Physicochemical Properties
| Molecular Formula | C14H14O4 |
| Molecular Weight | 246.2586 |
| Exact Mass | 246.089 |
| CAS # | 3804-70-4 |
| Related CAS # | (+)-Columbianetin acetate;23180-65-6 |
| PubChem CID | 92201 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 443.0±45.0 °C at 760 mmHg |
| Flash Point | 171.9±22.2 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.611 |
| LogP | 1.69 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 18 |
| Complexity | 387 |
| Defined Atom Stereocenter Count | 1 |
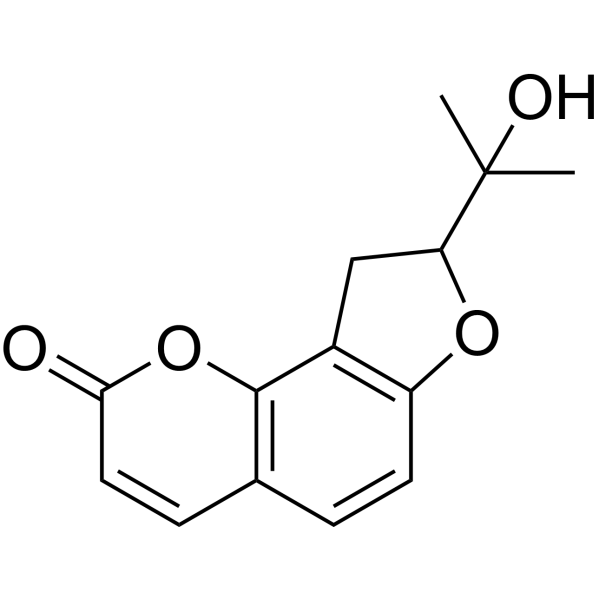
| SMILES | CC(C)([C@@H]1CC2=C(O1)C=CC3=C2OC(=O)C=C3)O |
| InChi Key | YRAQEMCYCSSHJG-NSHDSACASA-N |
| InChi Code | InChI=1S/C14H14O4/c1-14(2,16)11-7-9-10(17-11)5-3-8-4-6-12(15)18-13(8)9/h3-6,11,16H,7H2,1-2H3/t11-/m0/s1 |
| Chemical Name | (8S)-8-(2-hydroxypropan-2-yl)-8,9-dihydrofuro[2,3-h]chromen-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
- Against celery pathogens (including Botrytis cinerea and Fusarium oxysporum), Columbianetin showed dose-dependent inhibitory activity on mycelial growth and spore germination. At a concentration of 20 μg/mL, it inhibited the mycelial growth of Botrytis cinerea by 68% and the spore germination rate by 72%; at the same concentration, it inhibited the mycelial growth of Fusarium oxysporum by 59% [1] - In human mast cells (HMC-1 cell line) activated by phorbol 12-myristate 13-acetate (PMA, 10 nM) and calcium ionophore A23187 (1 μM), Columbianetin (1, 10, 30 μM) dose-dependently inhibited the release of pro-inflammatory mediators. At 30 μM, it reduced tumor necrosis factor-α (TNF-α) release by 62%, interleukin-6 (IL-6) release by 58%, and interleukin-8 (IL-8) release by 51% compared with the activated control group. It also inhibited histamine release by 45% at 30 μM [2] - Columbianetin (10, 30 μM) suppressed the activation of nuclear factor-κB (NF-κB) and mitogen-activated protein kinase (MAPK) pathways in activated HMC-1 cells: it reduced the phosphorylation of IκBα (inhibitor of NF-κB) by 70% at 30 μM, and decreased the phosphorylation levels of extracellular signal-regulated kinase (ERK) and p38 MAPK by 65% and 60%, respectively, at the same concentration [2] |
| Cell Assay |
- For human mast cell (HMC-1) experiment: HMC-1 cells were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin, maintained at 37°C in a 5% CO₂ incubator. Cells were seeded in 24-well plates at a density of 5×10⁵ cells/well and preincubated with Columbianetin (1, 10, 30 μM) for 1 hour. Then, the cells were stimulated with a combination of PMA (10 nM) and A23187 (1 μM) for 24 hours. After stimulation, the cell supernatant was collected by centrifugation. The levels of TNF-α, IL-6, and IL-8 in the supernatant were detected using enzyme-linked immunosorbent assay (ELISA) kits, and the histamine level was measured by a fluorometric assay. For Western blot analysis, cells were lysed with RIPA buffer containing protease and phosphatase inhibitors, and the lysates were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), then transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were probed with primary antibodies against phosphorylated IκBα (p-IκBα), phosphorylated ERK (p-ERK), phosphorylated p38 (p-p38), and β-actin (loading control), followed by incubation with secondary antibodies. The bands were visualized using an enhanced chemiluminescence (ECL) detection system [2] |
| Toxicity/Toxicokinetics |
- In the HMC-1 cell viability assay (MTT method), Columbianetin at concentrations up to 30 μM had no significant cytotoxicity after 24 hours of treatment; the cell viability remained above 90% compared with the untreated control group [2] |
| References |
[1]. Columbianetin, a phytoalexin associated with celery resistance to pathogens during storage. 1995. Volume 39, Issue 6, August 1995, Pages 1347-1350. [2]. Anti-inflammatory effect of Columbianetin on activated human mast cells. Biol Pharm Bull. 2009 Jun;32(6):1027-31. |
| Additional Infomation |
(S)-columbianetin is the (S)-(+)-enantiomer of columbianetin. It is an enantiomer of a (R)-columbianetin. Columbianetin has been reported in Zanthoxylum piasezkii, Campylotropis hirtella, and other organisms with data available. - Columbianetin is a phytoalexin naturally produced in celery during storage. Its accumulation in celery tissues is closely associated with the plant's resistance to post-harvest pathogenic infections (such as those caused by Botrytis cinerea and Fusarium oxysporum) [1] - The anti-inflammatory effect of Columbianetin on activated human mast cells is mainly mediated by inhibiting the NF-κB and MAPK signaling pathways, thereby reducing the release of pro-inflammatory cytokines and histamine. This suggests its potential application in the treatment of inflammatory diseases related to mast cell activation (such as allergic diseases) [2] - Columbianetin belongs to the coumarin chemical class, with a molecular structure containing a coumarin core and a hydroxyl group at the specific position, which is considered to be related to its biological activities (antimicrobial and anti-inflammatory) [1][2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~406.07 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (10.15 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (10.15 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (10.15 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.0607 mL | 20.3037 mL | 40.6075 mL | |
| 5 mM | 0.8121 mL | 4.0607 mL | 8.1215 mL | |
| 10 mM | 0.4061 mL | 2.0304 mL | 4.0607 mL |