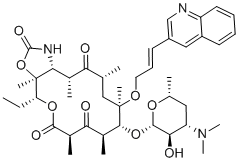
Cethromycin (also known as ABT-773, Abbott-195773, A-195773) is a ketolide antibiotic with a similar mechanism of action to telithromycin with an apparently better safety profile. Cethromycin displays in vitro activity against selected gram-positive, gram-negative, and atypical bacteria. The proposed indication of cethromycin is treatment of mild to moderate community-acquired bacterial pneumonia in patients. Cethromycin has an orphan drug designation for tularemia, plague, and anthrax prophylaxis. Preliminary data on adverse effects suggest that cethromycin is safe and gastrointestinal adverse effects appear to be dose-related.
Physicochemical Properties
| Molecular Formula | C42H59N3O10 |
| Molecular Weight | 765.93196 |
| Exact Mass | 765.42 |
| Elemental Analysis | C, 65.86; H, 7.76; N, 5.49; O, 20.89 |
| CAS # | 205110-48-1 |
| PubChem CID | 156417 |
| Appearance | Solid powder |
| Density | 1.22g/cm3 |
| Boiling Point | 927.1ºC at 760mmHg |
| Flash Point | 514.5ºC |
| Vapour Pressure | 0mmHg at 25°C |
| Index of Refraction | 1.578 |
| LogP | 5.438 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 55 |
| Complexity | 1410 |
| Defined Atom Stereocenter Count | 13 |
| SMILES | CC[C@H]1OC(=O)[C@H](C)C(=O)[C@H](C)[C@@H](O[C@@H]2O[C@H](C)C[C@H](N(C)C)[C@H]2O)[C@](C)(OC/C=C/C2C=NC3=CC=CC=C3C=2)C[C@@H](C)C(=O)[C@H](C)[C@H]2NC(O[C@]12C)=O |
| InChi Key | PENDGIOBPJLVBT-ONLVEXIXSA-N |
| InChi Code | InChI=1S/C42H59N3O10/c1-11-32-42(8)36(44-40(50)55-42)25(4)33(46)23(2)21-41(7,51-18-14-15-28-20-29-16-12-13-17-30(29)43-22-28)37(26(5)34(47)27(6)38(49)53-32)54-39-35(48)31(45(9)10)19-24(3)52-39/h12-17,20,22-27,31-32,35-37,39,48H,11,18-19,21H2,1-10H3,(H,44,50)/b15-14+/t23-,24-,25+,26+,27-,31+,32-,35-,36-,37-,39+,41-,42-/m1/s1 |
| Chemical Name | (3aS,4R,7R,9R,10R,11R,13R,15R,15aR)-10-(((2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-4-ethyl-3a,7,9,11,13,15-hexamethyl-11-(((E)-3-(quinolin-3-yl)allyl)oxy)octahydro-2H-[1]oxacyclotetradecino[4,3-d]oxazole-2,6,8,14(1H,7H,9H)-tetraone |
| Synonyms | Cethromycin; ABT-773; ABT 773; ABT773; A-195773; A 195773; A195773; Abbott-195773; Abbott195773; Abbott 195773 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
In vitro, cethromycin has marked activity against these pathogens with the ability to overcome both efflux and methylation mechanisms of resistance in S. pneumoniae.[1] The antibacterial activity of cethromycin is mediated through dual mechanisms of action: binding to the bacterial target, the 23S rRNA of the 50S subunit of the ribosome to prevent the translation of bacterial messenger RNAs into new proteins and interacting with partially assembled 50S subunit precursors to inhibit the complete formation of bacterial ribosomes.Cethromycin is able to overcome methylation-mediated resistance via a second point of contact with the ribosome . In addition, the enhanced binding of cethromycin is helpful in overcoming bacterial resistance mediated via efflux mechanisms, resulting in increases in antibacterial activity compared to both macrolide agents and the marketed ketolide agent, telithromycin . Cethromycin retains activity against clinical isolates of telithromycin-resistant S. pneumoniae, a phenomenon believed to be the result of the enhanced binding kinetics.[1] The reduced activity of cethromycin against enteric Gram-negative bacteria should limit the collateral damage often seen with quinolone treatment yet preserve the favorable activity against susceptible and resistant CAP-causative pathogens.[1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Cethromycin displays non-linear absorption kinetics. In healthy adults administered 150 mg cethromycin orally once daily for five doses, the calculated Cmax, Tmax, and AUC0-24 values were 0.181 ± 0.084 μg/ml, 2.01 ± 1.30 hrs, and 0.902 ± 0.469 μg\*h/ml, respectively. Similarly, the corresponding values for a 300 mg dose were 0.500 ± 0.168 μg/ml, 2.09 ± 0.03 hrs, and 3.067 ± 1.205 μg\*h/ml, respectively. In another study using a single oral dose of 150 mg cethromycin, the Cmax was 318 ± 161 ng/ml, the Tmax was 1.79 ± 0.50, the AUC0-24 was 1596 ± 876 ng\*h/ml, and the AUC0-∞ was 1662 ± 907 ng\*h/ml. Cethromycin is primarily excreted by the biliary route, with 87.2% of an initial dose recovered in feces and only 7.0% in urine. Unchanged cethromycin accounted for 35.7% of the radioactivity recovered in feces and an N-desmethyl metabolite for 39.8%; the remaining radioactivity was approximately evenly divided between three minor metabolites and a group of uncharacterized additional products. Cethromycin given in five 150 mg oral doses had an apparent volume of distribution at the terminal elimination phase of 1433 ± 843 L, and an apparent steady-state volume of distribution of 1453 ± 997 L. The corresponding values for a 300 mg dose was 761 ± 293 L and 769 ± 272 L. Cethromycin is known to accumulate in the epithelial lining fluid and alveolar cells, as well as within polymorphonuclear leukocytes. Cethromycin clearance in patients receiving a once-daily oral dose of 300 mg is reported to be approximately 63 L/h. Metabolism / Metabolites Extensive studies of cethromycin metabolism have not been conducted, although one study identified seven metabolites within feces of patients administered a single 150 mg oral dose. The major recovered products were unchanged cethromycin and an inactive N-desmethyl metabolite. It is likely that most of the metabolism occurs in the liver and is mediated, at least in part, by CYP3A4. Biological Half-Life Cethromycin given in five oral doses of 150 or 300 mg has a plasma half-life of 4.85 ± 1.10 and 4.94 ± 0.66 hrs, respectively. A single oral dose of 150 mg produced a measured half-life of 5.66 ± 0.77 hrs. |
| Toxicity/Toxicokinetics |
Protein Binding Cethromycin displays 86.7 to 95.6% human plasma protein binding over a range of concentrations between 0.1 to 30.0 μg/ml. |
| References |
[1]. Cethromycin versus clarithromycin for community-acquired pneumonia: comparative efficacyand safety outcomes from two double-blinded, randomized, parallel-group, multicenter, multinational noninferiority studies. Antimicrob Agents Chemot.2012 Apr;56(4):2037-47. |
| Additional Infomation |
Cethromycin is a 3-keto (ketolide) derivative of erythromycin A with an 11,12-carbamate group and an O-6-linked aromatic ring system. Cethromycin represents a joint development effort by Abbott Laboratories, Taisho Pharmaceuticals, and Advanced Life Sciences, intended to be marketed under the trade name Restanza for the treatment of community-acquired pneumonia. However, after completing phase III clinical trials, it was deemed safe but not sufficiently efficacious by the FDA. Since this time, cethromycin has received FDA orphan drug designations for the prophylactic treatment of anthrax inhalation, plague due to Yersinia pestis, and tularemia due to Francisella tularensis. It has also been investigated, by itself or together with [zoliflodacin], for the treatment of gonorrhea, and was recently suggested as a possible treatment for liver-stage Plasmodium sporozoite infection. Drug Indication Cethromycin currently has no FDA-approved indications; it was granted orphan drug designation for the prophylactic treatment of inhalation anthrax in 2007 and for the prophylactic treatment of both plague due to _Yersinia pestis_ and tularemia due to _Francisella tularensis_ in 2009. Mechanism of Action Respiratory tract infections can be caused by numerous strains of bacteria, requiring careful consideration of treatment and antibiotics effective against a broad spectrum of potential pathogens. Cethromycin, like other macrolide antibiotics, binds to the 23S rRNA of the 50S subunit of the bacterial ribosome. This binding, primarily mediated through regions II and V of the rRNA, occludes the peptide exit tunnel and inhibits bacterial protein synthesis. In addition, cethromycin is capable of binding to ribosomal intermediates during ribosome biogenesis, inhibiting the formation of functional 70S bacterial ribosomes. Due to the sequence and structural similarity of ribosomes between species, cethromycin displays broad-spectrum activity against diverse Gram-positive, Gram-negative, and atypical bacteria. Pharmacodynamics Cethromycin binds to the 50S subunit of the bacterial ribosome to inhibit both ribosome assembly and bacterial protein synthesis. Adverse effects such as diarrhea, nausea, vomiting, and headache may be due to off-target inhibition of molecules within mammalian cells. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~130.56 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3056 mL | 6.5280 mL | 13.0560 mL | |
| 5 mM | 0.2611 mL | 1.3056 mL | 2.6112 mL | |
| 10 mM | 0.1306 mL | 0.6528 mL | 1.3056 mL |