Physicochemical Properties
| Molecular Formula | C24H17CLN2O3 |
| Molecular Weight | 416.86 |
| Exact Mass | 416.093 |
| CAS # | 1421365-63-0 |
| PubChem CID | 50904505 |
| Appearance | White to off-white solid powder |
| LogP | 5.315 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 30 |
| Complexity | 693 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | QHVSQUYCVUHYKT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H17ClN2O3/c1-29-15-10-12-16(13-11-15)30-23-21(25)14-26-27(24(23)28)22-19-8-4-2-6-17(19)18-7-3-5-9-20(18)22/h2-14,22H,1H3 |
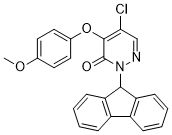
| Chemical Name | 5-chloro-2-(9H-fluoren-9-yl)-4-(4-methoxyphenoxy)pyridazin-3-one |
| Synonyms | ML 250; ML250; ML-250; CYM 50769; 1421365-63-0; CYM 50769; CYM50769; 5-chloro-2-(9H-fluoren-9-yl)-4-(4-methoxyphenoxy)pyridazin-3-one; CHEMBL1972527; 5-chloro-2-(9H-fluoren-9-yl)-4-(4-methoxyphenoxy)pyridazin-3(2H)-one; ML250; MLS003675924; CYM-50769; CYM50769; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Neuropeptides B and W receptor 1 (NPBWR1); GPR7 |
| ln Vitro |
In a dose-dependent manner, CYM 50769 (1 and 3 μM; 30 min) inhibits the proliferation of ATDC5 cells caused by NPW-23 [1]. In this Letter researchers report on the advances in their NPBWR1 antagonist program aimed at optimizing the 5-chloro-2-(3,5-dimethylphenyl)-4-(4-methoxyphenoxy)pyridazin-3(2H)-one lead molecule previously obtained from a high-throughput screening (HTS)-derived hit. Synthesis and structure-activity relationships (SAR) studies around the 3,5-dimethylphenyl and 4-methoxyphenyl regions resulted in the identification of a novel series of non-peptidic submicromolar NPBWR1 antagonists based on a 5-chloro-4-(4-alkoxyphenoxy)-2-(benzyl)pyridazin-3(2H)-one chemotype. Amongst them, 5-chloro-2-(9H-fluoren-9-yl)-4-(4-methoxyphenoxy)pyridazin-3(2H)-one 9h (CYM50769) inhibited NPW activation of NPBWR1 with a submicromolar IC(50), and displayed high selectivity against a broad array of off-targets with pharmaceutical relevance. Our medicinal chemistry study provides innovative non-peptidic selective NPBWR1 antagonists that may enable to clarify the biological role and therapeutic utility of the target receptor in the regulation of feeding behavior, pain, stress, and neuroendocrine function. [1] |
| Cell Assay |
Cell Proliferation Assay Cell Types: ATDC5 (24 hrs (hours) exposure to 200 ng/mL NPW-23) [1] Tested Concentrations: 1 and 3 μM Incubation Duration: 30 minutes Experimental Results: Attenuation of NPW-23-induced cell proliferation in a dose-dependent manner . |
| References |
[1]. SAR analysis of novel non-peptidic NPBWR1 (GPR7) antagonists. Bioorg Med Chem Lett. 2013 Feb 1;23(3):614-9. |
| Additional Infomation |
Amongst the synthesized compounds, 9h (CYM50769) was selected for further characterization. The solubility of 9h in a phosphate buffered saline (PBS) at pH 7.4 is 0.17 μM. The compound is non-cytotoxic to U2OS cells at 20 μM and chemically stable in PBS at pH 7.4 with half-life higher than 48 h. The selectivity profile was investigated against the Ricerca panel of off-target proteins including GPCRs, enzymes, transporters and ion channels at a concentration of 30 μM. Remarkably, out of 35 tested targets only CYP450 1A2, 5-HT2B and CYP450 2C19 showed 67%, 63% and 51% inhibition, respectively. In summary, we have reported the synthesis and SAR studies around the coil and 4-methoxyphenyl regions (a, b) of novel non-peptidic NPBWR1 antagonists based on a 5-chloro-4-(4-alkoxyphenoxy)-2-(benzyl)pyridazin-3(2H)-one chemotype. Small changes in region b had a negative impact on the potency, while the region a was found to interact with a liphophilic pocket that can accommodate a great variety of bulky quasi-planar substituents. Our studies resulted in the identification of a novel series of submicromolar NPBWR1 antagonists including 7y (CYM50719), 9h (CYM50769) and 15e (CYM50775) endowed with greater potency than our previously reported lead 1. Amongst them, 9h was further profiled and found to be highly selective against a broad array of off-targets with pharmaceutical relevance, making this compound suitable for further development. Our medicinal chemistry advances around this chemotype will be communicated in due course. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~239.89 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3989 mL | 11.9944 mL | 23.9889 mL | |
| 5 mM | 0.4798 mL | 2.3989 mL | 4.7978 mL | |
| 10 mM | 0.2399 mL | 1.1994 mL | 2.3989 mL |