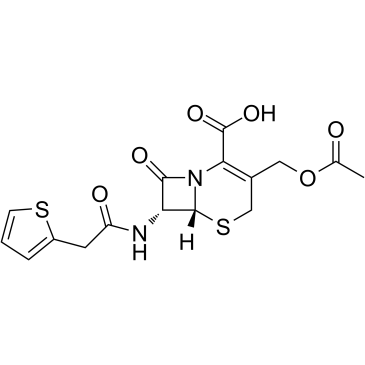
Cefalotin is a potent cephem antibiotic agent which inhibits class C β-lactamase AmpC, with an Ki of 0.32 µM. Cefalothin (Cefalotin) is a potent β-lactam cephalosporin antibiotic agent with a wide range antibacterial activity against gram-positive and gram-negative bacteria. It inhibits the growth of various Gram-positive and Gram-negative bacteria, including several strains of S. pyogenes, S. aureus, C. tetani, N. gonorrhoeae, Salmonella, and Shigella.
Physicochemical Properties
| Molecular Formula | C16H16N2O6S2 |
| Molecular Weight | 396.44 |
| Exact Mass | 396.044 |
| Elemental Analysis | C, 48.47; H, 4.07; N, 7.07; O, 24.21; S, 16.18 |
| CAS # | 153-61-7 |
| Related CAS # | Cephalothin sodium;58-71-9 |
| PubChem CID | 6024 |
| Appearance | White to off-white solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 757.2±60.0 °C at 760 mmHg |
| Melting Point | 160-161ºC |
| Flash Point | 411.8±32.9 °C |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C |
| Index of Refraction | 1.676 |
| LogP | 1.45 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 26 |
| Complexity | 680 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | S1C([H])([H])C(C([H])([H])OC(C([H])([H])[H])=O)=C(C(=O)O[H])N2C([C@]([H])([C@@]12[H])N([H])C(C([H])([H])C1=C([H])C([H])=C([H])S1)=O)=O |
| InChi Key | XIURVHNZVLADCM-IUODEOHRSA-N |
| InChi Code | InChI=1S/C16H16N2O6S2/c1-8(19)24-6-9-7-26-15-12(14(21)18(15)13(9)16(22)23)17-11(20)5-10-3-2-4-25-10/h2-4,12,15H,5-7H2,1H3,(H,17,20)(H,22,23)/t12-,15-/m1/s1 |
| Chemical Name | (6R,7R)-3-(acetyloxymethyl)-8-oxo-7-[(2-thiophen-2-ylacetyl)amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid |
| Synonyms | Cefalotin; Cephalothin; Cephalotin; Cefalothin; Cefalotina; Monosodium Salt, Cephalothin; Normon Brand of Cephalothin Sodium; Salt, Cephalothin Monosodium; Seffin; Sodium Cephalothin; Spaly Brand of Cephalothin Sodium; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | β-lactam |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion CEPHALOTHIN ENTERS AQUEOUS HUMOR AFTER SUBCONJUNCTIVAL INJECTIONS YIELDING PEAK LEVELS CA 1-2 HR AFTER DOSING. RATIO OF AQ HUMOR:SERUM ANTIBIOTIC LEVELS RANGES FROM 4.0 TO 67.0 DURING 5 HR AFTER DOSING, & LOSS OF ANTIBIOTIC FROM AQ HUMOR OCCURS BY BIPHASIC PROCESS. ...CEPHALOTHIN...WERE SHOWN...TO PENETRATE INTO BONE TO VERY LIMITED EXTENT AFTER SC OR ORAL DOSES TO RATS. RATIOS OF BONE TO SERUM CONCN AVG 1:4 FOR CEPHALOTHIN... DESPITE DIFFERENCES IN CONCN, T/2 IN BONE & SERUM WERE SIMILAR. DRUGS RECENTLY SHOWN TO ACTIVELY CROSS HUMAN PLACENTA INCL...CEPHALOTHIN... Concn of cephalothin present in urine after admin of 1 g range from 0.7 to 5 mg/mL. Excretion is delayed in presence of decr renal function ... For more Absorption, Distribution and Excretion (Complete) data for CEPHALOTHIN (11 total), please visit the HSDB record page. Metabolism / Metabolites Metabolized to a less active desacetyl metabolite, although 50-75% of the drug is eliminated unchanged in the urine. Approx 25% of cephalothin dose admin was eliminated in urine as deacetylcephalothin. Cephalothin ... /is/ deacetylated in vivo, and these metabolites have less antimicrobial activity than the parent cmpd ... The deacetylated metabolites also are excreted by the kidneys. Biological Half-Life 30 minutes Half-life = 0.6 hr /by/ injection /From table/ |
| Toxicity/Toxicokinetics |
Protein Binding 65-80% Interactions Concurrent administration with probenecid may prolong the serum half-life of cephalothin. ANIMAL STUDIES INDICATED THAT FUROSEMIDE ENHANCES NEPHROTOXICITY OF...CEPHALOTHIN... ...CEPHALOSPORINS...MAY BE AFFECTED BY CONCURRENT USE OF SULFINPYRAZONE. DIMINISHED TUBULAR SECRETION OF...WEAK ACIDS COULD RESULT IN HIGHER & MORE SUSTAINED SERUM LEVELS & HENCE, INTENSIFICATION OF DRUG ACTIVITY. /CEPHALOSPORINS/ NON-IONIC, ANIONIC, & ZWITTERIONIC SURFACTANTS INDUCED RAPIDLY REVERSIBLE HYPER-ABSORPTIVE STATE IN THOMAS CANINE FUNDIC POUCH FOR...CEPHALOTHIN... For more Interactions (Complete) data for CEPHALOTHIN (10 total), please visit the HSDB record page. Non-Human Toxicity Values LD50 Rat oral >10,000 mg/kg /sodium salt/ LD50 Rat ip 7716 mg/kg /sodium salt/ LD50 Mouse oral >20,000 mg/kg /sodium salt/ LD50 Mouse ip 5670 mg/kg /sodium salt/ |
| References |
[1]. Structure-based optimization of cephalothin-analogue boronic acids as beta-lactamase inhibitors. Bioorg Med Chem. 2008 Feb 1;16(3):1195-205. Epub 2007 Nov 7. |
| Additional Infomation |
Cefalotin is a semisynthetic, first-generation cephalosporin antibiotic with acetoxymethyl and (2-thienylacetyl)nitrilo moieties at positions 3 and 7, respectively, of the core structure. Administered parenterally during surgery and to treat a wide spectrum of blood infections. It has a role as an antimicrobial agent and an antibacterial drug. It is a semisynthetic derivative, a beta-lactam antibiotic allergen, a cephalosporin, a carboxylic acid, a member of thiophenes and an azabicycloalkene. It is a conjugate acid of a cefalotin(1-). Cefalotin is a cephalosporin antibiotic. Cephalothin has been reported in Bos taurus with data available. Cephalothin is a semisynthetic, beta-lactam, first-generation cephalosporin antibiotic with bactericidal activity. Cephalothin binds to and inactivates penicillin-binding proteins (PBP) located on the inner membrane of the bacterial cell wall. PBPs participate in the terminal stages of assembling the bacterial cell wall, and in reshaping the cell wall during cell division. Inactivation of PBPs interferes with the cross-linkage of peptidoglycan chains necessary for bacterial cell wall strength and rigidity. This results in the weakening of the bacterial cell wall and causes cell lysis. A cephalosporin antibiotic. Drug Indication Used to prevent infection during surgery and to treat many kinds of infections of the blood, bone or joints, respiratory tract, skin, and urinary tract. Mechanism of Action The bactericidal activity of cefalotin results from the inhibition of cell wall synthesis via affinity for penicillin-binding proteins (PBPs). The PBPs are transpeptidases which are vital in peptidoglycan biosynthesis. Therefore, their inhibition prevents this vital cell wall compenent from being properly synthesized. Bactericidal; action depends on ability to reach and bind penicillin-binding proteins located in bacterial cytoplasmic membranes. Cephalosporins inhibit bacterial septum and cell wall synthesis, probably by action of membrane-bound transpeptidase enzymes. This prevents cross-linkage of peptidoglycan chains, which is necessary for bacterial cell wall strength and rigidity. Also, cell division and growth are inhibited, and elongation of susceptible bacteria and lysis frequently occur. Rapidly dividing bacteria are those most susceptible to the actin of cephalosporins. /Cephalosporins/ Therapeutic Uses Since, among the cephalosporins, cephalothin is the most impervious to attack by Staphylococcal beta-lactamase, it is very effective in severe Staphylococcal infections, such as endocarditis. Mesh Heading: anti-bacterial agents Use should be restricted to treatment of serious infections caused by susceptible organisms, most commonly when patient is hypersensitive to penicillins. /Sodium/ ... Cephalosporin is ... drug of first choice ... for Klebsiella infections ... They are useful as alternative choices to penicillin. /Cephalosporins/ For more Therapeutic Uses (Complete) data for CEPHALOTHIN (28 total), please visit the HSDB record page. Drug Warnings Excretion is delayed in presence of decr renal function, and intervals between doses must be lengthened when renal failure is severe. Cephalothin should not be used to treat bacterial meningitis. Infections due to Enterococci are usually unaffected by these cmpd ... Enterococcal endocarditis cannot be cured with cephalosporin even when it is given concurrently with gentamicin or streptomycin. /Cephalosporins/ Patients with a history of a mild or a temporally distant reaction to penicillin appear to be at low risk of rash or other allergic reaction following the admin of a cephalosporin ... Patients who have had a recent severe, immediate reaction to a penicillin should be given a cephalosporin with great caution, if at all. /Cephalosporin/ For more Drug Warnings (Complete) data for CEPHALOTHIN (21 total), please visit the HSDB record page. Pharmacodynamics Cefalotin (INN) or cephalothin (USAN) is a semisynthetic first generation cephalosporin having a broad spectrum of antibiotic activity that is administered parenterally. |
Solubility Data
| Solubility (In Vitro) | DMSO : 79~100 mg/mL ( 199.27~252.24 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 10% DMSO+40% PEG300+5% Tween-80+45% Saline: ≥ 2.5 mg/mL (6.31 mM) (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5224 mL | 12.6122 mL | 25.2245 mL | |
| 5 mM | 0.5045 mL | 2.5224 mL | 5.0449 mL | |
| 10 mM | 0.2522 mL | 1.2612 mL | 2.5224 mL |