Physicochemical Properties
| Molecular Formula | C16H24N2O3 |
| Molecular Weight | 292.37336 |
| Exact Mass | 292.179 |
| CAS # | 51781-06-7 |
| Related CAS # | Carteolol hydrochloride;51781-21-6 |
| PubChem CID | 2583 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.13 g/cm3 |
| Boiling Point | 518.6ºC at 760 mmHg |
| Flash Point | 267.4ºC |
| Index of Refraction | 1.5800 (estimate) |
| LogP | 2.228 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 21 |
| Complexity | 354 |
| Defined Atom Stereocenter Count | 0 |
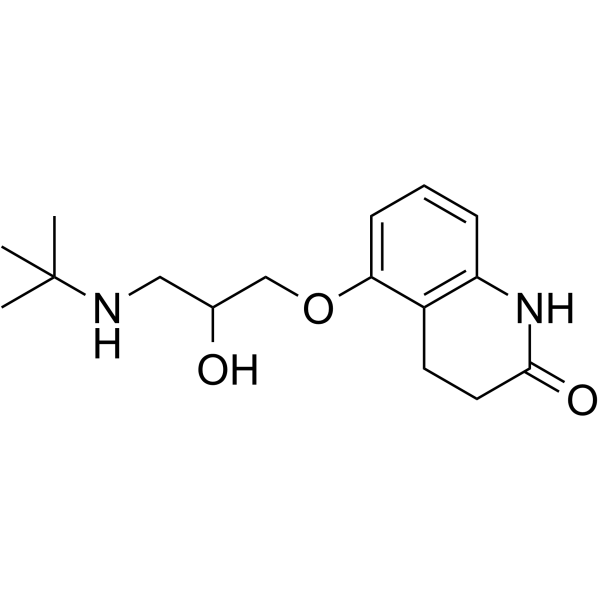
| SMILES | CC(C)(NCC(O)COC1=CC=CC2=C1CCC(N2)=O)C |
| InChi Key | LWAFSWPYPHEXKX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H24N2O3/c1-16(2,3)17-9-11(19)10-21-14-6-4-5-13-12(14)7-8-15(20)18-13/h4-6,11,17,19H,7-10H2,1-3H3,(H,18,20) |
| Chemical Name | 5-[3-(tert-butylamino)-2-hydroxypropoxy]-3,4-dihydro-1H-quinolin-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Carteolol is cytotoxic and decreases cell viability in a dose- and time-dependent manner (0–2%; 0-28 hours; HCEC) [1]. In HCEC cells, carteolol (0.25%; 4–12 hours) causes necrotic protein expression and apoptosis [1]. |
| Cell Assay |
Cell Viability Assay[1] Cell Types: HCEC Tested Concentrations: 0.00390625-2% Incubation Duration: 0, 2, 4, 8, 16, 20, 24 and 28 hrs (hours) Experimental Results: Decrease in cell viability and time at doses above 0.0015625% related way. Western Blot Analysis [1] Cell Types: HCEC Tested Concentrations: 0.25% Incubation Duration: 4, 8 and 12 hrs (hours) Experimental Results: The expression of anti-apoptotic proteins Bcl-2 and Bcl-xL was weakened, and the expression of pro-apoptotic protein Bax was enhanced Bad and Proapoptotic proteins Cyt.c and AIF released from mitochondria. Cell cycle analysis [1] Cell Types: HCEC Tested Concentrations: 0.25% Incubation Duration: 4, 8, 12 hrs (hours) Experimental Results: The number of G1 phase of the cell cycle increased, and the number of S phase diminished. |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Hepatic. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No data are available for the use of carteolol during breastfeeding. Because its excretion into breastmilk is probably extensive, other beta-adrenergic blocking drugs are preferred to oral carteolol while breastfeeding a neonate. Infants over 2 months of age have more mature kidney function and are less likely to be affected. Ophthalmic use of carteolol by the mother should pose little risk to the breastfed infant, although some guidelines state that gel formulations are preferred over solutions. To substantially diminish the amount of drug that reaches the breastmilk after using eye drops, place pressure over the tear duct by the corner of the eye for 1 minute or more, then remove the excess solution with an absorbent tissue. ◉ Effects in Breastfed Infants A study of mothers taking beta-blockers during nursing found a numerically, but not statistically significant increased number of adverse reactions in those taking any beta-blocker. Although the ages of infants were matched to control infants, the ages of the affected infants were not stated. None of the mothers were taking carteolol. Beta-adrenergic blocking drugs with similar breastmilk excretion characteristics have caused adverse effects in breastfed newborns. ◉ Effects on Lactation and Breastmilk A study in 6 patients with hyperprolactinemia and galactorrhea found no changes in serum prolactin levels following beta-adrenergic blockade with propranolol. There are no reports on the effects of beta-blockade or carteolol use during normal lactation. |
| References | [1]. Su W, et, al. Dose- and Time-Dependent Cytotoxicity of Carteolol in Corneal Endothelial Cells and the Underlying Mechanisms. Front Pharmacol. 2020 Mar 6;11:202. |
| Additional Infomation |
Carteolol is a quinolone and a secondary alcohol. It has a role as a beta-adrenergic antagonist, an antihypertensive agent, an antiglaucoma drug, an anti-arrhythmia drug and a sympatholytic agent. It is a conjugate base of a carteolol(1+). A beta-adrenergic antagonist used as an anti-arrhythmia agent, an anti-angina agent, an antihypertensive agent, and an antiglaucoma agent. Carteolol is a beta-Adrenergic Blocker. The mechanism of action of carteolol is as an Adrenergic beta-Antagonist. Carteolol is a synthetic quinolinone derivative and nonselective beta-adrenoceptor blocking agent with anti-glaucoma activity. Upon topical administration to the eye, carteolol decreases aqueous humor production, thereby reducing intraocular pressure (IOP). A beta-adrenergic antagonist used as an anti-arrhythmia agent, an anti-angina agent, an antihypertensive agent, and an antiglaucoma agent. See also: Carteolol Hydrochloride (has salt form). Drug Indication For the treatment of intraocular hypertension and chronic open-angle glaucoma Mechanism of Action The primary mechanism of the ocular hypotensive action of carteolol in reducing intraocular pressure is most likely a decrease in aqueous humor production. This process is initiated by the non-selective beta1 and beta2 adrenergic receptor blockade. Pharmacodynamics Carteolol is a beta1 and beta2 (non-selective) adrenergic receptor-blocking agent that does not have significant intrinsic sympathomimetic, direct myocardial depressant, or local anesthetic (membrane-stabilizing) activity. Carteolol, when applied topically to the eye, has the action of reducing elevated, as well as normal, intraocular pressure, whether or not accompanied by glaucoma. Elevated intraocular pressure is a major risk factor in the pathogenesis of glaucomatous visual field loss and optic nerve damage. Carteolol reduces intraocular pressure with little or no effect on pupil size or accommodation in contrast to the miosis which cholinergic agents are known to produce. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4203 mL | 17.1016 mL | 34.2032 mL | |
| 5 mM | 0.6841 mL | 3.4203 mL | 6.8406 mL | |
| 10 mM | 0.3420 mL | 1.7102 mL | 3.4203 mL |