Physicochemical Properties
| Molecular Formula | C15H15CLN4O6S |
| Molecular Weight | 414.82 |
| Exact Mass | 414.04 |
| CAS # | 90982-32-4 |
| PubChem CID | 56160 |
| Appearance |
Crystals from butyl chloride White solid Colorless crystals |
| Density | 1.493 g/cm3 |
| Melting Point | 180-182°C |
| Index of Refraction | 1.598 |
| LogP | 3.37 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 27 |
| Complexity | 628 |
| Defined Atom Stereocenter Count | 0 |
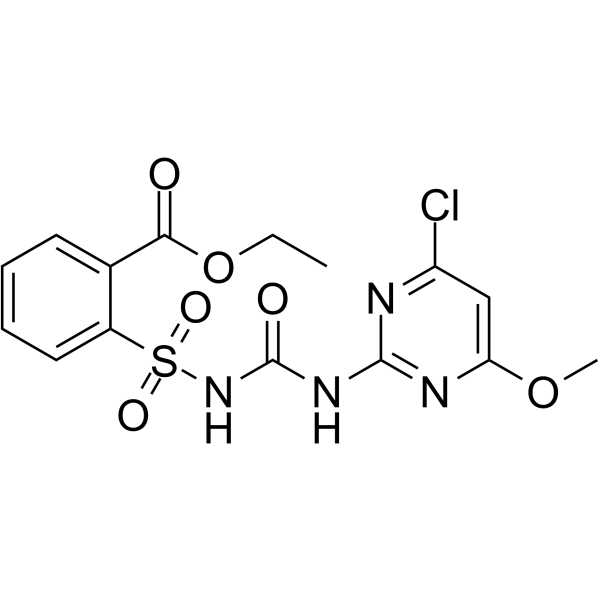
| SMILES | O=C(C1C(S(NC(NC2N=C(OC)C=C(Cl)N=2)=O)(=O)=O)=CC=CC=1)OCC |
| InChi Key | NSWAMPCUPHPTTC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C15H15ClN4O6S/c1-3-26-13(21)9-6-4-5-7-10(9)27(23,24)20-15(22)19-14-17-11(16)8-12(18-14)25-2/h4-8H,3H2,1-2H3,(H2,17,18,19,20,22) |
| Chemical Name | ethyl 2-[(4-chloro-6-methoxypyrimidin-2-yl)carbamoylsulfamoyl]benzoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Treatment with chlorimuron-ethyl (30 mg/kg) significantly damages CHL accumulation[1]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The compound /chlorimuron-ethyl/ is absorbed from the gastrointestinal tract and is eliminated equally in urine and feces with a biological half-life of about 50 hours. Chlorimuron-ethyl is distributed throughout the body, with the largest portions found in the liver. Metabolism / Metabolites Chlorimuron-ethyl was ...extensively metabolized by both male and female rats at the low and high dose. Excretion was monitored up to 168 hrs and the elimination of radioactivity was equal via the urine and feces for the low and high dose. The half-life is 50 hrs. /Five/... major metabolites .../were found/. (14)C Chlorimuron ethyl was readily absorbed by the roots of young intact corn seedlings and through the cut ends of excised leaves, but it was not readily absorbed by intact leaves. Under the conditions employed, (14)C-chlorimuron ethyl was metabolized at a moderate rate in both intact roots and excised leaves (ca 2.4 nmol/g fresh weight tissue/hr). Based upon HPLC analysis, (14)C-chlorimuron ethyl appeared to be metabolized by similar routes in both the roots and leaves. (14)C Chlorimuron ethyl and 10 radioactive metabolites were detected in the roots of corn 7 hr following herbicide treatment. (14)C-Chlorimuron ethyl and the following metabolites, listed in approximate order of their abundance, were isolated and characterized: chlorimuron ethyl (N-(4-chloro-6-methoxypyrimidine-2-yl)-N'-(2-ethoxycarbonylbenzene-sulfonyl)urea; N-(4-chloro-5-hydroxy-6-methoxypyrimidine-2-yl)-N'-(2-ethoxy carbonylbenzenesulfonyl)urea, 2-ethoxycarbonylbenzene sulfonamide, N-(4-(S-glutathionyl)-6-methoxypyrimidine-2-yl)-N'-(2-etho xycarbonyl benzenesulfonyl)urea, N-(4-(S-glutathionyl)-5-hydroxy-6-methoxypyrimidine-2-yl)- N'-(2- ethoxycarbonylbenzenesulfonyl)urea, N-(4-chloro-5-(O-beta-D-glucosyl)-6-methoxypyrimidine-2-yl)-N'ethoxy carbonylbenzenesulfonyl)urea, and N-(4-(S-cysteinyl)-6-methoxypyrimidine-2-yl)-N'-(2-ethoxycarbonyl- benzenesulfonyl)urea. Chlorimuron ethyl and these metabolites were purified by HPLC and were characterized by fast atom bombardment mass spectrometry. In addition to fast atom bombardment mass spectrometry, the following methods were used in the characterization of some metabolites: synthesis, hydrolysis with beta-glucosidase, analysis of hydrolysis products, electron impact MS, and proton nuclear magnetic resonance (400 MH). Biological Half-Life About 50 hours |
| Toxicity/Toxicokinetics |
Toxicity Data LC50 (rat) > 5,000 mg/m3/4h Interactions BAS 145 138 protected corn from injury due to low levels of chlorimuron ethyl. Inhibition of root growth was used to monitor injury. BAS 145 138 did not affect uptake, but it caused a two fold increase in the rate of chlorimuron ethyl metabolism in corn roots and shoots. The increase in chlorimuron ethyl metabolism was correlated positively to growth. Routes of metabolism that were accelerated in response to BAS 145 138 included hydroxylation of chlorimuron ethyl at the 5-position of the pyrimidine ring, the formation of the corresponding glucoside, the formation of two glutathione conjugates, and the formation of two unidentified metabolites. The most dramatic of the effects was on the formation of the glucoside of 5-hydroxychlorimuron ethyl. In the roots, the level of this metabolite was increased six fold in response to BAS 145 138. Part of this increase was due to an elevation in the in vivo rate of glucosylation of 5-hydroxychlorimuron ethyl. BAS 145 138 did not alter the qualitative routes of chlorimuron ethyl metabolism, nor did it appear to affect the levels or catalytic properties of acetolactate synthase, the target enzyme of chlorimuron ethyl. 5-Hydroxychlorimuron ethyl was 152 times less effective as an inhibitor of acetolactate synthase than chlorimuron ethyl. The glucoside of 5-hydroxychlorimuron ethyl was hydrolyzed during the acetolactate synthase assay; therefore, an I50 value for this metabolite was not obtained. Non-Human Toxicity Values LC50 Rat inhalation >5 mg/L/4 hr LD50 Rabbit dermal > 2000 mg/kg LD50 Rat oral >5000 mg/kg |
| References |
[1]. Effects of Herbicide Chlorimuron-Ethyl on Physiological Mechanisms in Wheat (Triticum Aestivum). Ecotoxicol Environ Saf. 2006 Jun;64(2):190-7. |
| Additional Infomation |
Chlorimuron ethyl appears as colorless crystals. Used as an herbicide. Chlorimuron-ethyl is an ethyl ester resulting from the formal condensation of the carboxy group of chlorimuron with ethanol. A proherbicide for chloimuron, it is used as herbicide for the control of broad-leaved weeds in peanuts, soya beans, and other crops. It has a role as a proherbicide, an EC 2.2.1.6 (acetolactate synthase) inhibitor and an agrochemical. It is a sulfamoylbenzoate, a N-sulfonylurea, an aromatic ether, an ethyl ester, an organochlorine pesticide and a member of pyrimidines. It is functionally related to a chlorimuron. It is a conjugate acid of a chlorimuron-ethyl(1-). Mechanism of Action Sulfonylurea class herbicide that inhibits acetolactate synthase, which regulates plant growth. Branched chain amino acid synthesis (ASL or AHAS) inhibitor. Acts by inhibiting biosynthesis of the essential amino acids valine and isoleucine, hence stopping cell division and plant growth. Crop selectivity derives from plant metabolism both by homoglutathione conjugation and by de-esterification. ...Acetolactate synthase /is/ the target enzyme of chlorimuron ethyl. |
Solubility Data
| Solubility (In Vitro) | DMSO: 250 mg/mL (602.67 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4107 mL | 12.0534 mL | 24.1068 mL | |
| 5 mM | 0.4821 mL | 2.4107 mL | 4.8214 mL | |
| 10 mM | 0.2411 mL | 1.2053 mL | 2.4107 mL |