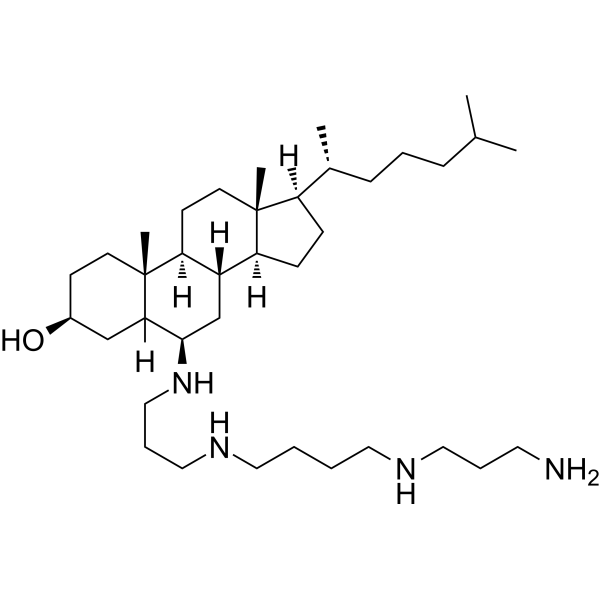
Physicochemical Properties
| Molecular Formula | C37H72N4O |
| Molecular Weight | 588.99 |
| CAS # | 1430194-56-1 |
| Related CAS # | 3030428-57-7 (TFA) |
| Appearance | Typically exists as solid at room temperature |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Lipid membrane |
| ln Vitro | Claramine (2-20 μM; 20 h) does not affect cell viability in human neuroblastoma cells (SH-SY5Y) at concentrations below 10 μM. Claramine (2-20 μM; 20 h) does not affect cell viability in HEK293 cells[1]. Claramine (2.5-10 μM; 20 h) protects human neuroblastoma (SH-SY5Y) cells from pore formers melittin (HY-P0233) (4 μM; 20 h) and α-hemolysin (50 μg/mL; 20 h) by inhibiting their binding to the cell membrane[1]. |
| Enzyme Assay |
Melittin Binding to the Cellular Membrane [1] To label melittin, 300 μM Alexa Fluor 488 N-hydroxysuccinimide (NHS) ester (succinimidyl ester) was incubated with gentle shaking for 2 h with 900 μM melittin in 0.1 mM sodium bicarbonate buffer (pH 8.0). SH-SY5Y cells were seeded on glass coverslips and treated for 5 min with 0.2 μM labeled melittin in the absence or presence of 0.1, 1.0, and 10 μM claramine. After incubation, the cells were washed with phosphate-buffered saline (PBS) and counterstained with 5 μg/mL Alexa Fluor 633-conjugated wheat germ agglutinin. After washing with PBS, cells were fixed in 2% paraformaldehyde. Fluorescence emission was detected after double excitation at 488 and 633 nm by the above-described scanning confocal microscopy system using a 60× oil immersion objective. A series of 1.0 μm thick optical sections (1024 × 1024) were acquired, and all sections were projected as a single composite image by superimposition. ImageJ was used to calculate the percentage of colocalization between cell membranes and melittin. α-Hemolysin Binding to the Cellular Membrane [1] SH-SY5Y cells were seeded on glass coverslips and treated for 15 min with 5 μg/mL (e.g., about 0.15 μM in monomer equivalents) of α-hemolysin in the absence or presence of 0.1 and 10 μM claramine. After incubation, the cells were washed with PBS, counterstained with 10 μg/ml Alexa Fluor 633-conjugated wheat germ agglutinin, and fixed in 2% paraformaldehyde. After washing with PBS, the presence of α-hemolysin was detected with 1:750 diluted rabbit antistaphylococcal α-toxin primary antibodies and subsequently with 1:1000 diluted Alexa Fluor 488-conjugated antirabbit secondary antibodies. Fluorescence emission was detected after double excitation at 488 and 633 nm by the above-described scanning confocal microscopy system using a 20× objective. A series of 1.0 μm thick optical sections (1024 × 1024) were acquired, and all sections were projected as a single composite image by superimposition. ImageJ was used to calculate the percentage of colocalization between cell membranes and α-hemolysin. [1] Turbidity Measurements [1] The same samples from the ANS preparation were analyzed for absorbance using a plate reader with spectral scanning. With the CD, ANS, and turbidity measurements, we elected to probe only up to 30 μM given that 40 and 50 μM concentrations of claramine in the absence of melittin caused an increase in ANS fluorescence intensity in the absence of a blue shift, indicating that there may be an avidity effect between claramine and ANS at such high concentrations, while concentrations of claramine at and below 30 μM did not clearly change the signal of free ANS or its absorbance (Figure S10). We note that it was necessary to use a 5-fold greater concentration of melittin in these in vitro measurements in comparison to the tissue culture experiments to resolve a sufficient and reproducible signal from the peptide alone. |
| Cell Assay |
MTT Reduction Assay [1] Melittin (2 μM, in monomer equivalents) or α-hemolysin (50 μg/mL corresponding to ca. 1.5 μM, in monomer equivalents) were added to the cell culture media and incubated with or without increasing concentrations of CL for 1 h at 37 °C under quiescent conditions. After this incubation, the culture media of cells seeded in 96-well plates was replaced with the aforementioned solutions containing melittin and claramine for 20 h or 30 min, as indicated in the text. Following treatment of the cells, the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide reduction assay was performed as previously described. Measurement of Intracellular ROS [1] Melittin (0.1 μM, monomer equivalents) or α-hemolysin (50 μg/mL, monomer equivalents) were added to the cell culture media of SH-SY5Y cells seeded on glass coverslips for 5 or 1 min, respectively, in the absence or presence of 0.01–10 μM claramine. To detect intracellular ROS production, cells were loaded with 10 μM 6-chloromethyl-2′,7′-dichlorodihydrofluorescein diacetate (CM-H2DCFDA) during the aforementioned treatment. The resulting fluorescence was analyzed by a Nikon C2 scanning laser confocal microscopy system. A series of 1.0 μm thick optical sections (1024 × 1024 or 2048 × 2048) were taken through the cells using a Nikon Eclipse Ti inverted microscope equipped with a 60× oil immersion objective and then projected as a single composite image by superimposition. The confocal microscope was set at optimal acquisition conditions, e.g., pinhole diameters, detector gain, and laser powers. Settings were maintained constant for all image acquisitions. For Figures 1C and 3B, the same images are shown with enhanced brightness and contrast such that all cells can be visualized (Figure S9), including those with low fluorescence signals. |
| References |
[1]. A Brain-Permeable Aminosterol Regulates Cell Membranes to Mitigate the Toxicity of Diverse Pore-Forming Agents. ACS Chem Neurosci. 2022;13(8):1219-1231. |
| Additional Infomation |
The molecular composition of the plasma membrane plays a key role in mediating the susceptibility of cells to perturbations induced by toxic molecules. The pharmacological regulation of the properties of the cell membrane has therefore the potential to enhance cellular resilience to a wide variety of chemical and biological compounds. In this study, we investigate the ability of claramine, a blood-brain barrier permeable small molecule in the aminosterol class, to neutralize the toxicity of acute biological threat agents, including melittin from honeybee venom and α-hemolysin from Staphylococcus aureus. Our results show that claramine neutralizes the toxicity of these pore-forming agents by preventing their interactions with cell membranes without perturbing their structures in a detectable manner. We thus demonstrate that the exogenous administration of an aminosterol can tune the properties of lipid membranes and protect cells from diverse biotoxins, including not just misfolded protein oligomers as previously shown but also biological protein-based toxins. Our results indicate that the investigation of regulators of the physicochemical properties of cell membranes offers novel opportunities to develop countermeasures against an extensive set of cytotoxic effects associated with cell membrane disruption. [1]
In conclusion, these findings collectively show that claramine is a potent molecule for protecting cells from membrane-disrupting toxins. As aminosterols have been shown to bind cell membranes, making them less negatively charged inducing a redistribution of cholesterol and ganglioside GM1 molecules and making them more resistant to indentation or oligomer embedding, this and other aminosterols may offer a unique therapeutic approach to protect the plasma membrane of cells from a wide range of toxic biomolecules implicit in numerous human pathologies. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6978 mL | 8.4891 mL | 16.9782 mL | |
| 5 mM | 0.3396 mL | 1.6978 mL | 3.3956 mL | |
| 10 mM | 0.1698 mL | 0.8489 mL | 1.6978 mL |