Physicochemical Properties
| Molecular Formula | C14H19BR2CLN4S |
| Molecular Weight | 470.6535 |
| Exact Mass | 467.938 |
| Elemental Analysis | C, 35.73; H, 4.07; Br, 33.95; Cl, 7.53; N, 11.90; S, 6.81 |
| CAS # | 145231-35-2 |
| Related CAS # | Clobenpropit;145231-45-4 |
| PubChem CID | 11213569 |
| Appearance | White to off-white solid powder |
| Melting Point | 205 °C |
| LogP | 5.86 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 22 |
| Complexity | 306 |
| Defined Atom Stereocenter Count | 0 |
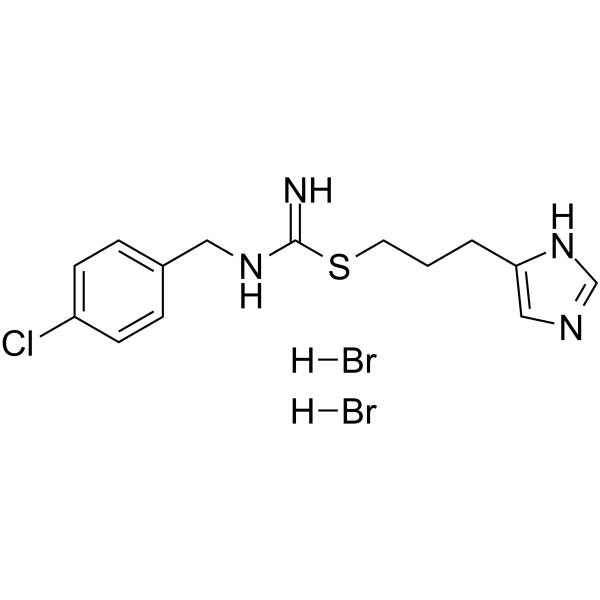
| SMILES | Br[H].Br[H].ClC1C([H])=C([H])C(=C([H])C=1[H])C([H])([H])/N=C(/N([H])[H])\SC([H])([H])C([H])([H])C([H])([H])C1=C([H])N=C([H])N1[H] |
| InChi Key | JIJQPEZAVLJZBO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H17ClN4S.2BrH/c15-12-5-3-11(4-6-12)8-18-14(16)20-7-1-2-13-9-17-10-19-13;;/h3-6,9-10H,1-2,7-8H2,(H2,16,18)(H,17,19);2*1H |
| Chemical Name | 3-(1H-imidazol-5-yl)propyl N'-[(4-chlorophenyl)methyl]carbamimidothioate;dihydrobromide |
| Synonyms | Clobenpropit HBr; Clobenpropit dihydrobromide; VUF 9153; VUF-9153; VUF9153; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Clobenpropit exhibits a pK of 9.44±0.04 for human H3LR and 9.75±0.01 for rat H3LR binding. The affinity of clobenpropit for histamine H1R or H2R is minimal (pK 5.2 and 5.6, respectively) [1]. Clobenpropit has a concentration-dependent effect on [3H]-dopamine transport in SH-SY5Y cells, with a maximal inhibition rate of 82.7±2.8%, IC50 of 490 nM (pIC50 6.31±0.11)[2]. The IC50 for NR1/NR2B receptors is 1 μM, and clobenpropit is a non-competitive, subunit-selective antagonist of recombinant NMDA receptors [2]. When Clobenpropit (50 μM) and Gemcitabine (5 μM) were administered together, Panc-1, MiaPCa-2, and AsPC-1 apoptosis increased dramatically in comparison to the control group [3]. |
| ln Vivo | Significant tumor growth inhibition was demonstrated by combination therapy with gemcitabine (125 mg/kg intraperitoneally twice a week for 40 days) and chlorprofen (20 μM/kg intraperitoneally every other day for 40 days) [ 3]. |
| Cell Assay |
Apoptosis analysis[3] Cell Types: Pancreatic cancer cells (Panc-1, MiaPaCa-2 and AsPC-1) Tested Concentrations: 50 μM Incubation Duration: Experimental Results: Gemcitabine (5 μM) combination enhanced apoptosis. |
| Animal Protocol |
Animal/Disease Models: 5weeks old male BALB/c nude mice, Panc-1 xenograft [3] Doses: 20 μM/kg Route of Administration: intraperitoneal (ip) injection; once every other day for 40 days. Gemcitabine (intraperitoneal (ip) injection twice a week, 125 mg/kg, for 40 days) Experimental Results: Compared with other treatment groups (control group 501±92 mg, gemcitabine 294±46 mg, chlorprofenate 444±167), The combination treatment demonstrated significant tumor growth inhibition. mg, and combination 154 ± 54 mg). |
| References |
[1]. Two novel and selective nonimidazole histamine H3 receptor antagonists A-304121 and A-317920: I. In vitro pharmacological effects. J Pharmacol Exp Ther. 2003 Jun;305(3):887-96. [2]. Clobenpropit, a histamine H3 receptor antagonist/inverse agonist, inhibits [3H]-dopamine uptake by human neuroblastoma SH-SY5Y cells and rat brain synaptosomes. Pharmacol Rep. 2018 Feb;70(1):146-155. [3]. Clobenpropit enhances anti-tumor effect of gemcitabine in pancreatic cancer. World J Gastroenterol. 2014 Jul 14;20(26):8545-57. |
| Additional Infomation | Clobenpropit dihydrobromide is a hydrobromide salt obtained by reaction of clobenpropit with two equivalents of hydrobromic acid. An extremely potent histamine H3 antagonist/inverse agonist (pA2 = 9.93). Also displays partial agonist activity at H4 receptors; induces eosinophil shape change with an EC50 of 3 nM. It has a role as a H4-receptor agonist and a H3-receptor antagonist. It contains a clobenpropit(2+). |
Solubility Data
| Solubility (In Vitro) | DMSO : ~125 mg/mL (~265.59 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.42 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.42 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.42 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1247 mL | 10.6236 mL | 21.2472 mL | |
| 5 mM | 0.4249 mL | 2.1247 mL | 4.2494 mL | |
| 10 mM | 0.2125 mL | 1.0624 mL | 2.1247 mL |