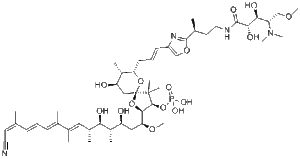
Calyculin A is a novel, potent, selective and cell-permeable inhibitor of protein phosphatase 1 (PP1) and protein phosphatase 2A (PP2A) with IC50s of 0.5 to 1 nM. Calyculin A is a natural product and a marine toxin isolated from Disodermia calyx. Calyculin A does not have significant effect against PP2B, PP2C, and PP4.2 Through its effects on PP1 and PP2A, calyculin A has been shown to either promote or inhibit cancer cell growth in tumor cell lines and animal models.
Physicochemical Properties
| Molecular Formula | C50H81N4O15P |
| Molecular Weight | 1009.16974 |
| Exact Mass | 1008.54 |
| Elemental Analysis | C, 59.51; H, 8.09; N, 5.55; O, 23.78; P, 3.07 |
| CAS # | 101932-71-2 |
| PubChem CID | 5311365 |
| Appearance | Colorless to light yellow liquid |
| Density | 1.25g/cm3 |
| Index of Refraction | 1.57 |
| LogP | 5.079 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 26 |
| Heavy Atom Count | 70 |
| Complexity | 1930 |
| Defined Atom Stereocenter Count | 15 |
| SMILES | O[C@@H]([C@H]([C@@H](N(C)C)COC)O)C(NCC[C@@H](C1=NC(/C=C/C[C@@H]([C@@H](C)[C@H](O)C2)O[C@@]32C(C)(C)[C@@H](OP(O)(O)=O)[C@@H]([C@@H](OC)C[C@H](O)[C@H](C)[C@H](O)[C@H](C)/C=C(C)/C(C)=C/C=C/C(C)=C\C#N)O3)=CO1)C)=O |
| InChi Key | FKAWLXNLHHIHLA-YCBIHMBMSA-N |
| InChi Code | InChI=1S/C50H81N4O15P/c1-29(20-22-51)16-14-17-30(2)32(4)24-33(5)42(57)35(7)38(55)25-41(65-13)45-46(69-70(61,62)63)49(8,9)50(68-45)26-39(56)34(6)40(67-50)19-15-18-36-27-66-48(53-36)31(3)21-23-52-47(60)44(59)43(58)37(28-64-12)54(10)11/h14-18,20,24,27,31,33-35,37-46,55-59H,19,21,23,25-26,28H2,1-13H3,(H,52,60)(H2,61,62,63)/b16-14+,18-15+,29-20-,30-17+,32-24+/t31-,33+,34-,35-,37-,38-,39+,40-,41-,42+,43-,44-,45+,46-,50+/m0/s1 |
| Chemical Name | (2R,3R,5R,7S,8S,9R)-2-((1S,3S,4S,5R,6R,7E,9E,11E,13Z)-14-cyano-3,5-dihydroxy-1-methoxy-4,6,8,9,13-pentamethyltetradeca-7,9,11,13-tetraen-1-yl)-7-((E)-3-(2-((S)-4-((2S,3S,4S)-4-(dimethylamino)-2,3-dihydroxy-5-methoxypentanamido)butan-2-yl)oxazol-4-yl)allyl)-9-hydroxy-4,4,8-trimethyl-1,6-dioxaspiro[4.5]decan-3-yl dihydrogen phosphate |
| Synonyms | (-)-Calyculin A; Calyculin A; calyculin A; 101932-71-2; (-)-calyculin A; CCRIS 3706; Calyculin-A; Calyculin A from Discodermia calyx; UNII-7D07U14TK3; 7D07U14TK3; Hemicalyculin A; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PP1/phosphatase 1 (IC50 = 2 nM); PP2A/protein phosphatase 2A (IC50 = 0.5-1 nM) |
| ln Vitro | In this report, it was demonstrated that calyculin A induced apoptosis in MG63 cells, as judged by WST-8 assay, nuclear fragmentation, and DNA ladder formation. Expression of PTEN, FasL, and FasR mRNA was stimulated by calyculin A treatment in MG63 cells. Calyculin A also enhanced the phosphorylation level of NF-kappaB, as judged from the results of Western blot analysis and an in vitro dephosphorylation assay. Western blot analysis with anti-phospho-p65NF-kappaB antibody disclosed that the NF-kappaB was phosphorylated on serine 536 in cytosol and translocated into nucleus with calyculin A-treatment. The phosphorylation status of p65NF-kappaB was further confirmed by using the phosphorylation site-mutated p65NF-kappaB gene transfected into HEK293 cells. Unlike TNF-alpha, calyculin A treatment did not degraded IkappaBalpha within 10 min, while it degraded IkappaBalpha at 2-h treatment. Our findings indicate that calyculin A elicit phosphorylation of NF-kappaB on serine 536 in MG63 cells, resulting in the translocation of phospho-NF-kappaB to the nucleus, thereby promoting transcriptional activity of NF-kappaB-related genes. [2] |
| Enzyme Assay | Calyculin A and okadaic acid induce contraction in smooth muscle fibers. Okadaic acid is an inhibitor of phosphatase activity and the aims of this study were to determine if calyculin A also inhibits phosphatase and to screen effects of both compounds on various phosphatases. Neither compound inhibited acid or alkaline phosphatases, nor the phosphotyrosine protein phosphatase. Both compounds were potent inhibitors of the catalytic subunit of type-2A phosphatase, with IC50 values of 0.5 to 1 nM. With the catalytic subunit of protein phosphatase type-1, calyculin A was a more effective inhibitor than okadaic acid, IC50 values for calyculin A were about 2 nM and for okadaic acid between 60 and 500 nM. The endogenous phosphatase of smooth muscle myosin B was inhibited by both compounds with IC50 values of 0.3 to 0.7 nM and 15 to 70 nM, for calyculin A and okadaic acid, respectively. The partially purified catalytic subunit from myosin B had IC50 values of 0.7 and 200 nM for calyculin A and okadaic acid, respectively. The pattern of inhibition for the phosphatase in myosin B therefore is similar to that of the type-1 enzyme.[1] |
| Cell Assay |
Cytotoxicity assay [2] Cell Types: human osteosarcoma MG63 cells Tested Concentrations: 0, 1, 2, 5, CalycuLin A (1-10 nM; 24 hrs (hours)) induces cytotoxicity in MG63 cells in a dosed manner [2]. 10 nM Incubation Duration: 24 hrs (hours) Experimental Results: Treatment with 0.1 nM for 24 hrs (hours) had minimal effect on MG63 cell survival. Cell rounding and shrinkage were evident in cultures treated with 5 nM for 24 hrs (hours). Cell viability levels treated with 10 nM were 27% of control cultures. |
| References |
[1]. Calyculin A and okadaic acid: inhibitors of protein phosphatase activity. Biochem Biophys Res Commun. 1989 Mar 31;159(3):871-7. [2]. Calyculin A induces apoptosis and stimulates phosphorylation of p65NF-kappaB in human osteoblastic osteosarcoma MG63 cells. Int J Oncol. 2007 Aug;31(2):389-96. |
| Additional Infomation | calyculin A has been reported in Lamellomorpha strongylata with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.9909 mL | 4.9546 mL | 9.9091 mL | |
| 5 mM | 0.1982 mL | 0.9909 mL | 1.9818 mL | |
| 10 mM | 0.0991 mL | 0.4955 mL | 0.9909 mL |