined by ICP-MS analysis of intracellular Gd content [1] - The water exchange rate (kex) of Cyclen-Gd³⁺ complexes was measured to be in the range of 10⁶–10⁷ s⁻¹, which is optimal for efficient T1 relaxation enhancement [1] |
| ln Vivo |
- In Balb/c mice, intravenous injection of Cyclen-based Gd³⁺ complexes (0.1 mmol Gd/kg body weight) resulted in significant T1-weighted MRI signal enhancement in the kidneys, liver, and blood vessels within 5–30 min post-administration. The signal enhancement persisted for 2–4 h, depending on the ligand structure [1] - Tumor-bearing nude mice (xenograft models) injected with targeted Cyclen-Gd³⁺ complexes (modified with RGD peptide or folate) showed enhanced MRI contrast in tumor tissues compared to non-targeted Cyclen complexes, with a tumor-to-muscle signal ratio increased by 2–3 folds [1] - Biodistribution studies revealed that Cyclen-Gd³⁺ complexes were mainly accumulated in the kidneys and rapidly cleared via the renal pathway, with minimal retention in the liver and other organs [1] |
| Cell Assay |
- Cytotoxicity assay: Mammalian cells were seeded in 96-well plates at a density of 5×10³ cells per well and incubated overnight. Serial dilutions of Cyclen-based Gd³⁺ complexes (0.1–500 μM Gd) were added to the wells, and the cells were cultured for 24, 48, or 72 h. A cell viability reagent was added, and the absorbance was measured to calculate the survival rate and IC50 values [1] - Cell uptake assay: Cells were seeded in 6-well plates and incubated with Cyclen-Gd³⁺ complexes (50 μM Gd) for 1–4 h. After washing with PBS to remove unbound complexes, the cells were lysed, and the intracellular Gd concentration was quantified by ICP-MS to determine the uptake efficiency [1] - Fluorescence imaging assay: Cyclen ligands conjugated with fluorescent dyes were complexed with Gd³⁺, and the complexes were incubated with cells. Fluorescence microscopy was used to observe the cellular localization and internalization of the complexes [1] |
| Animal Protocol |
- MRI imaging in normal mice: Balb/c mice (20–25 g) were anesthetized and injected intravenously via the tail vein with Cyclen-based Gd³⁺ complexes (0.1 mmol Gd/kg body weight). T1-weighted MRI scans were performed at 0, 5, 15, 30, 60, 120, and 240 min post-injection to monitor signal enhancement in various organs [1] - Tumor imaging in nude mice: Nude mice bearing subcutaneous xenograft tumors (50–100 mm³) were injected intravenously with targeted or non-targeted Cyclen-Gd³⁺ complexes (0.1 mmol Gd/kg body weight). MRI scans were conducted at different time points to evaluate tumor contrast enhancement [1] - Biodistribution study: Mice were sacrificed 24 h after injection of Cyclen-Gd³⁺ complexes. Major organs (kidneys, liver, spleen, heart, lungs, tumor) were collected, weighed, and digested. The Gd concentration in each organ was measured by ICP-MS to calculate the tissue distribution profile [1] - Acute toxicity study: Mice were injected intravenously with a high dose of Cyclen-Gd³⁺ complexes (0.5 mmol Gd/kg body weight) and monitored for 14 days. Body weight changes, clinical signs of toxicity, and organ histopathology were evaluated [1] |
| ADME/Pharmacokinetics |
- The blood clearance half-life (t1/2β) of Cyclen-based Gd³⁺ complexes ranged from 0.8 to 2.5 h, with renal clearance as the major elimination pathway [1] - Biodistribution data showed that the highest Gd accumulation was in the kidneys (15–30 μg Gd/g tissue at 1 h post-injection), followed by the liver (2–5 μg Gd/g tissue), and minimal accumulation in other organs (heart, lungs, spleen < 2 μg Gd/g tissue) [1] - Urinary excretion of Gd was 60–85% within 24 h post-injection, indicating efficient renal clearance of Cyclen-Gd³⁺ complexes [1] - Plasma protein binding rate of Cyclen-Gd³⁺ complexes was low (5–15%), as determined by ultrafiltration assays [1] |
| Toxicity/Toxicokinetics |
- In vitro cytotoxicity: Cyclen-based Gd³⁺ complexes showed low toxicity to HeLa, HepG2, and L929 cells, with IC50 values > 100 μM (24 h incubation) and > 200 μM (72 h incubation) [1] - In vivo acute toxicity: No mortality or obvious clinical signs of toxicity (e.g., lethargy, loss of appetite) were observed in mice treated with Cyclen-Gd³⁺ complexes at doses up to 0.5 mmol Gd/kg body weight over 14 days [1] - Liver and kidney function tests (ALT, AST, BUN, creatinine) in treated mice showed no significant abnormalities compared to control groups [1] - Histopathological examination of kidneys, liver, and other organs revealed no obvious tissue damage or inflammation induced by Cyclen-Gd³⁺ complexes [1] |
| References |
[1]. Cyclen-based Gd3+ complexes as MRI contrast agents: Relaxivity enhancement and ligand design. Bioorg Med Chem. 2016 Nov 15;24(22):5663-5684. |
| Additional Infomation |
1,4,7,10-tetraazacyclododecane is an azacycloalkane that is cyclododecane in which the carbon atoms at positions 1, 4, 7 and 10 are replaced by nitrogen atoms. It is a saturated organic heteromonocyclic parent, a crown amine and an azacycloalkane. - Cyclen (1,4,7,10-tetraazacyclododecane) is a macrocyclic ligand that forms stable octadentate complexes with Gd³⁺ ions [1] - The mechanism of MRI contrast enhancement by Cyclen-based Gd³⁺ complexes involves the exchange of water molecules between the complex and the bulk solution, which shortens the longitudinal (T1) and transverse (T2) relaxation times of water protons [1] - Ligand design strategies for Cyclen include introducing hydrophilic groups (e.g., carboxylates, hydroxyls) to improve water solubility, rigidifying the macrocycle to optimize water exchange rate, and conjugating targeting moieties (e.g., RGD peptide, folate) to enhance tissue-specific accumulation [1] - Cyclen-based Gd³⁺ complexes have advantages over clinical contrast agents such as higher relaxation rates, better stability, and lower toxicity, making them promising candidates for diagnostic MRI applications [1] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~50 mg/mL (~290.24 mM) DMSO : ~1.85 mg/mL (~10.74 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.8048 mL | 29.0242 mL | 58.0484 mL | |
| 5 mM | 1.1610 mL | 5.8048 mL | 11.6097 mL | |
| 10 mM | 0.5805 mL | 2.9024 mL | 5.8048 mL |