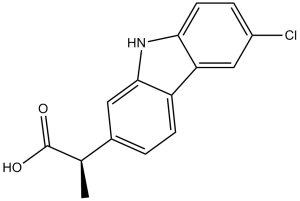
Carprofen (Rimadyl; Ro-205720; C 5720; quellin; Novox; Imafen; Rovera), a nonsteroid anti-inflammatory drug (NSAID), is a potent and multi-target FAAH/COX inhibitor with potential anti-inflammatory activity. It inhibits COX-2, COX-1 and FAAH with IC50s of 3.9 μM, 22.3 μM and 78.6 μM, respectively. Veterinarians prescribe it as a supportive treatment for various conditions in animals, is a COX2 inhibitor that inhibits canine COX2 with IC50 of 30 nM. Carprofen (S and R stereoisomers) inhibits canine COX2 with IC50 of 0.102 microM for the racemic mixture, the inhibition is primarily attributable to the S enantiomer (IC50, 0.0371 microM), which is approximately 200-fold more potent than the R enantiomer (IC50, 5.97 microM). It acts as a multi-target FAAH/COX inhibitor, with IC50s of 3.9 μM, 22.3 μM and 78.6 μM for COX-2, COX-1 and FAAH, respectively.
Physicochemical Properties
| Molecular Formula | C15H12CLNO2 | |
| Molecular Weight | 273.71 | |
| Exact Mass | 273.055 | |
| Elemental Analysis | C, 65.82; H, 4.42; Cl, 12.95; N, 5.12; O, 11.69 | |
| CAS # | 53716-49-7 | |
| Related CAS # | Carprofen-d3;1173019-42-5;Carprofen-13C,d3;2012598-34-2 | |
| PubChem CID | 2581 | |
| Appearance | Typically exists as White to off-white solids at room temperature | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 509.1±35.0 °C at 760 mmHg | |
| Melting Point | 186-188ºC | |
| Flash Point | 261.7±25.9 °C | |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C | |
| Index of Refraction | 1.732 | |
| LogP | 4.03 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 2 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 19 | |
| Complexity | 362 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | ClC1C([H])=C([H])C2=C(C=1[H])C1C([H])=C([H])C(=C([H])C=1N2[H])C([H])(C(=O)O[H])C([H])([H])[H] |
|
| InChi Key | PUXBGTOOZJQSKH-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C15H12ClNO2/c1-8(15(18)19)9-2-4-11-12-7-10(16)3-5-13(12)17-14(11)6-9/h2-8,17H,1H3,(H,18,19) | |
| Chemical Name | 2-(6-chloro-9H-carbazol-2-yl)propanoic acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
COX-2 (IC50 = 3.9 μM); COX-1 (IC50 = 22.3 μM); FAAH (IC50 = 78.6 μM) Fatty Acid Amide Hydrolase (FAAH) (Ki: 2.3 ± 0.2 μM for Carprofen, measured in recombinant human FAAH enzyme assay) [1] - Cyclooxygenase-1 (COX-1) (IC50: 0.35 ± 0.03 μM for Carprofen, measured in sheep seminal vesicle microsomes) [1] - Cyclooxygenase-2 (COX-2) (IC50: 0.18 ± 0.02 μM for Carprofen, measured in LPS-stimulated human monocytes; selectivity ratio (COX-1/COX-2) = 1.94) [1] |
| ln Vitro |
Compound 1, or carprofen, is a non-steroidal anti-inflammatory drug. The IC50 values for COX-2, COX, and FAAH are 3.9 μM, 22.3 μM, and 78.6 μM-1, respectively, as a multi-target inhibitor of FAAH/COX. In CCL and CaCL cells, carprofen (10 μg/mL) has cytoprotective properties and lowers both cell apoptosis. When compared to the corresponding CCL or CaCL controls, PGE2 concentrations were not significantly increased by carprofen (10 μg/mL) [2]. Cytoprotective effects of NSAIDs were dependent on the extent of SNP-induced apoptosis and were greatest in CCL and CaCL cell cultures with moderate SNP-induced cytotoxic effects. Preincubation with an NSAID improved cell viability by 15% to 45% when CCL and CaCL cells were subsequently incubated with SNP. Carprofen (10 μg/mL) had the greatest cytoprotective effects for CCL and CaCL cells. Incubation with NSAIDs resulted in a nonsignificant decrease in PGE(2) production from SNP-damaged cells. Conclusions and clinical relevance: Results indicated that carprofen, meloxicam, and robenacoxib may reduce apoptosis in cells originating from canine cruciate ligaments[2]. 1. Inhibition of FAAH and COX activity ([1]): - FAAH inhibition: Recombinant human FAAH was treated with Carprofen (0.1-10 μM) for 30 min, then incubated with fluorescent substrate (4-methylumbelliferyl arachidonate, AMC). At 2 μM, Carprofen inhibited FAAH activity by 85 ± 4%; at 10 μM, inhibition reached 92 ± 3% [1] - COX-2 inhibition: LPS-stimulated human monocytes (1 μg/mL LPS, 16 h) were treated with Carprofen (0.05-1 μM) for 30 min + arachidonic acid (100 μM) for 15 min. At 0.2 μM, Carprofen reduced COX-2-mediated PGE2 production by 85 ± 5%; at 0.5 μM, inhibition reached 96 ± 2% [1] - COX-1 inhibition: Sheep seminal vesicle microsomes (COX-1 source) were treated with Carprofen (0.1-2 μM) + arachidonic acid (100 μM). At 0.4 μM, Carprofen reduced COX-1-mediated TXB2 production by 82 ± 4% [1] 2. Cytoprotective effect on canine cruciate ligament cells ([2]): - Cell model: Canine cranial cruciate ligament (CCL) cells were cultured in DMEM + 10% FBS. Apoptosis was induced by sodium nitroprusside (SNP, 1 mM) for 24 h. - Anti-apoptotic activity: Carprofen (1 μM, 5 μM, 10 μM) was added 1 h before SNP treatment. At 10 μM, Carprofen reduced apoptotic rate from 35 ± 3% (SNP-only group) to 12 ± 2% (Annexin V-FITC/PI staining) [2] - Protein regulation: Western blot showed 10 μM Carprofen upregulated anti-apoptotic Bcl-2 by 2.1 ± 0.2-fold and downregulated pro-apoptotic Bax by 45 ± 5%, with cleaved caspase-3 reduced by 58 ± 4% vs. SNP group [2] |
| ln Vivo |
On days 3 and 10, carprofen (2.2 mg/kg, po) dramatically lowered the levels of PGE2 in canine blood. On day 3, carprofen similarly decreased the synthesis of PGE2 in the stomach; however, by day 10, the inhibition was not as great. Moreover, on days 3 and 10, it was demonstrated that carprofen had no effect on the synthesis of stomach PGE1 in dogs [3]. 1. Modulation of prostanoid production in dogs with chronic osteoarthritis ([3]): - Animals: 18 client-owned dogs (10-15 kg, 4-8 years old) with radiographically confirmed chronic osteoarthritis of the stifle joint, randomly divided into 3 groups: control (vehicle), Carprofen 4 mg/kg/day, deracoxib 2 mg/kg/day (n=6/group). - Treatment: Carprofen was orally administered once daily for 7 days; control received 0.5% carboxymethyl cellulose (CMC-Na). - Prostanoid changes: - Blood TXB2 (COX-1 product): Carprofen group reduced by 42 ± 4% vs. baseline (control: no significant change) [3] - Synovial fluid PGE2 (COX-2 product): Carprofen group reduced by 68 ± 5% vs. baseline (greater than deracoxib group’s 52 ± 4%) [3] - Gastric mucosa PGE2 (COX-1/COX-2 product): Carprofen group reduced by 25 ± 3% vs. baseline (less than deracoxib group’s 38 ± 4%), indicating lower gastrointestinal impact [3] |
| Enzyme Assay |
In vitro assays[1] FAAH activity was measured by incubating for 30 minutes at 37°C [3H] anandamide (1 uM cold AEA and 0.6 nM (1 mCi/mL) [3H]-AEA (Arachidonyl-[1-3H] ethanolamine, Specific activity 60 Ci/mmol) in the presence of 50 ug protein/sample of total rat brain homogenates in assay buffer (50 mM TRIS pH 7.4, 0.05 % fatty acid free BSA). The reaction was stopped with cold 1:1 CHCl3/MeOH. The aqueous phase was counted by liquid scintillation (Microbeta2 Lumijet, adapted from Kathuria et al, 2003). Inhibitors were pre-incubated with the enzyme preparation at the appropriate concentration for 10 minutes prior to substrate addition. COX activity was measured using a commercial enzyme immunoassay kit. The manufacturer protocol was followed except for the substrate concentration. Briefly, inhibitors were pre-incubated with either ovine COX-1 or human COX-2 for 10 min at 37 °C, and the reaction was carried out in the presence of 5 μM arachidonic acid for 2 minutes at 37 °C. The reaction was stopped with hydrochloric acid and COX-derived PGH2 was then converted to PGF2α with SnCl2. The PGF2α product is then quantified via enzyme immunoassay (EIA) using a PG-specific antibody and competing with a PG-acetylcholinesterase conjugate. Absorbance is measured at 412 nM with a Tecan Infinite M200 plate reader and data processed according to manufacturer’s instructions[1]. 1. FAAH activity assay ([1]): - Reaction system (100 μL): 50 mM Tris-HCl (pH 7.4), 1 mM EDTA, 0.1 mg/mL recombinant human FAAH, 10 μM 4-methylumbelliferyl arachidonate (AMC, substrate), and serial dilutions of Carprofen (0.1-10 μM). - Incubation: Mixtures were incubated at 37°C for 20 min. The reaction was terminated by adding 10 μL of 1 M HCl. - Detection: Fluorescence intensity of the product (4-methylumbelliferone) was measured using a fluorometer (excitation: 360 nm, emission: 460 nm). Inhibition rate = (1 - sample fluorescence/control fluorescence) × 100%. Ki was calculated via Lineweaver-Burk plot analysis [1] 2. COX-1/COX-2 activity assay ([1]): - COX-1 sample preparation: Sheep seminal vesicles were homogenized and centrifuged (100,000×g for 60 min, 4°C) to isolate microsomes, resuspended in 50 mM Tris-HCl (pH 8.0) containing 2 μM heme. - COX-2 sample preparation: Human monocytes were stimulated with LPS (1 μg/mL) for 16 h, lysed, and centrifuged (10,000×g for 10 min, 4°C) to collect supernatant. - Reaction system (200 μL): - COX-1: 10 μL sheep microsomes + Carprofen (0.1-2 μM) + 100 μM arachidonic acid. - COX-2: 20 μL monocyte supernatant + Carprofen (0.05-1 μM) + 100 μM arachidonic acid. - Incubation: 37°C for 15 min, terminated by 20 μL 1 M HCl. - Detection: TXB2 (COX-1) and PGE2 (COX-2) were measured via radioimmunoassay (RIA) kits. IC50 was calculated via nonlinear regression [1] |
| Cell Assay |
Primary cultures of CCL and CaCL cells were created via enzymatic dissociation of cruciate explants. Purified cell cultures were incubated for 2 hours without (controls) or with 1 of 3 concentrations of 1 of 4 NSAIDs (10, 100, or 200 μg of acetylsalicylic acid/mL; 0.1, 1, or 10 μg of carprofen/mL; 0.1, 1, or 10 μg of meloxicam/mL; or 0.1, 1, or 10 μg of robenacoxib/mL) and subsequently incubated for 18 hours with 1 of 3 concentrations of SNP in an attempt to induce mild, moderate, or severe cytotoxic effects. Cell viability and apoptosis were analyzed via a cell proliferation assay and flow cytometry, respectively. Prostaglandin E(2) concentrations were measured via an ELISA[2]. 1. Canine CCL cell anti-apoptosis assay ([2]): - Cell isolation and culture: Canine cranial cruciate ligaments were harvested from healthy dogs (euthanized for non-orthopedic reasons), minced, and digested with 0.1% collagenase type II (37°C, 4 h). Cells were filtered through a 70 μm strainer, cultured in DMEM + 10% FBS + 1% penicillin-streptomycin at 37°C in 5% CO₂ (passages 3-5 used). - Drug treatment: Cells were plated in 24-well plates (1×10⁵ cells/well) and adhered overnight. Carprofen (1 μM, 5 μM, 10 μM) was added for 1 h pre-treatment, then sodium nitroprusside (SNP, 1 mM) was added for 24 h. - Apoptosis detection: Cells were harvested, stained with Annexin V-FITC and PI for 15 min in dark, analyzed by flow cytometry to calculate apoptotic rate. - Protein detection: Cells were lysed with RIPA buffer (protease inhibitors). Bcl-2, Bax, and cleaved caspase-3 were detected by Western blot, with GAPDH as loading control [2] |
| Animal Protocol |
Each dog receives Carprofen (2.2 mg/kg, PO, q 12 h), deracoxib (2 mg/kg, PO, q 24 h), or etodolac (10 to 15 mg/kg, PO, q 24 h) for 10 days in a crossover design with a 30- to 60-day washout period between treatments. On days 0, 3, and 10 of each treatment period, blood is collected for evaluation of TXB2 and PGE2 concentrations. In addition, anesthesia is induced with propofol (4 mg/kg) and maintained with isoflurane. Synovial fluid is collected from both stifle joints by use of a standard arthrocentesis technique for evaluation of PGE2 concentrations. Gastroscopy is performed during each anesthetic episode, and 3 to 6 endoscopic biopsy specimens are collected from the gastric antrum for evaluation of PGE1 and PGE2 synthesis. On day 0 for each dog, a gastric biopsy specimen is placed into a Campylobacter-like organism test kit and evaluated for up to 24 hours for Helicobacter spp. Stained slides (H&E) of gastric biopsy specimens are also evaluated for the presence of underlying inflammation Dogs 1. Canine chronic osteoarthritis model ([3]): - Animals: 18 client-owned dogs (10-15 kg, 4-8 years old) with chronic stifle osteoarthritis (duration ≥3 months), confirmed by clinical signs (lameness, joint swelling) and radiography (osteophytes, joint space narrowing). Dogs with hepatic/renal disease were excluded. - Grouping: Randomly divided into control (0.5% CMC-Na), Carprofen 4 mg/kg/day, deracoxib 2 mg/kg/day groups (n=6/group). - Drug preparation: Carprofen was ground into powder, suspended in 0.5% CMC-Na to a concentration of 0.4 mg/mL (for 4 mg/kg dose, 10 μL/g body weight). - Administration: Oral gavage once daily for 7 days; control received equal volume of 0.5% CMC-Na. - Sample collection: - Blood: Collected via cephalic vein at baseline and day 7, centrifuged (3000×g, 10 min) to separate plasma for TXB2 detection. - Synovial fluid: Collected via arthrocentesis of the affected stifle joint at baseline and day 7, stored at -80°C for PGE2 detection. - Gastric mucosa: Dogs were euthanized at day 7, gastric fundus mucosa biopsied for PGE2 measurement and histological examination [3] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Rapidly and nearly completely absorbed (more than 90% bioavailable) when administered orally. Metabolism / Metabolites Hepatic. Biological Half-Life Approximately 8 hours (range 4.5–9.8 hours) in dogs. |
| Toxicity/Toxicokinetics |
Protein Binding High (99%) 1. In vivo safety in dogs ([3]): - Gastrointestinal safety: Carprofen 4 mg/kg/day (oral, 7 days) reduced gastric mucosa PGE2 by 25 ± 3% (vs. deracoxib’s 38 ± 4%), with no macroscopic gastric ulcers or mucosal erosion observed in any dogs (histological score: 0.3 ± 0.1 vs. control 0.2 ± 0.1) [3] - Hepatic/renal safety: Serum alanine transaminase (ALT: 45 ± 5 U/L vs. baseline 43 ± 4 U/L) and creatinine (0.8 ± 0.1 mg/dL vs. baseline 0.7 ± 0.1 mg/dL) in Carprofen group remained within normal canine ranges [3] 2. In vitro cytotoxicity ([2]): - Carprofen at concentrations up to 10 μM had no significant cytotoxicity on canine CCL cells (MTT assay: viability ≥ 90% vs. untreated control) after 24 h treatment [2] |
| References |
[1]. Identification and characterization of carprofen as a multitarget fatty acid amide hydrolase/cyclooxygenase inhibitor. J Med Chem. 2012 Oct 25;55(20):8807-26. [2]. In vitro cytoprotective effects of acetylsalicylic acid, carprofen, meloxicam, or robenacoxib against apoptosis induced by sodium nitroprusside in canine cruciate ligament cells. Am J Vet Res. 2012 Nov;73(11):1752-8. [3]. In vivo effects of carprofen, deracoxib, and etodolac on prostanoid production in blood, gastric mucosa, and synovial fluid in dogs with chronic osteoarthritis. Am J Vet Res. 2005 May;66(5):812-7. |
| Additional Infomation |
Carprofen is propanoic acid in which one of the methylene hydrogens is substituted by a 6-chloro-9H-carbazol-2-yl group. A non-steroidal anti-inflammatory drug, it is no longer used in human medicine but is still used for treatment of arthritis in elderly dogs. It has a role as a non-steroidal anti-inflammatory drug, an EC 1.14.99.1 (prostaglandin-endoperoxide synthase) inhibitor and a photosensitizing agent. It is a member of carbazoles and an organochlorine compound. Carprofen is a non-steroidal anti-inflammatory drug (NSAID) that is used by veterinarians as a supportive treatment for the relief of arthritic symptoms in geriatric dogs. Carprofen was previously used in human medicine for over 10 years (1985-1995). It was generally well tolerated, with the majority of adverse effects being mild, such as gastro-intestinal pain and nausea, similar to those recorded with aspirin and other non-steroidal anti-inflammatory drugs. It is no longer marketed for human usage, after being withdrawn on commercial grounds. Carprofen is a propionic acid derivate and nonsteroidal anti-inflammatory drug (NSAID) with anti-inflammatory, analgesic, and antipyretic activities, used exclusively in veterinary medicine. Carprofen inhibits the activity of the enzymes cyclo-oxygenase (COX) I and II, resulting in a decreased formation of precursors of prostaglandins and thromboxanes. This inhibits the formation of prostaglandins, by prostaglandin synthase, that are involved in pain, inflammation and fever. Ibuprofen also causes a decrease in the formation of thromboxane A2 synthesis, by thromboxane synthase, thereby inhibiting platelet aggregation. Drug Indication For use as a pain reliever in the treatment of joint pain and post-surgical pain. Mechanism of Action The mechanism of action of carprofen, like that of other NSAIDs, is believed to be associated with the inhibition of cyclooxygenase activity. Two unique cyclooxygenases have been described in mammals. The constitutive cyclooxygenase, COX-1, synthesizes prostaglandins necessary for normal gastrointestinal and renal function. The inducible cyclooxygenase, COX-2, generates prostaglandins involved in inflammation. Inhibition of COX-1 is thought to be associated with gastrointestinal and renal toxicity while inhibition of COX-2 provides anti-inflammatory activity. In an in vitro study using canine cell cultures, carprofen demonstrated selective inhibition of COX-2 versus COX-1. Pharmacodynamics Carprofen is a non-steroidal anti-inflammatory drug (NSAID) of the propionic acid class that includes ibuprofen, naproxen, and ketoprofen. It is no longer used in the clinical setting, but is approved for use in dogs. Carprofen is non-narcotic and has characteristic analgesic and antipyretic activity approximately equipotent to indomethacin in animal models. 1. Carprofen is a non-steroidal anti-inflammatory drug (NSAID) with unique multitarget activity: it inhibits both FAAH (regulating endocannabinoid signaling) and COX-1/COX-2 (reducing prostaglandin synthesis), distinguishing it from single-target NSAIDs [1] 2. In veterinary medicine, Carprofen is widely used to treat chronic osteoarthritis in dogs due to its strong COX-2 inhibitory effect (reducing synovial inflammation) and low gastrointestinal toxicity (minimal impact on gastric PGE2) [3] 3. Carprofen exerts cytoprotective effects on canine orthopedic cells (e.g., CCL cells) by regulating the Bcl-2/Bax/caspase-3 apoptotic pathway, suggesting potential for adjuvant treatment of joint injuries [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.60 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (7.60 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (7.60 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6535 mL | 18.2675 mL | 36.5350 mL | |
| 5 mM | 0.7307 mL | 3.6535 mL | 7.3070 mL | |
| 10 mM | 0.3654 mL | 1.8268 mL | 3.6535 mL |