Physicochemical Properties
| Molecular Formula | C19H25CLN6O5S2 |
| Molecular Weight | 517.022000074387 |
| Exact Mass | 516.101638 |
| CAS # | 107648-79-3 |
| Related CAS # | Cefepime;88040-23-7; Cefepime Dihydrochloride Monohydrate;123171-59-5; Cefepime chloride;107648-79-3; 107648-78-2 (sulfate); 103296-32-8 (compound E); 123171-59-5 (2HCl hydrate);107648-80-6 (2HCl) |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 33 |
| Complexity | 874 |
| Defined Atom Stereocenter Count | 2 |
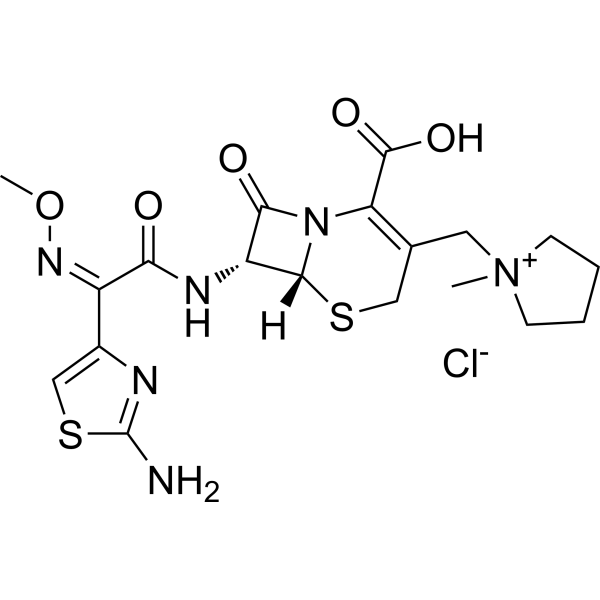
| SMILES | [Cl-].S1CC(=C(C(=O)O)N2C([C@H]([C@@H]12)NC(/C(/C1=CSC(N)=N1)=N\OC)=O)=O)C[N+]1(C)CCCC1 |
| InChi Key | MMRINLZOZVAPDZ-LSGRDSQZSA-N |
| InChi Code | InChI=1S/C19H24N6O5S2.ClH/c1-25(5-3-4-6-25)7-10-8-31-17-13(16(27)24(17)14(10)18(28)29)22-15(26)12(23-30-2)11-9-32-19(20)21-11;/h9,13,17H,3-8H2,1-2H3,(H3-,20,21,22,26,28,29);1H/b23-12-;/t13-,17-;/m1./s1 |
| Chemical Name | (6R,7R)-7-[[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-methoxyiminoacetyl]amino]-3-[(1-methylpyrrolidin-1-ium-1-yl)methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid chloride |
| Synonyms | Cefepime (chloride); 107648-79-3; 1-(((6R,7R)-7-((Z)-2-(2-Aminothiazol-4-yl)-2-(methoxyimino)acetamido)-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl)methyl)-1-methylpyrrolidin-1-ium chloride; orb1744769; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PBP/penicillin-binding proteins |
| ln Vitro | Cefepime chloride binds to proteins that bind penicillin to produce its antibacterial action [2]. |
| ln Vivo | Cefepime chloride (80 mg/kg; intraperitoneally) considerably lengthened the half-life and the mice survived [4]. |
| Animal Protocol |
Animal/Disease Models: Male CD-1 mice [4] Doses: 80 mg/kg Route of Administration: intraperitoneal (ip) injection Experimental Results: The half-life of cefepime was Dramatically prolonged in all mice treated with cisplatin 18-22 mg/kg and 26 mg /kg all survived during pretreatment. kg cisplatin Dramatically diminished survival rate, and the half-life of cefepime was not Dramatically longer than 18 mg/kg cisplatin. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Healthy adult male volunteers (n=9) given a single intravenous infusion of 500 mg, 1 g, and 2 g of cefepime had a corresponding Cmax of 39.1, 81.7 and 163.9 μg/mL, and a corresponding AUC of 70.8, 148.5 and 284.8 h⋅μg/mL. On the other hand, healthy adult male volunteers given a single intramuscular infusion of 500 mg, 1 g, and 2 g of cefepime had a corresponding Cmax of 13.9, 29.6 and 57.5 μg/mL, a corresponding AUC of 60, 137 and 262 h⋅μg/mL, and a corresponding Tmax of 1.4, 1.6 and 1.5 h. A study in healthy adult male volunteers (n=7) that received clinically relevant doses for 9 days suggests that cefepime is not accumulated in the body. Between 250 mg and 2 g, cefepime follows a linear pharmacokinetic model, and the absolute bioavailability of cefepime in pediatric patients (n=8) given an intramuscular dose of 50 mg/kg was 82.3%. Cefepime is mainly eliminated by the kidneys, and most of it is excreted unchanged. Approximately 85% of cefepime administered to normal subjects is excreted unchanged in urine. Less than 1% of the administered dose is recovered from urine as N-methylpyrrolidine (NMP), 6.8% as NMP-N-oxide, and 2.5% as an epimer. Dosage adjustments are required in patients with renal dysfunction or those undergoing hemodialysis, due to the importance of renal excretion in eliminating cefepime. The average steady-state volume of distribution of cefepime is 18.0 L. In pediatric patients, the average steady-state volume of distribution is 0.3 L/kg. Cefepime has a total body clearance of 120 mL/min in healthy volunteers, and in pediatric patients, the average total body clearance is 3.3 mL/min/kg. In geriatric patients (65 years of age and older) and patients with abnormal renal function, cefepime total body clearance decreases proportionally with creatinine clearance. Metabolism / Metabolites Less than 1% of cefepime is metabolized in the liver. Cefepime is metabolized to N-methylpyrrolidine (NMP), which then undergoes rapid oxidation to form NMP-N-oxide, a more stable compound. NMP-N-oxide is the predominant metabolite of cefepime, while NMP and the 7-epimer of cefepime are minor byproducts. It has been suggested that flavin-containing mixed-function oxygenase mediates the oxidation of NMP to NMP-N-oxide. Biological Half-Life Healthy adult male volunteers (n=9) given cefepime had an average half-life of 2 hours. In patients requiring hemodialysis, the average half-life was 13.5 hours, and in patients requiring continuous peritoneal dialysis, the average half-life was 19 hours. Absorption Healthy adult male volunteers (n=9) given a single intravenous infusion of 500 mg, 1 g, and 2 g of cefepime had a corresponding Cmax of 39.1, 81.7 and 163.9 μg/mL, and a corresponding AUC of 70.8, 148.5 and 284.8 h⋅μg/mL. On the other hand, healthy adult male volunteers given a single intramuscular infusion of 500 mg, 1 g, and 2 g of cefepime had a corresponding Cmax of 13.9, 29.6 and 57.5 μg/mL, a corresponding AUC of 60, 137 and 262 h⋅μg/mL, and a corresponding Tmax of 1.4, 1.6 and 1.5 h. A study in healthy adult male volunteers (n=7) that received clinically relevant doses for 9 days suggests that cefepime is not accumulated in the body. Between 250 mg and 2 g, cefepime follows a linear pharmacokinetic model, and the absolute bioavailability of cefepime in pediatric patients (n=8) given an intramuscular dose of 50 mg/kg was 82.3%. Route of Elimination Cefepime is mainly eliminated by the kidneys, and most of it is excreted unchanged. Approximately 85% of cefepime administered to normal subjects is excreted unchanged in urine. Less than 1% of the administered dose is recovered from urine as N-methylpyrrolidine (NMP), 6.8% as NMP-N-oxide, and 2.5% as an epimer. Dosage adjustments are required in patients with renal dysfunction or those undergoing hemodialysis, due to the importance of renal excretion in eliminating cefepime. Volume of Distribution The average steady-state volume of distribution of cefepime is 18.0 L. In pediatric patients, the average steady-state volume of distribution is 0.3 L/kg. Clearance Cefepime has a total body clearance of 120 mL/min in healthy volunteers, and in pediatric patients, the average total body clearance is 3.3 mL/min/kg. In geriatric patients (65 years of age and older) and patients with abnormal renal function, cefepime total body clearance decreases proportionally with creatinine clearance. Protein Binding The serum protein binding of cefepime is approximately 20%, independent of its concentration in serum. Metabolism / Metabolites Less than 1% of cefepime is metabolized in the liver. Cefepime is metabolized to N-methylpyrrolidine (NMP), which then undergoes rapid oxidation to form NMP-N-oxide, a more stable compound. NMP-N-oxide is the predominant metabolite of cefepime, while NMP and the 7-epimer of cefepime are minor byproducts. It has been suggested that flavin-containing mixed-function oxygenase mediates the oxidation of NMP to NMP-N-oxide. Biological Half-Life Healthy adult male volunteers (n=9) given cefepime had an average half-life of 2 hours. In patients requiring hemodialysis, the average half-life was 13.5 hours, and in patients requiring continuous peritoneal dialysis, the average half-life was 19 hours. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Although no published information is available on the use of cefepime during breastfeeding, the levels in breastmilk appear to be low and cephalosporins are generally not be expected to cause serious adverse effects in breastfed infants. Occasionally disruption of the infant's gastrointestinal flora, resulting in diarrhea or thrush have been reported with cephalosporins, but these effects have not been adequately evaluated. Cefepime is acceptable in nursing mothers. The combination of cefepime and enmetazobactam has not been studied in humans during breasteeding, but the same adverse effects should apply. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding The serum protein binding of cefepime is approximately 20%, independent of its concentration in serum. Toxicity Summary In the event of a suspected overdose, the clinician should discontinue the drug or adjust the dose. Determining if the symptoms result from an actual cefepime overdose or preexisting comorbidities may be difficult. If suspicion is high or symptoms do not subside after dose adjusting or discontinuing, blood and cerebral spinal fluid levels should be obtained to evaluate if the toxicity stems from elevated cefepime levels. Cefepime-induced neurotoxicity (CIN) can lead to generalized periodic discharges and a triphasic wave pattern on EEG. Dialysis may be necessary in severe cases. Adverse Effects Cefepime is usually well tolerated by both adult and pediatric patients. The most common adverse effects in adults are diarrhea and rash. The most common adverse effects in the pediatric population are fevers, diarrhea, and rash. There are multitudes of other less common adverse effects listed according to the system affected: * Neurological: headache, fever, and neurotoxicity * Gastrointestinal: nausea, vomiting, abdominal pain, hepatic injury, colitis including pseudomembranous colitis, oral candidiasis * Genitourinary: vaginitis, renal injury * Dermatological: local site injection irritation, pruritus, urticaria, Stevens-Johnson syndrome, and erythema multiforme * Hematological: positive Coombs test without hemolysis, pancytopenia, and anaplastic anemia Adverse effects typically reverse upon removal of the medication. Neurotoxicity is a serious, life-threatening adverse effect that deserves special mention. Symptoms can include altered mental status, encephalopathy, seizures, myoclonus, hallucinations, coma, and stroke-like symptoms. The onset of symptoms is typically 4 days after starting cefepime. Risk factors include renal failure (creatinine ≤60 mL/min), older patients, critically ill patients in ICU, strokes, Alzheimer disease, brain malignancy, history of seizures, and a compromised blood-brain barrier (BBB). The theorized mechanism is that cefepime can cross the BBB and antagonize gamma-aminobutyric acid receptors. Treatment consists of stopping the drug, seizure management with benzodiazepines, or renal replacement therapy in severe refractory cases. The clinical team must monitor and adjust dosing with renal dysfunction; however, neurotoxicity is also reported in patients with normal kidney function. Drug-Drug Interactions * Significant drug interaction exists when using cefepime. Concurrent use of cefepime with aminoglycoside antibiotics increases the risk of cytotoxicity and nephrotoxicity. Concurrent use of cephalosporins (cefepime) and potent diuretics (eg, furosemide) can result in nephrotoxicity. Monitor renal function when these medicines are administered to the patient. * Cefepime can lead to false positive glucose in urine; urinary glucose tests based on enzymatic glucose oxidase reactions are recommended. * Concurrent use of intravesical BCG for bladder carcinoma is not recommended with cefepime as it may adversely affect carcinoma outcomes. * Concurrent administration of cholera and typhoid vaccines is not recommended due to the reduced efficacy of vaccines. |
| References |
[1]. Efficacy and safety of cefepime: a systematic review and meta-analysis. Lancet Infect Dis. 2007 May;7(5):338-48. [2]. Barradell LB, Bryson HM. Cefepime. A review of its antibacterial activity, pharmacokinetic properties and therapeutic use. Drugs. 1994;47(3):471-505. [3]. Cefepime-induced neurotoxicity: a systematic review. Crit Care. 2017 Nov 14;21(1):276. doi: 10.1186/s13054-017-1856-1. [4]. The effect of different doses of cisplatin on the pharmacokinetic parameters of cefepime in mice. Lab Anim. 2006 Jul;40(3):296-300. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9342 mL | 9.6708 mL | 19.3416 mL | |
| 5 mM | 0.3868 mL | 1.9342 mL | 3.8683 mL | |
| 10 mM | 0.1934 mL | 0.9671 mL | 1.9342 mL |