Physicochemical Properties
| Molecular Formula | C28H32O16 |
| Molecular Weight | 624.54408 |
| Exact Mass | 642.143 |
| CAS # | 146501-37-3 |
| PubChem CID | 101682254 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 975.0±65.0 °C at 760 mmHg |
| Flash Point | 320.2±27.8 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.727 |
| LogP | -0.13 |
| Hydrogen Bond Donor Count | 12 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 45 |
| Complexity | 1060 |
| Defined Atom Stereocenter Count | 10 |
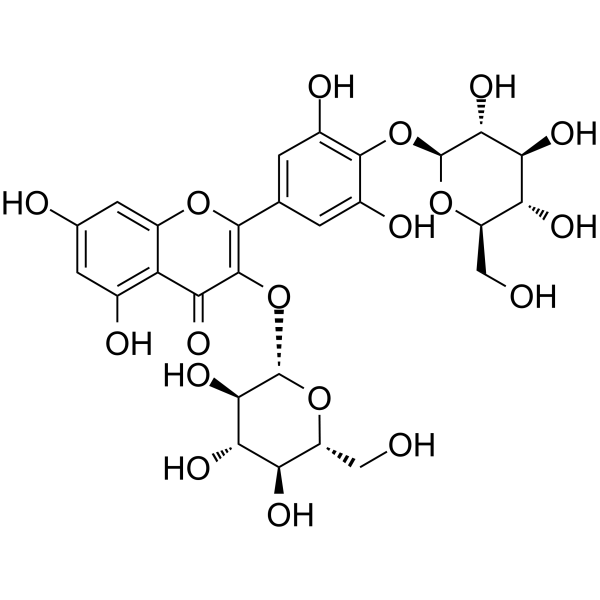
| SMILES | O=C1C(O[C@@H]([C@@H]([C@@H](O)[C@@H]2O)O)O[C@@H]2CO)=C(C3=CC(O)=C(O[C@@H]([C@@H]([C@@H](O)[C@@H]4O)O)O[C@@H]4CO)C(O)=C3)OC5=CC(O)=CC(O)=C15 |
| InChi Key | LLVOJEYSNCNXJN-XUAXIHHBSA-N |
| InChi Code | InChI=1S/C27H30O18/c28-5-13-16(34)19(37)21(39)26(42-13)44-24-10(32)1-7(2-11(24)33)23-25(18(36)15-9(31)3-8(30)4-12(15)41-23)45-27-22(40)20(38)17(35)14(6-29)43-27/h1-4,13-14,16-17,19-22,26-35,37-40H,5-6H2/t13-,14-,16-,17-,19+,20+,21-,22-,26+,27+/m1/s1 |
| Chemical Name | 2-[3,5-dihydroxy-4-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyphenyl]-5,7-dihydroxy-3-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-4-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | simple and sensitive LC-MS/MS technique was established for the measurement of saponin A in rat plasma in the range 2.3–575 ng/mL. Nereosin A was isolated from plasma by a protein precipitation process, separated by LC, and identified by MS/MS in positive electrospray ionization mode. The lower limit of quantification was determined at 2.3 ng/mL. Intraday and interday precision (LLOQ, low QC, medium QC, and high QC) were below 7.9%, and accuracy ranged from 94.0 to 105.1%. Matrix effects were acceptable (97.9–103.0%) and extraction recoveries were reproducible (88.5–94.4%). complanatoside A is stable under research conditions. This approach was utilized to explore the pharmacokinetics of sasoside A in rats [1]. |
| Animal Protocol |
A pharmacokinetic study was conducted in six healthy male Sprague-Dawley rats (200–230 g). Complanatoside A was formulated in water. A single oral dose of 30 mg/kg Complanatoside A was administered to each rat by oral gavage. Blood samples were collected from the vena orbitalis just before dosing and at 0.083, 0.167, 0.25, 0.5, 0.75, 1, 2, 3, 5, 7, 10, and 14 hours after administration. Plasma was separated by centrifugation and stored at –20°C until analysis.[1] |
| ADME/Pharmacokinetics |
Following a single oral dose of 30 mg/kg Complanatoside A to rats, the mean plasma concentration-time profile was obtained. Key pharmacokinetic parameters were calculated using non-compartmental analysis: Area under the curve from time zero to the last measurable time point (AUC0-t) was 566.0 ± 162.8 ng·h/mL. Area under the curve extrapolated to infinity (AUC0-∞) was 614.2 ± 192.6 ng·h/mL. Mean residence time (MRT) was 3.98 ± 0.87 hours. Elimination half-life (t1/2) was 3.20 ± 1.09 hours. Time to reach maximum plasma concentration (Tmax) was 1.08 ± 0.49 hours. Maximum plasma concentration (Cmax) was 110.8 ± 22.6 ng/mL. Apparent plasma clearance (CLz/F) was 53.3 ± 17.3 L/h/kg. Apparent volume of distribution (Vz/F) was 227.3 ± 45.4 L/kg.[1] |
| References |
[1]. Quantification of complanatoside A in rat plasma using LC-MS/MS and its application to a pharmacokinetic study. Biomed Chromatogr. 2016 Jun;30(6):888-93. |
| Additional Infomation |
Complanatoside A has been reported in Picea abies with data available. Complanatoside A (myricetin 3,4′-diglucoside) is a flavonol glycoside isolated from Astragalus complanatus. It is used as a phytochemical marker for the quality control of Semen Astragali Complanati in the 2010 edition of the Chinese Pharmacopoeia. This study developed and validated a simple, sensitive LC-MS/MS method for quantifying Complanatoside A in rat plasma for the first time, with a linear range of 2.3–575 ng/mL and a lower limit of quantification of 2.3 ng/mL. The method was successfully applied to a pharmacokinetic study in rats.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 35.7 mg/mL (~55.56 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6012 mL | 8.0059 mL | 16.0118 mL | |
| 5 mM | 0.3202 mL | 1.6012 mL | 3.2024 mL | |
| 10 mM | 0.1601 mL | 0.8006 mL | 1.6012 mL |