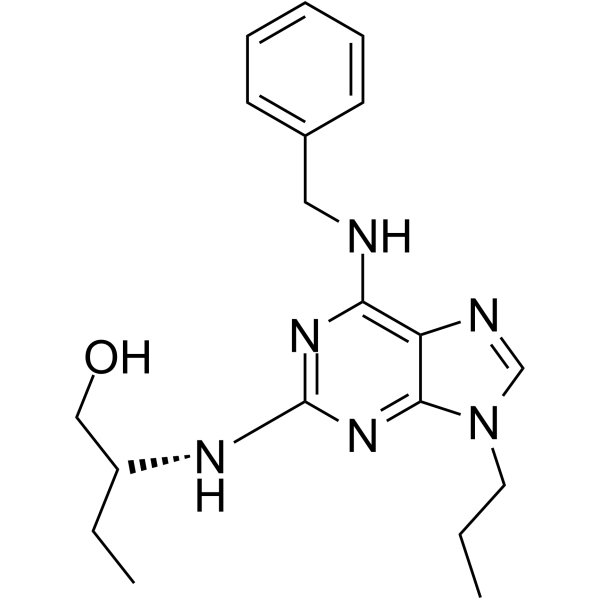
Physicochemical Properties
| Molecular Formula | C19H26N6O |
| Molecular Weight | 354.4493 |
| Exact Mass | 354.216 |
| CAS # | 1402821-24-2 |
| PubChem CID | 69472592 |
| Appearance | White to off-white solid powder |
| LogP | 3.3 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 26 |
| Complexity | 406 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | O([H])C([H])([H])[C@@]([H])(C([H])([H])C([H])([H])[H])N([H])C1=NC(=C2C(=N1)N(C([H])=N2)C([H])([H])C([H])([H])C([H])([H])[H])N([H])C([H])([H])C1C([H])=C([H])C([H])=C([H])C=1[H] |
| InChi Key | LKXPLOHGQSEPEM-OAHLLOKOSA-N |
| InChi Code | InChI=1S/C19H26N6O/c1-3-10-25-13-21-16-17(20-11-14-8-6-5-7-9-14)23-19(24-18(16)25)22-15(4-2)12-26/h5-9,13,15,26H,3-4,10-12H2,1-2H3,(H2,20,22,23,24)/t15-/m1/s1 |
| Chemical Name | (2R)-2-[[6-(benzylamino)-9-propylpurin-2-yl]amino]butan-1-ol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
N-type (Cav2.2) calcium channels (EC50 = 7.21 ± 0.86 µM) P/Q-type (Cav2.1) calcium channels (EC50 = 8.8 ± 1.1 µM) L-type (Cav1.3) calcium channels (EC50 > 100 µM) Cyclin-dependent kinase 2 (Cdk2) (IC50 = 3.29 ± 0.43 µM) Cyclin-dependent kinase 5 (Cdk5) (IC50 = 3.03 ± 0.32 µM) Cyclin-dependent kinase 1 (Cdk1) (IC50 > 20 µM) Mitogen-activated protein kinase 1 (MAPK1) (IC50 > 20 µM) Myosin light-chain kinase (MLCK) (IC50 > 20 µM). [1] |
| ln Vitro |
Ca2+ channel agonist 1 (Compound 13d) has an EC50 of 3.34 μM and 14.23 μM, respectively, making it an inhibitor of Cdk2 and an agonist of N-type Ca2+ channels. A1: Ca2+ channel agonist displays ca. Agonism is increased twofold and cdk2 kinase activity is decreased twenty-twofold when compared to standard (R)-roscovitine [1]. 13x is a synthetic analogue of (R)-roscovitine. [1] In whole-cell patch-clamp recordings, 13x acted as an agonist for N-type calcium channels with an EC50 of 7.21 ± 0.86 µM, representing approximately a 4-fold improvement in agonist efficacy compared to (R)-roscovitine (EC50 = 27.58 ± 1.65 µM). [1] 13x also acted as an agonist for P/Q-type calcium channels with an EC50 of 8.8 ± 1.1 µM, but showed no agonist activity on L-type calcium channels up to 100 µM, indicating selectivity for N- and P/Q-type over L-type channels. [1] In a kinase inhibition panel, 13x showed an IC50 of 3.29 ± 0.43 µM against Cdk2, which is approximately a 22-fold decrease in inhibitory activity compared to (R)-roscovitine (IC50 = 0.151 ± 0.004 µM). Its IC50 against Cdk5 was 3.03 ± 0.32 µM, against Cdk1 was >20 µM, and it showed no significant inhibition (IC50 >20 µM) of MAPK1 or MLCK. [1] |
| Enzyme Assay |
The inhibitory activities of 13x and reference compounds against Cdk1/cyclinB(h), Cdk2/cyclinA(h), Cdk5/p35(h), MAPK1(h), and MLCK(h) were determined by a commercial service. The assays were performed at three different concentrations of the agent (0.2, 2, and 20 µM), with (R)-roscovitine included as a positive control. IC50 values were derived from the results. [1] |
| Cell Assay |
The effects of 13x and other (R)-roscovitine derivatives on calcium channels were evaluated using whole-cell perforated patch-clamp recordings. The pipette solution contained Cs₂SO₄, CsCl, MgCl₂, and HEPES (pH 7.4). Cells were bathed in a saline solution containing choline chloride, TEA-Cl, CaCl₂, MgCl₂, and HEPES (pH 7.4). Patch pipettes were fabricated from borosilicate glass. Capacitive currents and passive membrane responses were subtracted. Currents were amplified, filtered at 5 kHz, and digitized at 10 kHz for analysis. A liquid junction potential was corrected during recordings. To measure effects on calcium channel tail currents, the tail current integral was measured before and after compound application, with each trace normalized to its peak. All experiments were conducted at room temperature (22°C). Compounds were dissolved in DMSO as 100 mM stocks and stored at -20°C. For recordings, compounds were diluted in saline to final concentrations of 1–100 µM and applied via bath perfusion. Control experiments with DMSO alone showed no significant effects. [1] |
| References |
[1]. Synthesis and biological evaluation of a selective N- and p/q-type calcium channel agonist. ACS Med Chem Lett. 2012 Oct 1;3(12):985-990. |
| Additional Infomation |
13x was developed through systematic structure-activity relationship (SAR) studies based on modifying the purine scaffold of (R)-roscovitine. The goal was to reduce cyclin-dependent kinase (Cdk) inhibitory activity while enhancing selectivity and potency as an agonist for N- and P/Q-type voltage-gated calcium channels. [1] The synthesis of 13x started from 2,6-dichloropurine. Alkylation at the N-9 position introduced an n-propyl group (R¹), followed by nucleophilic aromatic substitution at C-6 with a (2-methyl-5-thienyl)methylamine. Finally, displacement of the C-2 chloride with (R)-2-amino-1-butanol yielded the target compound. [1] Molecular docking studies to the Cdk2/(R)-roscovitine complex were performed to analyze the structural basis for the reduced Cdk2 inhibitory activity of analogues like 13x. The docking poses and binding scores (MolDock Score) were consistent with the experimental trend showing reduced kinase inhibition. [1] The long-term goal of developing such selective calcium channel agonists is for the therapy of Lambert-Eaton myasthenic syndrome (LEMS) and other neurological diseases. The mechanism involves directly increasing calcium influx through presynaptic N- and P/Q-type channels during action potentials to enhance neurotransmitter release, compensating for the channel loss in LEMS. [1] Based on the clinical experience with the indirect potassium channel blocker 3,4-diaminopyridine (DAP) in LEMS, selective use-dependent N- and P/Q-type calcium channel agonists like 13x are predicted to be well tolerated, assuming they do not cross the blood-brain barrier. [1] 13x is also noted as a useful experimental tool for studying the basic properties of P/Q- and N-type calcium channels and the calcium control of neurotransmitter release. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~141.06 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.05 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.05 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8213 mL | 14.1064 mL | 28.2127 mL | |
| 5 mM | 0.5643 mL | 2.8213 mL | 5.6425 mL | |
| 10 mM | 0.2821 mL | 1.4106 mL | 2.8213 mL |