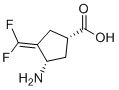
CPP-115 is a novel and potent GABA-aminotransferase inhibitor
Physicochemical Properties
| Molecular Formula | C7H9F2NO2 |
| Molecular Weight | 177.1508 |
| Exact Mass | 177.06 |
| Elemental Analysis | C, 47.46; H, 5.12; F, 21.45; N, 7.91; O, 18.06 |
| CAS # | 640897-20-7 |
| Related CAS # | 760947-97-5 (HCl) ; 640897-20-7 |
| PubChem CID | 9794014 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.659 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 12 |
| Complexity | 236 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | N[C@@H]1/C(=C(\F)/F)/C[C@H](C(O)=O)C1 |
| InChi Key | CBSRETZPFOBWNG-UCORVYFPSA-N |
| InChi Code | InChI=1S/C7H9F2NO2/c8-6(9)4-1-3(7(11)12)2-5(4)10/h3,5H,1-2,10H2,(H,11,12)/t3-,5-/m0/s1 |
| Chemical Name | (1S,3S)-3-amino-4-(difluoromethylene)cyclopentane-1-carboxylic acid |
| Synonyms | CPP115; CPP 115; CPP-115 free base; CPP-115; 640897-20-7; UNII-5TD9324Z2U; 5TD9324Z2U; CHEMBL146927; (1S,3S)-3-Amino-4-difluoromethylenyl-1-cyclopentanoic acid; (+)-(1S,4S)-4-Amino-3-(difluoromethylene)-1-cyclopentanecarboxylic acid CPP-115 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | GABA-aminotransferase |
| ln Vitro | GABA aminotransferase is an enzyme that degrades GABA, the principal inhibitory neurotransmitter in the brain. CPP-115 increases GABA levels in the brain by inhibiting GABA aminotransferase with a 187× greater potency than vigabatrin. [1] |
| ln Vivo |
CPP-115 is a GABA aminotransferase inactivator and a highly potent analog of vigabatrin, a first-line therapeutic for infantile spasms. CPP-115 has shown markedly lower retinal toxicity than vigabatrin, a major adverse effect reported in patients. CPP-115 was granted orphan drug designation for the treatment of infantile spasms.[1] A proof-of-concept study of CPP-115 in the multiple-hit rat model of infantile spasms demonstrated that it decreased spasms and normalized EEG activity at considerably lower and better tolerated doses than vigabatrin Results showed that CPP-115 decreased spasms at a dose 400× lower than vigabatrin with effects lasting almost three times longer. CPP-115 also demonstrated effectiveness in the 6-Hz, corneal kindling, and picrotoxin-induced seizure models as part of the National Institutes of Health’s (NIH’s) anticonvulsant screening program.[1] |
| Enzyme Assay | In this study, researchers report a novel inactivation mechanism for CPP-115, a mechanism-based inactivator that undergoes GABA-AT-catalyzed hydrolysis of the difluoromethylene group to a carboxylic acid with concomitant loss of two fluoride ions and coenzyme conversion to pyridoxamine 5'-phosphate (PMP). The partition ratio for CPP-115 with GABA-AT is about 2000, releasing cyclopentanone-2,4-dicarboxylate (22) and two other precursors of this compound (20 and 21). Time-dependent inactivation occurs by a conformational change induced by the formation of the aldimine of 4-aminocyclopentane-1,3-dicarboxylic acid and PMP (20), which disrupts an electrostatic interaction between Glu270 and Arg445 to form an electrostatic interaction between Arg445 and the newly formed carboxylate produced by hydrolysis of the difluoromethylene group in CPP-115, resulting in a noncovalent, tightly bound complex. This represents a novel mechanism for inactivation of GABA-AT and a new approach for the design of mechanism-based inactivators in general.[2] |
| Animal Protocol | CPP-115 has been granted fast track status for the treatment of cocaine dependency and orphan drug status for infantile spasms. A recent case report published the results from an open-label investigation for pharmacologically resistant infantile spasms in a 3-year-old boy. Prior to investigational therapy, the patient was reported to fail 10 drugs and ketogenic diet. The patient had approximately 100 seizures per day before administration of CPP-115. One year after starting CPP-115 and tapering off clobazam and vigabatrin, his reported seizure frequency decreased to 25–30 seizure per day. The patient’s cognition and behavior also improved, and there was no evidence of retinal damage. CPP-115 shows promising results for management of infantile spasms and more comprehensive analysis in a controlled setting is warranted. |
| ADME/Pharmacokinetics | A phase I safety, tolerability, and pharmacokinetic study of CPP-115 has been completed to evaluate oral doses of CPP-115 (NCT01493596). The study demonstrated favorable pharmacokinetics with rapid and complete oral absorption and elimination. CPP-115 demonstrated low potential for drug–drug interaction, showing no inhibition or induction of the 12 most common CYP enzymes |
| References |
[1]. https://www.tandfonline.com/doi/full/10.1080/17512433.2018.1386553#d1e807 [2]. Mechanism of inactivation of γ-aminobutyric acid aminotransferase by (1S,3S)-3-amino-4-difluoromethylene-1-cyclopentanoic acid (CPP-115). J Am Chem Soc. 2015 Feb 25;137(7):2628-40. |
| Additional Infomation | See also: Cpp-115 (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.6449 mL | 28.2247 mL | 56.4493 mL | |
| 5 mM | 1.1290 mL | 5.6449 mL | 11.2899 mL | |
| 10 mM | 0.5645 mL | 2.8225 mL | 5.6449 mL |