Physicochemical Properties
| Molecular Formula | C22H30O5 |
| Molecular Weight | 374.4706 |
| Exact Mass | 374.209 |
| Elemental Analysis | C, 70.56; H, 8.08; O, 21.36 |
| CAS # | 94079-80-8 |
| Related CAS # | 95722-07-9 |
| PubChem CID | 5311044 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 591.1±50.0 °C at 760 mmHg |
| Flash Point | 203.4±23.6 °C |
| Vapour Pressure | 0.0±3.8 mmHg at 25°C |
| Index of Refraction | 1.569 |
| LogP | 1.94 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 27 |
| Complexity | 683 |
| Defined Atom Stereocenter Count | 6 |
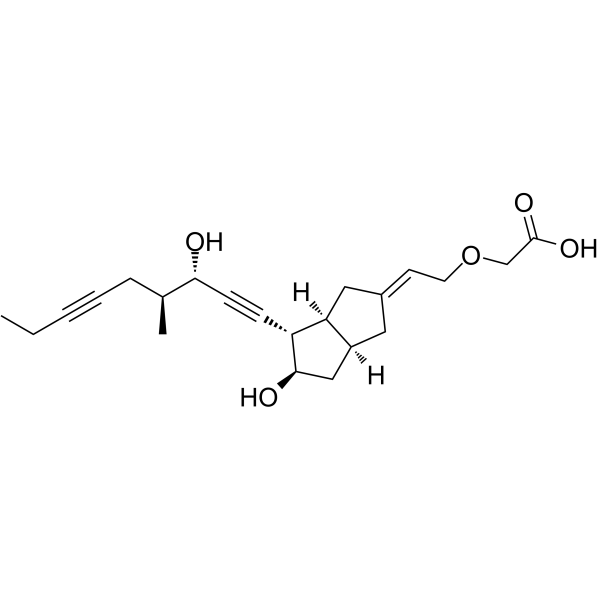
| SMILES | CCC#CC[C@H](C)[C@@H](C#C[C@@H]1[C@H]2C/C(=C/COCC(=O)O)/C[C@H]2C[C@H]1O)O |
| InChi Key | ARUGKOZUKWAXDS-SEWALLKFSA-N |
| InChi Code | InChI=1S/C22H30O5/c1-3-4-5-6-15(2)20(23)8-7-18-19-12-16(9-10-27-14-22(25)26)11-17(19)13-21(18)24/h9,15,17-21,23-24H,3,6,10-14H2,1-2H3,(H,25,26)/b16-9+/t15-,17-,18+,19-,20+,21+/m0/s1 |
| Chemical Name | 2-[(2E)-2-[(3aS,4S,5R,6aS)-5-hydroxy-4-[(3S,4S)-3-hydroxy-4-methylnona-1,6-diynyl]-3,3a,4,5,6,6a-hexahydro-1H-pentalen-2-ylidene]ethoxy]acetic acid |
| Synonyms | Cicaprost; 94079-80-8; Cicaprost [INN]; ZK 96480; ZK-96480; NE94J8CAMD; 2-[(2E)-2-[(3aS,4S,5R,6aS)-5-hydroxy-4-[(3S,4S)-3-hydroxy-4-methylnona-1,6-diynyl]-3,3a,4,5,6,6a-hexahydro-1H-pentalen-2-ylidene]ethoxy]acetic acid; (2-((2E,3aS,4S,5R,6aS)-Hexahydro-5-hydroxy-4-((3S,4S)-3-hydroxy-4-methyl-1,6-nonadiynyl)-2(1H)-pentalenylidene)ethoxy)acetic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | IP/prostacyclin receptor |
| ln Vitro | Cicaprost effectively inhibits the proliferation of human pulmonary artery smooth muscle cells (HPASMC) induced by fetal bovine serum (FBS). At 30 nM, cicaprost has notable antiproliferative action [2]. With the exception of SK-N-SH cells, caprocost promotes the synthesis of [3H] cyclic AMP (EC50 values of 1.5–22 nM) and [3H phosphoinositide (EC50 values of 49–457 nM) in all cells [3]. |
| ln Vivo | Cicaprost changes pain perception and inflammatory responses in mice lacking prostacyclin receptors. Intravenous infusion of cicaprost (1 μg/kg) results in a drop in blood pressure of approximately 30 mm Hg in anesthetized wild-type mice [4]. |
| Enzyme Assay | IL-33 has pleiotropic functions in immune responses and promotes the development of allergic diseases and asthma. IL-33 induces Th2 differentiation and enhances type 2 cytokine production by CD4+ T cells. However, the regulation of IL-33-driven type 2 cytokine responses is not fully defined. In this study, we investigated the effect of PGI2, a lipid mediator formed in the cyclooxygenase pathway of arachidonic acid metabolism, on naive CD4+ T cell activation, proliferation, and differentiation by IL-33. Using wild-type and PGI2 receptor (IP) knockout mice, we found that the PGI2 analog cicaprost dose-dependently inhibited IL-33-driven IL-4, IL-5, and IL-13 production by CD4+ T cells in an IP-specific manner. In addition, cicaprost inhibited IL-33-driven IL-2 production and CD25 expression by CD4+ T cells. Furthermore, IP knockout mice had increased IL-5 and IL-13 responses of CD4+ T cells to Alternaria sensitization and challenge in mouse lungs. Because IL-33 is critical for Alternaria-induced type 2 responses, these data suggest that PGI2 not only inhibits IL-33-stimulated CD4+ Th2 cell responses in vitro but also suppresses IL-33-induced Th2 responses caused by protease-containing allergens in vivo[1]. |
| Cell Assay |
Cell proliferation assay[2] Cell Types: HPASMC Tested Concentrations: 10 pM, 100 pM, 1 nM, 10 nM, 100 nM, 1 μM and 10 μM Incubation Duration: Experimental Results: Dose-dependent inhibition of proliferation with EC50 of 24.1 nM. |
| Animal Protocol |
Animal/Disease Models: wild-type (+/+) and IP−/− mice [4] Doses: 1 μg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results: Anesthetized wild-type mice induced hypotension of approximately 30 mm Hg, whereas no Even at an IP dose of 10 μg/kg, blood pressure changes occurred in IP-deficient mice. |
| References |
[1]. The PGI2 Analog Cicaprost Inhibits IL-33-Induced Th2 Responses, IL-2 Production, and CD25 Expression in Mouse CD4+ T Cells. J Immunol. 2018 Oct 1;201(7):1936-1945. [2]. Lucie H Clapp, et al. Differential effects of stable prostacyclin analogs on smooth muscle proliferation and cyclic AMP generation in human pulmonary artery. Am J Respir Cell Mol Biol. 2002 Feb;26(2):194-201. [3]. Kevin B S Chow, et al. Protein kinase A-dependent coupling of mouse prostacyclin receptors to Gi is cell-type dependent. Eur J Pharmacol. 2003 Aug 1;474(1):7-13. [4]. T Murata, et al. Altered pain perception and inflammatory response in mice lacking prostacyclin receptor. Nature. 1997 Aug 14;388(6643):678-82. |
| Additional Infomation | Cicaprost is a monoterpenoid. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6704 mL | 13.3522 mL | 26.7044 mL | |
| 5 mM | 0.5341 mL | 2.6704 mL | 5.3409 mL | |
| 10 mM | 0.2670 mL | 1.3352 mL | 2.6704 mL |