Physicochemical Properties
| Molecular Formula | C26H30D3FNNAO5 |
| Molecular Weight | 484.55 |
| Exact Mass | 482.232 |
| CAS # | 916314-45-9 |
| PubChem CID | 23684377 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 4.88 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 34 |
| Complexity | 626 |
| Defined Atom Stereocenter Count | 2 |
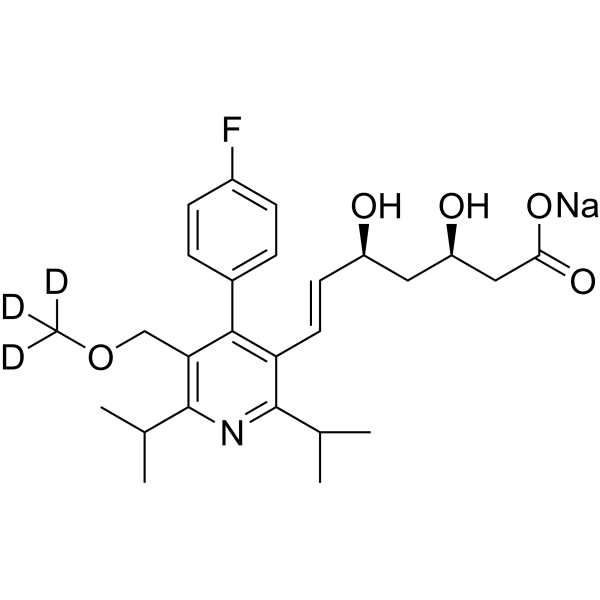
| SMILES | [2H]C([2H])([2H])OCC1=C(C(=C(N=C1C(C)C)C(C)C)/C=C/[C@H](C[C@H](CC(=O)[O-])O)O)C2=CC=C(C=C2)F.[Na+] |
| InChi Key | GPUADMRJQVPIAS-ARMPMJDMSA-M |
| InChi Code | InChI=1S/C26H34FNO5.Na/c1-15(2)25-21(11-10-19(29)12-20(30)13-23(31)32)24(17-6-8-18(27)9-7-17)22(14-33-5)26(28-25)16(3)4;/h6-11,15-16,19-20,29-30H,12-14H2,1-5H3,(H,31,32);/q;+1/p-1/b11-10+;/t19-,20-;/m1./s1/i5D3; |
| Chemical Name | sodium;(E,3R,5S)-7-[4-(4-fluorophenyl)-2,6-di(propan-2-yl)-5-(trideuteriomethoxymethyl)pyridin-3-yl]-3,5-dihydroxyhept-6-enoate |
| Synonyms | Cerivastatin Sodium-d3 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, primarily as quantitative tracers during drug development. Studies involving humans using deuterium-labeled drugs have shown that these compounds may have certain advantages over their non-deuterium-labeled counterparts. Deuterated drugs have attracted attention due to their potential to affect the pharmacokinetic and metabolic profiles of drugs. Deuttetrabenazine is the first deuterated drug approved by the U.S. Food and Drug Administration. Deuttetrabenazine is indicated for the treatment of chorea associated with Huntington's disease as well as tardive dyskinesia. Ongoing clinical trials indicate that many other deuterated compounds are being evaluated for use as therapeutics in humans, not just as research tools. [1] Cerivastatin sodium (5-50 ng/mL; 3 days; MDA-MB-231 cells) treatment induces a dose-dependent decrease in the proliferation of MDA-MB-231 cells (up to 40% inhibition at 25 ng/mL) [2]. Cerivastatin sodium (25 ng/mL; 18-36 hours; MDA-MB-231 cells) treatment induced cell cycle arrest at the G1/S phase after 36 hours of treatment. This arrest was not observed with a shorter incubation time (18 hours) [2]. Cerivastatin sodium (25 ng/mL; 18 hours; MDA-MB-231 cells) treatment induced a significant increase in p21Waf1/Cip1 levels [2]. Cerivastatin sodium (25 ng/mL; 12 hours; MDA-MB-231 cells) treatment increased p21 transcripts in MDA-MB-231 cells [2]. Cerivastatin sodium (10-25 ng/mL; 18 hours) inhibited the invasion of MDA-MB-231 cells by Matrigel [2]. Cerivastatin sodium (25 ng/mL; 18-36 hours) delocalizes RhoA and Ras from the cell membrane to the cytosol and induces morphological changes[2]. Cerivastatin sodium (25 ng/mL; 4-36 hours) induces NFκB inactivation in a RhoA inhibition-dependent manner, resulting in decreased expression of urokinase and metalloproteinase 9, while increasing IκB[2]. |
| ln Vivo | Cerivastatin sodium is well absorbed, reaching maximum plasma concentrations within 1-3 hours after oral administration. In the circulation, cerivastatin sodium is highly bound to plasma proteins (99.5%), with an elimination half-life of 2-4 hours. Cerivastatin sodium is metabolized primarily in the liver to three polar metabolites. Two of these metabolites are active, but to a lesser extent than the parent drug, and the third metabolite is inactive. Plasma concentrations of all metabolites are substantially lower than those of the parent drug. Metabolites are eliminated via urine (20-25%) and feces (66-73%), with essentially no excretion of the parent compound[3]. |
| References |
[1]. Impact of Deuterium Substitution on the Pharmacokinetics of Pharmaceuticals. Ann Pharmacother. 2019 Feb;53(2):211-216. [2]. Cerivastatin, an inhibitor of HMG-CoA reductase, inhibits the signaling pathways involved in the invasiveness and metastatic properties of highly invasive breast cancer cell lines: an in vitro study. Carcinogenesis. 2001 Aug;22(8):1139-48. [3]. Cerivastatin, a New Potent Synthetic HMG Co-A Reductase Inhibitor: Effect of 0.2 mg Daily in Subjects With Primary Hypercholesterolemia. J Cardiovasc Pharmacol Ther. 1997 Jan;2(1):7-16. [4]. Withdrawal of cerivastatin from the world market. Curr Control Trials Cardiovasc Med. 2001;2(5):205-207. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0638 mL | 10.3189 mL | 20.6377 mL | |
| 5 mM | 0.4128 mL | 2.0638 mL | 4.1275 mL | |
| 10 mM | 0.2064 mL | 1.0319 mL | 2.0638 mL |