Physicochemical Properties
| Molecular Formula | C19H14CL2FN3O |
| Molecular Weight | 390.2394 |
| Exact Mass | 389.049 |
| CAS # | 1193314-23-6 |
| PubChem CID | 44469321 |
| Appearance | Light brown to brown solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 621.3±55.0 °C at 760 mmHg |
| Flash Point | 329.6±31.5 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.738 |
| LogP | 4.61 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 26 |
| Complexity | 609 |
| Defined Atom Stereocenter Count | 2 |
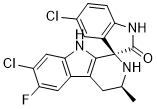
| SMILES | C[C@H]1CC2=C([C@]3(N1)C4=C(C=CC(=C4)Cl)NC3=O)NC5=CC(=C(C=C25)F)Cl |
| InChi Key | CKLPLPZSUQEDRT-WPCRTTGESA-N |
| InChi Code | InChI=1S/C19H14Cl2FN3O/c1-8-4-11-10-6-14(22)13(21)7-16(10)23-17(11)19(25-8)12-5-9(20)2-3-15(12)24-18(19)26/h2-3,5-8,23,25H,4H2,1H3,(H,24,26)/t8-,19+/m0/s1 |
| Chemical Name | (3R,3'S)-5,7'-dichloro-6'-fluoro-3'-methylspiro[1H-indole-3,1'-2,3,4,9-tetrahydropyrido[3,4-b]indole]-2-one |
| Synonyms | GNF609 KAE609, KAE 609,GNF 609 NITD609, NITD 609, NITD-609, KAE-609, GNF-609 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | At the highest studied concentration (10 μM), cipragamin (NITD609) is non-toxic to human foreskin fibroblasts (HFF) and inhibits Toxoplasma gondii, with a tachyzoite MIC90 of 5 μM and a MIC50 of 1 μM [1]. At 50 and 500 nM, the most effective inhibitor of early gametocyte growth is ciprgamin (NITD609). The formation of late gametocytes is inhibited by sipragamine in a dose-dependent manner [2]. With ICIC50 values ranging from 0.5 to 1.4 nM, cipragmin (NITD609) demonstrates strong efficacy against a panel of Plasmodium falciparum strains that have been culture-adapted. Like artesunate, sipargamine is active against all isolates of P. falciparum and P. vivax in the low nanomolar range (ICIC50 values consistently <10 nM) [3]. |
| ln Vivo | In malaria mouse models, dipergamin (NITD609) has good pharmacokinetic features and single-dose curative effectiveness. In all treated mice, cipramer (100 mg/kg) totally eradicated Plasmodium berghei infection; a single oral dose of 30 mg/kg produced a 50% partial cure [3]. |
| References |
[1]. Spiroindolone that inhibits PfATPase4 is a potent, cidal inhibitor of Toxoplasma gondii tachyzoites in vitro and in vivo. Antimicrob Agents Chemother. 2014;58(3):1789-92. [2]. The spiroindolone drug candidate NITD609 potently inhibits gametocytogenesis and blocks Plasmodium falciparum transmission to anopheles mosquito vector. Antimicrob Agents Chemother. 2012 Jul;56(7):3544-8. [3]. Spiroindolones, a potent compound class for the treatment of malaria. Science. 2010 Sep 3;329(5996):1175-80. |
| Additional Infomation | Cipargamin has been used in trials studying the treatment of Malaria, Cure Rate, and Plasmodium Falciparum Malaria. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~128.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3 mg/mL (7.69 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 30.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5625 mL | 12.8126 mL | 25.6253 mL | |
| 5 mM | 0.5125 mL | 2.5625 mL | 5.1251 mL | |
| 10 mM | 0.2563 mL | 1.2813 mL | 2.5625 mL |