Physicochemical Properties
| Molecular Formula | C12H22O2 |
| Molecular Weight | 198.3019 |
| Exact Mass | 198.161 |
| CAS # | 150-84-5 |
| PubChem CID | 9017 |
| Appearance | Colorless to light yellow liquid |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 258.5±19.0 °C at 760 mmHg |
| Flash Point | 88.2±19.9 °C |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.442 |
| LogP | 4.28 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 14 |
| Complexity | 191 |
| Defined Atom Stereocenter Count | 0 |
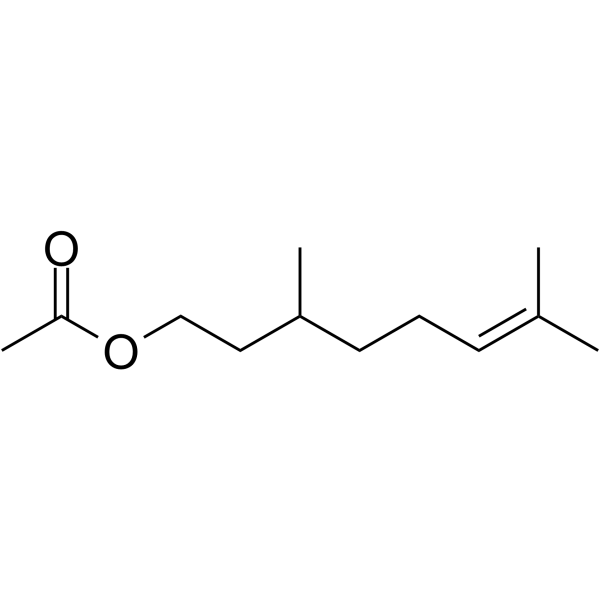
| SMILES | O(C(C([H])([H])[H])=O)C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])/C(/[H])=C(\C([H])([H])[H])/C([H])([H])[H] |
| InChi Key | JOZKFWLRHCDGJA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C12H22O2/c1-10(2)6-5-7-11(3)8-9-14-12(4)13/h6,11H,5,7-9H2,1-4H3 |
| Chemical Name | 3,7-dimethyloct-6-enyl acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo |
- Acetic Acid-Induced Writhing Test: Intragastric administration of CAT (100 and 200 mg/kg) significantly reduced the number of abdominal writhes in mice compared to the vehicle control. The dose of 200 mg/kg produced antinociception lasting up to 240 minutes post-administration. The estimated ED50 in this model was 74.42 mg/kg (95% CI: 67.65 – 81.88 mg/kg). [1] - Formalin-Induced Paw Licking Test: CAT (100 and 200 mg/kg, intragastric) significantly decreased the paw licking time in both the early (neurogenic, 0-5 min) and late (inflammatory, 20-25 min) phases of the formalin test in mice. [1] - Glutamate-Induced Paw Licking Test: CAT (100 and 200 mg/kg, intragastric) significantly inhibited nociceptive behavior (paw licking time) induced by intraplantar injection of glutamate in mice. [1] - Capsaicin-Induced Nociception Test: CAT (100 and 200 mg/kg, intragastric) produced a dose-dependent attenuation of nociceptive behavior (paw licking time) induced by intraplantar injection of capsaicin (a TRPV1 agonist) in mice. [1] - Menthol-Induced Nociception Test: CAT (100 and 200 mg/kg, intragastric) significantly inhibited nociceptive behavior induced by intraplantar injection of menthol (a TRPM8 agonist) in mice. [1] - Acidified Saline-Induced Nociception Test: CAT (100 and 200 mg/kg, intragastric) significantly inhibited nociceptive behavior induced by intraplantar injection of acidified saline (pH 2.04, an ASIC activator) in mice. [1] - PMA-Induced Nociception Test: CAT (200 mg/kg, intragastric) significantly reduced nociceptive behavior induced by intraplantar injection of PMA (a PKC activator). [1] - 8-Br-cAMP-Induced Nociception Test: CAT (200 mg/kg, intragastric) significantly reduced nociceptive behavior induced by intraplantar injection of 8-Br-cAMP (a PKA activator). [1] - Cinnamaldehyde-Induced Nociception Test: CAT (100 and 200 mg/kg, intragastric) did not significantly alter nociceptive behavior induced by intraplantar injection of cinnamaldehyde (a TRPA1 agonist), suggesting TRPA1 is not involved in its mechanism. [1] - Mechanism Investigation (L-arginine/NO pathway): Pretreatment with L-arginine did not reverse the antinociceptive effect of CAT (200 mg/kg) in the acetic acid writhing test, suggesting the L-arginine-NO pathway is not involved. [1] - Mechanism Investigation (KATP channels): Pretreatment with glibenclamide (a KATP channel blocker) partially reversed the antinociceptive effect of CAT (200 mg/kg) in the acetic acid writhing test, suggesting the involvement of KATP channels. [1] |
| Animal Protocol |
- Animals and Housing: Male Swiss mice (4 weeks old, 26-32 g) were used. They were housed in a temperature-controlled room (25 ± 2 °C) with a 12/12 h light/dark cycle and free access to food and water. Animals were fasted for 8 hours before experiments with free access to water. [1] - Drug Formulation and Administration: Citronellyl acetate is an oily substance. For administration, it was emulsified in a solution of 2% Tween 80 in distilled water using sonic agitation for 5 minutes. The drug was administered intragastrically (by gavage) at doses of 25, 50, 75, 100, and 200 mg/kg. The control group received an equivalent volume of the vehicle (2% Tween 80 in distilled water). [1] - Acetic Acid-Induced Writhing Test: Mice were pretreated with vehicle, CAT, or indomethacin (positive control, 10 mg/kg, p.o.). After a predetermined time (e.g., 60 minutes for dose-response), 0.6% acetic acid was injected intraperitoneally (10 µL/g body weight). Ten minutes later, the number of writhing episodes (abdominal constriction and hind leg extension) was counted over a 20-minute period. [1] - Paw Licking Tests (Formalin, Glutamate, etc.): Mice were pretreated with vehicle or CAT. After a set time (30 or 60 minutes), they received a 20 µL intraplantar injection into the right hind paw of one of the following: 1% formalin, glutamate (10 µmol/paw), capsaicin (1.6 µg/paw), menthol (1.2 µmol/paw), cinnamaldehyde (10 µmol/paw), acidified saline (2% acetic acid in saline, pH 2.04), PMA (500 pmol/paw), or 8-Br-cAMP (500 nmol/paw). The duration of paw licking was recorded over specific time windows post-injection (e.g., 0-5 min for formalin early phase, 20-25 min for late phase). [1] - Locomotor Activity Tests (Open Field and Rotarod): To rule out motor impairment, mice were treated with vehicle or CAT (25-200 mg/kg, p.o.). After 60 minutes, spontaneous locomotor activity was assessed for 5 minutes in an open field arena (number of quadrants crossed). Motor coordination was assessed using a rotarod apparatus (time spent on a rotating rod at 12 rpm, with a maximum of three attempts). [1] |
| Toxicity/Toxicokinetics |
- Acute Behavioral Toxicity: In the rotarod and open field tests, CAT at antinociceptive doses (up to 200 mg/kg, intragastric) did not significantly alter motor coordination or spontaneous locomotor activity in mice 60 minutes after administration, indicating no acute sedative or motor-impairing effects at these doses. [1] |
| References |
[1]. TRP and ASIC channels mediate the antinociceptive effect of citronellyl acetate. Chem Biol Interact. 2013 May 25;203(3):573-9. |
| Additional Infomation |
Citronellol acetate is a monoterpenoid that is the acetate ester of citronellol. It has been isolated from Citrus hystrix. It has a role as a plant metabolite. It is an acetate ester and a monoterpenoid. It is functionally related to a citronellol. Citronellyl acetate has been reported in Alpinia latilabris, Alpinia hainanensis, and other organisms with data available. Citronellyl acetate is a metabolite found in or produced by Saccharomyces cerevisiae. See also: Java citronella oil (part of). - Citronellyl acetate is a monoterpene ester found in plants like Eucalyptus citriodora. It is chemically similar to citronellol and citronellal, which have known biological activities. [1] - Prior to this study, CAT was known to possess fungicidal, larvicidal, bactericidal, insecticidal, and antitumor (pro-apoptotic in hepatoma cells) activities, but its antinociceptive properties had not been investigated. [1] - The study concludes that the systemic (intragastric) administration of CAT produces pronounced antinociceptive effects in mouse models of acute pain. Its mechanism involves, at least in part, the modulation of TRPV1, TRPM8, ASIC channels, glutamate receptors, KATP channels, and the inhibition of PKC and PKA signaling pathways. TRPA1 channels and the L-arginine-NO pathway do not appear to be involved. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~504.29 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (12.61 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (12.61 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (12.61 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.0429 mL | 25.2143 mL | 50.4286 mL | |
| 5 mM | 1.0086 mL | 5.0429 mL | 10.0857 mL | |
| 10 mM | 0.5043 mL | 2.5214 mL | 5.0429 mL |