CRX-526 is a novel and potent TLR4 antagonist,used to prevent advanced diabetic nephropathy.
Physicochemical Properties
| Molecular Formula | C69H127N2O19P |
| Molecular Weight | 1319.7438 |
| Exact Mass | 1318.88 |
| Elemental Analysis | C, 62.80; H, 9.70; N, 2.12; O, 23.03; P, 2.35 |
| CAS # | 245515-64-4 |
| Related CAS # | 245515-64-4; |
| PubChem CID | 24849153 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 16.254 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 19 |
| Rotatable Bond Count | 65 |
| Heavy Atom Count | 91 |
| Complexity | 1960 |
| Defined Atom Stereocenter Count | 9 |
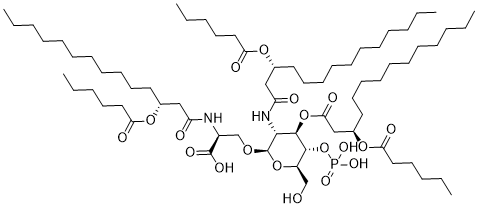
| SMILES | P(=O)(O)(O)O[C@@H]1[C@@H](CO)O[C@H]([C@@H]([C@H]1OC(C[C@@H](CCCCCCCCCCC)OC(CCCCC)=O)=O)NC(C[C@@H](CCCCCCCCCCC)OC(CCCCC)=O)=O)OC[C@@H](C(=O)O)NC(C[C@@H](CCCCCCCCCCC)OC(CCCCC)=O)=O |
| InChi Key | PRIXXGNJDNLMBH-DPGPRPECSA-N |
| InChi Code | InChI=1S/C69H127N2O19P/c1-7-13-19-22-25-28-31-34-40-43-54(85-61(75)46-37-16-10-4)49-59(73)70-57(68(79)80)53-84-69-65(71-60(74)50-55(86-62(76)47-38-17-11-5)44-41-35-32-29-26-23-20-14-8-2)67(66(58(52-72)88-69)90-91(81,82)83)89-64(78)51-56(87-63(77)48-39-18-12-6)45-42-36-33-30-27-24-21-15-9-3/h54-58,65-67,69,72H,7-53H2,1-6H3,(H,70,73)(H,71,74)(H,79,80)(H2,81,82,83)/t54-,55-,56-,57+,58-,65-,66-,67-,69-/m1/s1 |
| Chemical Name | (2S)-2-[[(3R)-3-hexanoyloxytetradecanoyl]amino]-3-[(2R,3R,4R,5S,6R)-3-[[(3R)-3-hexanoyloxytetradecanoyl]amino]-4-[(3R)-3-hexanoyloxytetradecanoyl]oxy-6-(hydroxymethyl)-5-phosphonooxyoxan-2-yl]oxypropanoic acid |
| Synonyms | CRX 526; CRX-526; 245515-64-4; UNII-8NLO017HHA; 8NLO017HHA; CHEMBL505526; (2S)-2-[[(3R)-3-hexanoyloxytetradecanoyl]amino]-3-[(2R,3R,4R,5S,6R)-3-[[(3R)-3-hexanoyloxytetradecanoyl]amino]-4-[(3R)-3-hexanoyloxytetradecanoyl]oxy-6-(hydroxymethyl)-5-phosphonooxyoxan-2-yl]oxypropanoic acid; Hexanoic acid, (1R)-1-(2-(((1S)-1-carboxy-2-((2-deoxy-3-O-((3R)-1-oxo-3-((1-oxohexyl)oxy)tetradecyl)-2-(((3R)-1-oxo-3-((1-oxohexyl)oxy)tetradecyl)amino)-4-O-phosphono-beta-D-glucopyranosyl)oxy)ethyl)amino)-2-oxoethyl)dodecyl ester; HEXANOIC ACID, (1R)-1-(2-(((1S)-1-CARBOXY-2-((2-DEOXY-3-O-((3R)-1-OXO-3-((1-OXOHEXYL)OXY)TETRADECYL)-2-(((3R)-1-OXO-3-((1-OXOHEXYL)OXY)TETRADECYL)AMINO)-4-O-PHOSPHONO-.BETA.-D-GLUCOPYRANOSYL)OXY)ETHYL)AMINO)-2-OXOETHYL)DODECYL ESTER; CRX-526; CRX526 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | TLR4 |
| ln Vitro |
CRX-526 inhibits the activation of human monocytes by LPS in vitro [1]> We have established previously that CRX-526, though structurally similar to lipid A (Fig. 1), did not by itself induce the production of proinflammatory cytokines from human monocytes. To determine whether CRX-526 interacts with the TLR4R complex, we tested whether CRX-526 could antagonize the proinflammatory properties of a known TLR4 ligand, LPS. Human monocyte-derived macrophages were preincubated with increasing concentrations of CRX-526 and then exposed to various concentrations of LPS. Preincubation of macrophages with CRX-526 resulted in an inhibition of TNF-α production after exposure to LPS (Fig. 2,A). In agreement with our previous studies, incubation of CRX-526 alone with macrophages did not result in any TNF-α production. A minimum w/w ratio of 5:1 CRX-526: LPS was necessary to see a clearly detectable inhibition of TNF-α production; a w/w ratio of 50:1 was required to completely abolish TNF-α production in response to LPS. Preincubation of human monocytes with CRX-526 also resulted in inhibition of IL-6, MIP-1α, and production after exposure to LPS (Fig. 2,B). Because IFN-γ activation of monocytes not only enhances the secretion of TNF-α but also is required for the production of biological active IL-12p70 in response to LPS, we determined the inhibitory effect of CRX-526 on the LPS-induced cytokines secretion in IFN-γ-activated monocytes (Fig. 2,C). In contrast to resting monocytes where the secretion of IL-6 and TNF-α were inhibited at similar levels (Fig. 2,B), CRX-526 had a differential inhibitory effect on the cytokine secretion of IFN-γ-activated monocytes in response to LPS (Fig. 2 C). A 10:1 w/w ratio of CRX-526 to LPS inhibited IL-12p70 and TNF-α production by 79 and 57%, respectively, but had no effect on the LPS-induced IL-6 production. In a murine system, CRX-526 inhibited the LPS-induced production of IL-6, IL-12p70, and TNF-α by murine splenic dendritic cells (data not shown), indicating that CRX-526 inhibits LPS signaling through either human or murine TLR4R complexes. Antagonism of LPS signaling by CRX-526 in human dendritic cells has also been demonstrated (P. Probst, unpublished observations). To confirm that CRX-526 is a pure antagonist and does not induce any gene transcription by signaling through the TLR4R complex, we used a custom microarray containing ∼300 inflammatory gene targets arranged in triplicate to look at mRNA expression in human monocytes after exposure to CRX-526 alone and/or in the presence of LPS. Exposure of monocytes to LPS alone (Fig. 2,D, left) stimulated the expression of >60 array elements >2-fold, while exposure to CRX-526 alone (Fig. 2,D, right) did not induce expression of any genes found on the microarray. Furthermore, pretreatment of monocytes with increasing amounts of CRX-526 before exposure to LPS completely suppressed all LPS-induced gene transcription (Fig. 2,D). NF-κB is known to be involved in the transcriptional regulation of many of the elements on this microarray (22). The level of inhibition by CRX-526 of representative NF-κB-regulated genes is summarized in Table I. Thus, CRX-526 can act as an antagonist for the TLR4R complex and can inhibit the ability of LPS to signal through the TLR4R complex. The antagonistic activity of CRX-526 is dependent on SAC length [1]> The structure of CRX-526 differs significantly from monophosphoryl lipid A (MPL) and other TLR4-agonist AGP in the length of its SAC: CRX-526 contains 3 SAC of 6 carbons in length, whereas MPL and other AGP, which signal through the TLR4R complex, contain SAC of ≥10 carbons in length (Fig. 1). To confirm whether hexanoic SAC are a requirement for the antagonistic activity of CRX-526, we synthesized molecules that are variants of CRX-526, containing either hexanoic or decanoic SAC at three different positions (Fig. 3). CRX-567 differs from CRX-526 by only a decanoic SAC at the right position, whereas CRX-568 has a decanoic SAC in the middle position, and CRX-570 has a decanoic SAC at the left position (compare Fig. 1 vs Fig. 3). These molecules were then tested for their ability to inhibit LPS signaling through the TLR4R complex. Using a previously described model system, HeLa cells were transfected transiently with DNA encoding TLR4, MD-2, and CD14, along with human IL-8 promoter-driver luciferase reporter plasmid and β-galactosidase vector. These transfected cells were stimulated with increasing concentrations of either AGP alone (⋄) or AGP + 100 ng/ml LPS (▪), and luciferase activity was monitored (Fig. 3). CRX-526 inhibited, in a dose-dependent manner, the ability of LPS to signal through the TLR4/MD2/CD14 complex. LPS was not able to stimulate HeLa cells that were transfected with a control vector nor did CRX-526 alone induce a signal in the TLR4/MD2/CD14-transfected cells (Fig. 3). CRX-567 has antagonistic activity for the TLR4/MD2/CD14R complex similar to CRX-526 (Fig. 3). However, CRX-568 has less antagonistic activity compared with CRX-526 or CRX-567 and even some partial agonist activity. CRX-570 has almost no antagonistic activity and significant agonistic activity. These findings show that a shortened (hexanoic) SAC on the left and middle position of the molecule are required for the inhibition of LPS signaling through the TLR4/MD2/CD14 complex by CRX-526. |
| ln Vivo |
CRX-526 can inhibit LPS-induced TNF-α release in vivo [1] Our data clearly show that CRX-526 can inhibit LPS signaling through the TLR4R complex in vitro. To test whether CRX-526 can also inhibit LPS activity in vivo, we injected LPS i.v. in the presence or absence of CRX-526 into mice and 1 h later measured TNF-α in the serum. We were able to detect significant amounts of TNF-α in the serum of mice given as little as 5 ng of LPS (data not shown). Therefore, we injected mice with either 5 ng of LPS alone or with increasing amounts of CRX-526. Significant inhibition of TNF-α release was seen with as little as 80 ng of CRX-526 (data not shown), and background levels of TNF-α were reached in the presence of 10 μg of CRX-526 (Fig. 4). These data confirm that CRX-526 acts as an antagonist for LPS in vivo as well as in vitro. CRX-526 can inhibit the development of disease in two different mouse models of colitis [1] Because LPS is the major inflammatory portion of the cell membrane of Gram-negative bacteria, we hypothesized that CRX-526 may be able to inhibit the ability of Gram-negative bacteria to cause or perpetrate the inflammation seen in IBD. Therefore, we tested the ability of CRX-526 to inhibit the development of disease in two different mouse models of colonic inflammation: dextran sodium sulfate-induced colitis, and MDR1a-deficient mice. Exposure to DSS in drinking water causes an acute colitis in mice due to damage to the epithelium layer. DSS-exposed animals show distinct clinical signs, including weight loss, changes in stool consistency, and blood in the stool and/or gross intestinal bleeding, that correlate directly with histological changes of the large intestine). Mice that are deficient in TLR4 expression do develop acute colitis when exposed to DSS. However, Lange et al. have observed that TLR4-mutant mouse strains, such as C3H/HeJ and C57BL/10ScN, show delayed onset of intestinal bleeding compared with strain-matched TLR4-wild-type mice and show increased survival upon exposure to DSS for ≥10 days. Their data indicate that TLR4 may play a minor, but detectable, role in DSS-induced colitis. Therefore, we exposed BALB/c females to DSS for 7 days and simultaneously treated the mice with CRX-526. Mice were assessed for clinical signs of colitis (DAI) and for histological changes in the large intestine on day 7. As shown in Fig. 5, exposure to DSS caused the development of significant clinical and histological changes in these mice. Furthermore, in agreement with previous studies, DAI correlated with the histological score. Cotreatment with CRX-526 significantly decreased the histological score at all doses tested but decreased DAI only at the 10- and 50-μg doses (experiment no. 1, Fig. 5,A). These data show that blocking TLR4R complex signaling can decrease disease severity in an acute model of colitis and that a decrease in DAI is a rigorous measure of a decrease in disease severity in our model system. A repeat experiment confirmed the ability of 50 μg of CRX-526 to significantly decrease the DAI score in DSS-exposed mice (experiment no. 2, Fig. 5,B). To confirm that our results were due to inhibition of signaling through the TLR4R complex, we compared the ability of CRX-526 to affect disease severity in TLR4-wild-type (BALB/cJ) and TLR4-mutant (C.C3H.Tlr4Lps-d) mice. Both TLR4-wild-type and TLR4-mutant mice developed acute disease when exposed to DSS, although the TLR4-mutant mice had a significantly lower DAI (mean DAI of 1.9 ± 0.4 and 1.4 ± 0.2, respectively, for TLR4-wild-type and TLR4-mutant mice; p = 0.004; Fig. 6). Treatment with 50 μg of CRX-526 by s.c. injection significantly decreased the DAI score in TLR4-wild-type but not TLR4-mutant mice (Fig. 6). These data confirm that the decreased disease severity seen in CRX-526-treated animals is due to blocking of TLR4R complex signaling. Interestingly, treatment of TLR4-wild-type mice with CRX-526 brought the DAI score to a level similar to that of the TLR4-mutant mice (mean DAI of 1.4 ± 0.3 for TLR4-mutant mice and 1.3 ± 0.8 for CRX-526-treated TLR4-wild-type mice), suggesting that at the 50-μg dose we obtained an almost complete inhibition of TLR4R complex signaling (Fig. 6). Current evidence indicates that the chronic inflammation observed in the intestines of patients with inflammatory bowel disease is due to an aberrant immune response to enteric flora. Researchers have developed a lipid A-mimetic, CRX-526, which has antagonistic activity for TLR4 and can block the interaction of LPS with the immune system. CRX-526 can prevent the expression of proinflammatory genes stimulated by LPS in vitro. This antagonist activity of CRX-526 is directly related to its structure, particularly secondary fatty acyl chain length. In vivo, CRX-526 treatment blocks the ability of LPS to induce TNF-alpha release. Importantly, treatment with CRX-526 inhibits the development of moderate-to-severe disease in two mouse models of colonic inflammation: the dextran sodium sulfate model and multidrug resistance gene 1a-deficient mice. By blocking the interaction between enteric bacteria and the innate immune system, CRX-526 may be an effective therapeutic molecule for inflammatory bowel disease [1]. |
| Cell Assay |
Human monocyte-derived macrophages stimulation with LPS [1] Human PBMC were isolated by Ficoll-Hypaque 1.077 centrifugation of leukapheresis product before aliquoting and freezing. Monocytes and monocyte-derived macrophages were prepared by an adherent step as described previously. Phenotypic analysis of the cells revealed that >95% of the cells are CD14 positive. Stimulation of human monocyte-derived macrophages in the presence of LPS and/or AGP has been described previously in detail. Briefly, monocyte-derived macrophages were exposed to CRX-526 at various concentrations. Thirty minutes later, LPS was added to the cultures. After 6 h of stimulation, the supernatant was removed and analyzed by ELISA for the presence of TNF-α. RNA was isolated from adherent cells as previously described for the generation of cDNA for microarray analysis. Inhibition of LPS-induced cytokine production in monocytes [1] Human monocytes were generated from 4 × 106 PBMC by an adherent step and were cultured for 48 h in RPMI 1640 medium containing 2% human AB serum. For experiments determining IL-12p70 production, the culture medium was supplemented with IFN-γ (1000 U/ml) for the last 24 h. After incubating with CRX-526 for 30 min, monocytes were stimulated with 100 ng/ml E. coli LPS. Cytokines and MIP-1α were determined in supernatants taken after 18 h. Quantification of IL-6, IL-12p70, and TNF-α production was done by cytokine-specific ELISA as described previously. The working sensitivity of all ELISA was shown to be ∼10 pg/ml. MIP-1α and murine TNF-α levels were determined by using ELISA kits, according to the manufacturer’s protocol. Cytokines were not detectable in the absence of stimulation. HeLa cell transfection and reporter assay [1] A previously described HeLa transient transfection system was used to test the ability of CRX-526, CRX-567, CRX-568, and CRX-570 to act as antagonist/agonist for the TLR4-MD2-CD14R complex. Briefly, HeLa cells were transfected with 50 ng of IL-8 promoter-derived luciferase reporter and 10 ng each of TLR4, MD-2, and CD14 plasmids. Eighteen hours after transfection, cells were stimulated with varying concentrations of AGP in the presence of vehicle or 100 ng/ml LPS for 6 h, lysed, and then analyzed for luciferase and β-galactosidase activity. |
| Animal Protocol |
DSS model of intestinal inflammation [1] Two separate protocols were used to cause DSS-induced intestinal inflammation, depending on the strain of mice and the lot of DSS used. For experiment no. 1 (see Fig. 1), BALB/cAnN females were given 4.0% DSS in their water ad libitum starting on day 0, with all mice sacrificed on day 7. Negative control animals remained on standard facility water. Starting on day 0, mice were given s.c. injections of either AF vehicle or varying doses of CRX-526-AF every other day for a total of four doses (n = 5/group). For experiment no. 2 and Fig. 5, BALB/cJ females and C.C3H Tlr4Lps-d/J mice were given 3.0% DSS in their drinking water from days 0 to 7 and were s.c. injected daily with either AF-vehicle or 50 μg of CRX-526-AF from days −3 through 6 for a total of nine doses (n = 10/group). Disease activity index (DAI), a clinical score that factors in weight loss, changes in stool consistency, and the presence of blood in the stool was assessed by a previously published method. Paraffin-embedded sections of the large intestines, prepared as “Swiss rolls”, were stained with H&E, and histological scoring of disease in the cecums and colons was determined in a blinded fashion by a certified veterinary pathologist (H. Bielefeldt-Ohmann). Briefly, the cecum, proximal colon, mid-colon, distal colon, and rectum were assessed for loss of mucosal architecture (0–4), the presence of mucoid crypt cysts (0–4), goblet cell depletion (0–4), erosion (0–4), edema (0–4), mononuclear cell infiltration (0–4), polymorphonucleocyte and/or eosinophil infiltration (0–4), and transmural inflammation (0–4). To obtain a total score for each mouse, the total scores for each segment were added together; the maximal possible score is 160. Treatment and assessment of colitis in MDR1a-deficient mice [1] Starting at 5–6 wk of age, MDR1a-deficient female mice were injected s.c. on a weekly basis with 50 μg (experiment no. 1) or 100 μg of CRX-526-TeoA or with an equal volume of TeoA alone (vehicle treated). Treatment continued for 5–6 wk, and then, mice were sacrificed and the tissues prepared as described above. Histological scoring of disease in the large intestine was performed in a blinded fashion by Dr. H. Bielefeldt-Ohmann, using a previously described scoring system. Briefly, the cecum, proximal colon, distal colon, and rectum from each mouse were scored on severity of mucosal epithelial changes (0–4), degree of inflammation (0–4), the percentage of intestinal segment affected in any manner (extent score A), and the percentage of intestinal segment affected by the most severe score (≥2) given for inflammation or mucosa (extent score B). The segment score was determined by summing the severity scores (mucosal score + inflammation score + extent score A + extent score B), and then, the segment scores were added together to reach a total score for each mouse, with a maximum possible score of 64. |
| References | [1]. Anti-inflammatory effect of miltirone on inflammatory bowel disease via TLR4/NF-κB/IQGAP2 signaling pathway. Biomed Pharmacother. 2017 Jan;85:531-540. |
| Additional Infomation | Although the exact mechanism of action of CRX-526 is still under investigation, we hypothesize that CRX-526 binds directly to the TLR4R complex and sterically inhibits the ability of LPS and, perhaps, other ligands to bind. We have demonstrated previously that MPL and other AGP molecules do not induce cytokine production from cells lacking TLR4 expression, suggesting these lipid A-derived molecules are strictly ligands for TLR4. Recently, it has been shown by Akashi et al. that LPS binds directly to the TLR4-MD-2 complex, while CD14 can augment this binding. They also tested a TLR4R complex antagonist, E5531, and showed it also bound directly to TLR4-MD-2. As E5531 has very similar LPS-antagonist activity compared with CRX-526 it is possible that CRX-526 also binds directly to a TLR4-MD-2 complex. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.7577 mL | 3.7886 mL | 7.5773 mL | |
| 5 mM | 0.1515 mL | 0.7577 mL | 1.5155 mL | |
| 10 mM | 0.0758 mL | 0.3789 mL | 0.7577 mL |