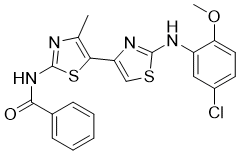
Corrector C4 is a novel and potent class II mutation corrector of ΔF508-CFTR (IC50 = 6.0 uM). Loss-of-function mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene cause the autosomal-recessive disorder cystic fibrosis (CF). Whereas therapies for CF have historically targeted disease symptoms, novel therapies are being developed that target mutation-specific defects of the CFTR protein, such as repair of 1) CFTR apical trafficking by correctors (e.g. VX-809) [1] and 2) CFTR gating by potentiators (e.g. VX-770).
Physicochemical Properties
| Molecular Formula | C21H17CLN4O2S2 |
| Molecular Weight | 456.96 |
| Exact Mass | 456.048 |
| Elemental Analysis | C, 55.20; H, 3.75; Cl, 7.76; N, 12.26; O, 7.00; S, 14.03 |
| CAS # | 421580-53-2 |
| PubChem CID | 1144671 |
| Appearance | Solid powder |
| Density | 1.44g/cm3 |
| Index of Refraction | 1.711 |
| LogP | 6.038 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 30 |
| Complexity | 583 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC1=C(SC(=N1)NC(=O)C2=CC=CC=C2)C3=CSC(=N3)NC4=C(C=CC(=C4)Cl)OC |
| InChi Key | RDOBOPJBMQURAT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H17ClN4O2S2/c1-12-18(30-21(23-12)26-19(27)13-6-4-3-5-7-13)16-11-29-20(25-16)24-15-10-14(22)8-9-17(15)28-2/h3-11H,1-2H3,(H,24,25)(H,23,26,27) |
| Chemical Name | N-(2-((5-chloro-2-methoxyphenyl)amino)-4'-methyl-[4,5'-bithiazol]-2'-yl)benzamide |
| Synonyms | Corrector C 4; Corrector C-4; Corrector C4 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CFTR (cystic fibrosis transmembrane conductance regulator) |
| ln Vitro | C4 acts as a class II corrector, targeting the nucleotide-binding domain (NBD)2–membrane-spanning domain (MSD)1 interface, which repairs CFTR-p.Phe508del expression and function synergistically when combined with class I correctors (VX-809) that target the NBD1–MSD2 interface [1]. |
| ln Vivo | Small-molecule therapies that restore defects in cystic fibrosis transmembrane conductance regulator (CFTR) gating (potentiators) or trafficking (correctors) are being developed for cystic fibrosis (CF) in a mutation-specific fashion. Options for pharmacological correction of CFTR-p.Phe508del (F508del) are being extensively studied but correction of other trafficking mutants that may also benefit from corrector treatment remains largely unknown.We studied correction of the folding mutants CFTR-p.Phe508del, -p.Ala455Glu (A455E) and -p.Asn1303Lys (N1303K) by VX-809 and 18 other correctors (C1-C18) using a functional CFTR assay in human intestinal CF organoids.Function of both CFTR-p.Phe508del and -p.Ala455Glu was enhanced by a variety of correctors but no residual or corrector-induced activity was associated with CFTR-p.Asn1303Lys. Importantly, VX-809-induced correction was most dominant for CFTR-p.Phe508del, while correction of CFTR-p.Ala455Glu was highest by a subgroup of compounds called bithiazoles (C4, C13, C14 and C17) and C5.These data support the development of mutation-specific correctors for optimal treatment of different CFTR trafficking mutants, and identify C5 and bithiazoles as the most promising compounds for correction of CFTR-p.Ala455Glu [1]. |
| Enzyme Assay |
Quantification of FIS [1] Forskolin-stimulated organoid swelling was automatically quantified using Volocity imaging software. The total organoid area (xy-plane) increase relative to t=0 min of forskolin treatment was calculated and averaged from two individual wells per condition. In some cases, cell debris and unviable structures were manually excluded based on criteria described in detail in a standard operating procedure. The area under the curve (AUC) (t=60 or 120 min, baseline 100%) was calculated using Prism.[1] Western blot analysis[1] Organoids that were untreated or treated with VX-809 or C17 were lysed in Laemmli buffer supplemented with complete protease inhibitor tablets. Lysates were analysed by SDS-PAGE and electrophoretically transferred to a polyvinylidene difluoride membrane. The membrane was blocked with 5% milk protein in Tris-buffered saline–Tween (0.3% Tween, 10 mM Tris (pH 8) and 150 mM sodium chloride in water) and probed for 3 h at room temperature with mouse monoclonal E-cadherin-specific (1 in 10 000 dilution; BD Biosciences) or CFTR-specific antibodies (Cystic Fibrosis Folding Consortium 450, 570 and 596; 1 in 3000 dilution;), followed by incubation with horseradish peroxidase-conjugated secondary antibodies and enhanced chemiluminescence development.[1] |
| Cell Assay |
Crypt isolation and organoid culture from rectal suction biopsies [1] Methods were slightly adapted from protocols described previously. In short, crypts were isolated and seeded in 50% Matrigel (growth factor reduced, phenol-free) in 24-well plates (∼10–30 crypts in three 10-μL matrigel droplets per well). Growth medium was further supplemented with Primocin (diluted 1 in 500). Vancomycin and gentamycin were added during the first week of culture. The medium was refreshed every 2–3 days and organoids were passaged ∼1 in 5 every 7–10 days.[1] The FIS assay[1] Methods to measure forskolin-induced organoid swelling described previously were slightly adapted. In short, rectal CF organoids (passage 1–30) from a 7–10-day-old culture were seeded in a flat-bottom 96-well culture plate in 5 μL 50% Matrigel commonly containing 20–80 organoids immersed in 100 μL complete culture medium, with or without 3 μM VX-809 or C1–C18 at concentrations as indicated in figure 2. 1 day after seeding, organoids were incubated for 30 min with 3 μM calcein green in complete culture medium. After calcein green staining, forskolin was added at concentrations as indicated in the figures and organoids were directly analysed by confocal live cell microscopy (LSM710), 5× objective) for 60 min (p.Phe508del/p.Phe508del and p.Phe508del/p.Ala455Glu) or 120 min (p.Phe508del/class I and p.Phe508del/p.Asn1303Lys). Two or three wells were used per condition. Per individual, each experiment was repeated at three to five different time-points. |
| Animal Protocol |
Human material [1] Approval for this study was obtained by the Ethics Committee of the University Medical Centre Utrecht and the Erasmus Medical Centre Rotterdam. Informed consent was obtained from all subjects participating in the study. Organoids from healthy controls and CF subjects (CFTR genotypes indicated in figure 1) were generated from four rectal suction biopsies after intestinal current measurements obtained 1) during standard CF care, 2) for diagnostic purposes or 3) during voluntary participation in studies. |
| References |
[1]. Mechanisms for rescue of correctable folding defects in CFTRDelta F508. Mol Biol Cell. 2009 Sep;20(18):4059-69. |
Solubility Data
| Solubility (In Vitro) | > 10 mM in DMSO |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1884 mL | 10.9419 mL | 21.8838 mL | |
| 5 mM | 0.4377 mL | 2.1884 mL | 4.3768 mL | |
| 10 mM | 0.2188 mL | 1.0942 mL | 2.1884 mL |