Physicochemical Properties
| Molecular Formula | C25H25N3O2 |
| Molecular Weight | 399.4849 |
| Exact Mass | 399.195 |
| CAS # | 123437-32-1 |
| PubChem CID | 135439140 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.3 g/cm3 |
| Boiling Point | 568.2ºC at 760 mmHg |
| Flash Point | 297.4ºC |
| Vapour Pressure | 1.63E-13mmHg at 25°C |
| Index of Refraction | 1.685 |
| LogP | 4.718 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 30 |
| Complexity | 541 |
| Defined Atom Stereocenter Count | 0 |
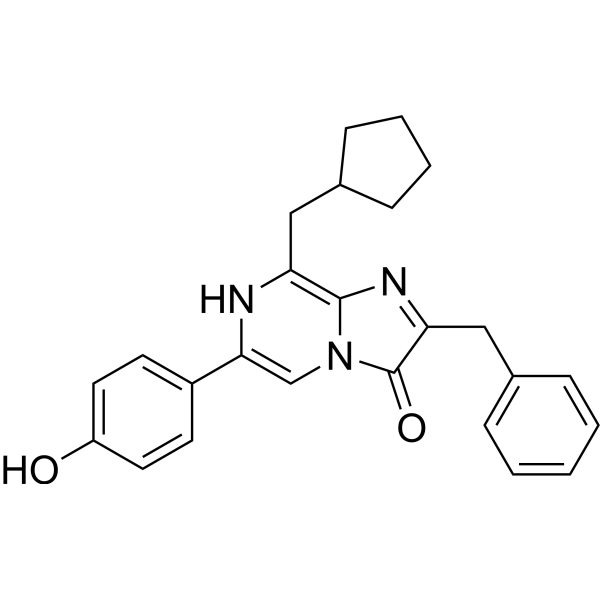
| SMILES | C1=CC=C(C=C1)CC2=NC3=C(CC4CCCC4)NC(=CN3C2=O)C5=CC=C(C=C5)O |
| InChi Key | UCSBOFLEOACXIR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C25H25N3O2/c29-20-12-10-19(11-13-20)23-16-28-24(21(26-23)14-17-8-4-5-9-17)27-22(25(28)30)15-18-6-2-1-3-7-18/h1-3,6-7,10-13,16-17,29-30H,4-5,8-9,14-15H2 |
| Chemical Name | 2-benzyl-8-(cyclopentylmethyl)-6-(4-hydroxyphenyl)imidazo[1,2-a]pyrazin-3-ol |
| Synonyms | 2-benzyl-8-(cyclopentylmethyl)-6-(4-hydroxyphenyl)-7H-imidazo[1,2-a]pyrazin-3-one; CLZN-hcp; 2-benzyl-8-(cyclopentylmethyl)-6-(4-hydroxyphenyl)imidazo[1,2-a]pyrazin-3-ol; Coelenterazine hcp, solid; SCHEMBL14117493; DTXSID40376337; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Luminescent enzyme substrate |
| ln Vitro | Bioluminescence-based techniques, such as bioluminescence imaging, BRET and dual-luciferase reporter assay systems, have been widely used to examine a myriad of biological processes. Coelenterazine (CTZ), a luciferin or light-producing compound found in bioluminescent organisms, has sparked great curiosity and interest in searching for analogues with improved photochemical properties. This review summarizes the current development of coelenterazine analogues, their bioluminescence properties, and the rational design of caged coelenterazine towards biotargets, as well as their applications in bioassays. It should be emphasized that the design of caged luciferins can provide valuable insight into detailed molecular processes in organisms and will be a trend in the development of bioluminescent molecules.[1] |
| Enzyme Assay | Caged-coelenterazine derivatives as bioluminescent probes for investigating enzyme activity. Bioluminescence imaging (BLI) is becoming an important technique that is readily applied in the life sciences. The coelenterate-luciferase system, which does not need extra co-factors in addition to molecular oxygen, is simpler than the firefly luciferase system or bacterial system. Therefore, coelenterazine derivatives could act as bioluminescent probes to monitor and image events in vitro and in vivo. In 2013, the Kazuya Kikuchi lab reported two caged coelenterazine derivatives, 132 and 133 (Fig. 11). They were designed and synthesized for imaging of β-galactosidase activity and expression in HEK-293T cell cultures that expressed a mutant Gaussia luciferase. These compounds were introduced with β-galactosidase cleavable caging groups at the key site of bioluminescence, the carbonyl group of the imidazopyrazinone moiety, which resulted in low auto-oxidation of coelenterazine and high specificity for β-galactosidase. The caged coelenterazine probes themselves did not emit bioluminescence with luciferase. However, in the presence of β-galactosidase, the probes containing β-galactosidase cleavable groups were cleaved by enzymes and free coelenterazine was generated to react with Gaussia luciferase to produce a bioluminescent signal (Fig. 11). Furthermore, the free coelenterazine could be generated from compound 133 in β-Gal-expressing cells. Also, it would only generate bioluminescence when it readily diffused to GLucM23-expressing cells with outer membrane bound Gluc or intracellularly localized Gluc (Fig. 11). In other words, the probes only induce bioluminescence when they are cleaved by β-galactosidase and encounter luciferase. Therefore, compound 133 has the potential to be used as a dual reporter in two different cell populations.83 These were the first probes based on a caged coelenterazine strategy to evaluate enzyme activity successfully. Their study can be helpful for further research on bioluminescent probes based on coelenterazine and their applications. It is difficult to introduce a large caging group at the C-3 position of the imidazopyrazinone core. Hence, this is an excellent example to guide the design of caged coelenterazine-type probes.[1] |
| Cell Assay | Coelenterazine-type bioluminescent probes for monitoring cell membrane fusion In 2013, the Kikuchi lab reported the first membrane impermeable coelenterazine derivative (Fig. 13), compound 145, which had the potential to be used as a bioluminescent probe for the monitoring of cell membrane fusion events (Fig. 14).85 This compound was synthesized by alkylating the coelenterazine with a linker containing a terminal anionic phosphonate moiety. Meanwhile, two other coelenterazine derivatives (143 and 144) containing a polyethylene glycol (PEG) linker with terminal benzyl-protecting groups were synthesized to explore the effect on bioluminescence. However, their performance was not as good as that of probe 145. Probe 145 was highly cell-impermeable due to its negative charge, resulting in failure to penetrate the cell membrane to react with intracellularly localized GlucER. However, probe 145 could emit a bioluminescence signal with outer-membrane bound GlucM23Mem. Also, if the secretory vesicle carrying Gluc fused with cell membranes, Gluc would be exposed to probe 145, thereby leading to the generation of bioluminescence. In addition, it is interesting that compound 145 showed a 30 fold higher bioluminescence activity for Gaussia luciferase (Gluc) over Renilla luciferase (RLuc), which indicated that Gluc was more appropriate. This probe is an example of the use of small molecular probes to explore biological processes, considering the lower application of coelenterazine-type probes.[1] |
| References |
[1]. Lighting up bioluminescence with coelenterazine: strategies and applications. Photochem Photobiol Sci. 2016;15(4):466-480. |
| Additional Infomation | The development of coelenterazine is focused on two main directions. One strategy is traditional modification of the coelenterazine substrate, especially the optimization of substitution at the C-2, C-6 and C-8 positions of the imidazopyrazinone core. Much work has been done in the past two decades in order to discover more suitable compounds with increased luminescence output, red-shifted emission and high stability of bioluminescence. Overall, the introduction of electron donating groups into the C-2, C-6 and C-8 positions which made the molecule more polar or increased the conjugation degree made a significant contribution to improving the bioluminescence intensity or influenced the maximum peak of emission. However, only a few of these compounds displayed excellence in all aspects that could be applied to bioluminescence assays, taking the place of native coelenterazine. The other strategy is to cage the 3-carbonyl position of the imidazopyrazinone skeleton of coelenterazine, which forms the critical active site of the bioluminescence reaction. The caged coelenterazine derivatives with high stability can emit bioluminescence only in particular situations. Therefore, the caged coelenterazine derivatives could be used as slow release substrates or as bioluminescent probes to detect biomacromolecules and bioactive small molecules. Very recently, some caged firefly luciferins as bioluminescent probes have been reported, and some of them display remarkable behavior.4,88–95 However, only a handful of studies have been done on caged coelenterazine. The reason why they have seen so little application may be that the yields of the caged reactions reported were very low. The explanation may be that it is difficult to cage the 3-carbonyl of the imidazopyrazinone ring due to its sensitivity to base and oxygen. Moreover, coelenterazine and its derivatives seem to be more unstable compared with firefly luciferins, which could be the main drawback of its application. Another disadvantage of coelenterazine is that its light emission maximum is less than 600 nm, which makes it inappropriate for deep tissue imaging. However, the coelenterazine bioluminescence system still has obvious advantages. This universal luciferin, coelenterazine, could be used in numerous bioluminescent luciferase systems, such as Renilla and Gaussia. Moreover, most of the coelenterazines involved a bioluminescence system that only consists of luciferin and luciferase without other constituents, which is much simpler. So far, the application of luciferin–luciferase bioluminescence has been broadly extended to a variety of fields in the chemobiological sciences. It should be emphasized that the design of caged luciferins can provide valuable insight into the detailed molecular processes of organisms and represents a new trend in the development of bioluminescent molecules. Caged coelenterazine probes, an important class of bioluminescent molecules, have the potential to become popular in future research fields. Meanwhile, novel strategies and techniques should be proposed and developed to promote the application of coelenterazine-type bioluminescence.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5033 mL | 12.5163 mL | 25.0325 mL | |
| 5 mM | 0.5007 mL | 2.5033 mL | 5.0065 mL | |
| 10 mM | 0.2503 mL | 1.2516 mL | 2.5033 mL |