Physicochemical Properties
| Molecular Formula | C25H24N8O7S2 |
| Molecular Weight | 612.63746 |
| Exact Mass | 612.121 |
| CAS # | 70797-11-4 |
| Related CAS # | Cefpiramide sodium;74849-93-7 |
| PubChem CID | 636405 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.75 g/cm3 |
| Melting Point | 213-215° (dec) |
| Index of Refraction | 1.821 |
| LogP | 1.046 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 42 |
| Complexity | 1270 |
| Defined Atom Stereocenter Count | 3 |
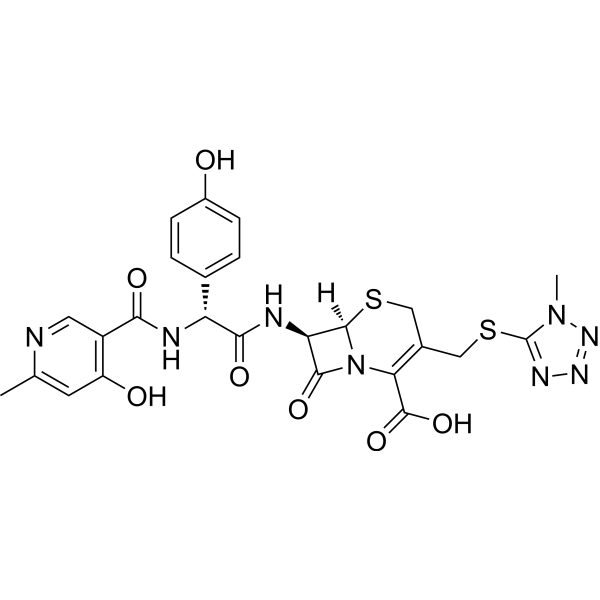
| SMILES | CC1=CC(C(C(N[C@@H](C(N[C@@H]2C(N3C(C(O)=O)=C(CSC4=NN=NN4C)CS[C@H]23)=O)=O)C5=CC=C(O)C=C5)=O)=CN1)=O |
| InChi Key | PWAUCHMQEXVFJR-PMAPCBKXSA-N |
| InChi Code | InChI=1S/C25H24N8O7S2/c1-11-7-16(35)15(8-26-11)20(36)27-17(12-3-5-14(34)6-4-12)21(37)28-18-22(38)33-19(24(39)40)13(9-41-23(18)33)10-42-25-29-30-31-32(25)2/h3-8,17-18,23,34H,9-10H2,1-2H3,(H,26,35)(H,27,36)(H,28,37)(H,39,40)/t17-,18-,23-/m1/s1 |
| Chemical Name | (6R,7R)-7-[[(2R)-2-(4-hydroxyphenyl)-2-[(6-methyl-4-oxo-1H-pyridine-3-carbonyl)amino]acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The majority of non-fermenting Gram-negative bacilli and enterococci are effectively inhibited by cefpiramide (0–2048 µg/mL; 16–18 hours) (32 µg/mL, 97% inhibition) [1]. Cefpiramide, with MIC[50] and MIC[90] values of 4 µg/mL and 16 µg/mL, respectively, shows considerable effectiveness against Pseudomonas aeruginosa (0-2048 µg/mL; 16-18 hours)[1]. |
| ln Vivo | Cefpiramide (25 mg/kg; IV; once) has proven efficacy against Streptococcus pneumoniae in experimental pneumococcal meningitis [2]. Cefpiramide (25 mg/kg; IV; once) decreases bacteria in cerebrospinal fluid by more than 10 [4] CFU/ml [2]. |
| Cell Assay |
Cell Viability Assay[1] Cell Types: 761 bacterial isolates (35 different common bacterial pathogens) Tested Concentrations: 0-2048 µg/mL Incubation Duration: 16-18 hrs (hours) Experimental Results: Demonstrated broad spectrum antimicrobial activity. |
| Animal Protocol |
Animal/Disease Models: New Zealand white rabbit (2-3 kg) [2]. Doses: 25 mg/kg Route of Administration: intravenous (iv) (iv)injection; once Experimental Results: Anti-Streptococcus pneumoniae. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Rapidly absorbed following intramuscular injection. Biological Half-Life 4.44 hours |
| References |
[1]. Cefpiramide: comparative in-vitro activity and beta-lactamase stability. J Antimicrob Chemother. 1985 Sep;16(3):315-25. [2]. Pharmacokinetics and bacteriological efficacies of apalcillin and cefpiramide in experimental pneumococcal meningitis. Antimicrob Agents Chemother. 1984 Oct;26(4):578-9. |
| Additional Infomation |
Cefpiramide is a third-generation cephalosporin antibiotic with [(1-methyl-1H-tetrazol-5-yl)sulfanyl]methyl and (R)-2-{[(4-hydroxy-6-methylpyridin-3-yl)carbonyl]amino}-2-(4-hydroxyphenyl)acetamido groups at positions 3 and 7, respectively, of the cephem skeleton. It has a broad spectrum of antibacterial activity. It has a role as an antibacterial drug. It is a cephalosporin and a carboxylic acid. It is a conjugate acid of a cefpiramide(1-). Cefpiramide is a third-generation cephalosporin antibiotic. Cefpiramide is a third-generation, semi-synthetic, beta-lactam cephalosporin antibiotic with antibacterial activity. Cefpiramide binds to penicillin-binding proteins (PBPs), transpeptidases that are responsible for crosslinking of peptidoglycan. By preventing crosslinking of peptidoglycan, cell wall integrity is lost and cell wall synthesis is halted. Drug Indication For treatment of severe infections caused by susceptible bacteria such as P. aeruginosa. Mechanism of Action The bactericidal activity of cefpiramide results from the inhibition of cell wall synthesis via affinity for penicillin-binding proteins (PBPs). Pharmacodynamics Cefpiramide is a cephalosporin active against Pseudomonas aeruginosa. It has a broad spectrum of antibacterial activity. Cefpiramide works by inhibiting bacterial cell wall biosynthesis. The plasma half-lives of cefpiramide in rabbits, dogs, and rhesus monkeys were much longer than those of cefoperazone and cefazolin. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6323 mL | 8.1614 mL | 16.3228 mL | |
| 5 mM | 0.3265 mL | 1.6323 mL | 3.2646 mL | |
| 10 mM | 0.1632 mL | 0.8161 mL | 1.6323 mL |