Physicochemical Properties
| Molecular Formula | C18H19NO4 |
| Molecular Weight | 313.34776 |
| Exact Mass | 313.131 |
| CAS # | 223532-02-3 |
| PubChem CID | 44374998 |
| Appearance | White to off-white solid powder |
| Density | 1.274 |
| LogP | 2.39 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 23 |
| Complexity | 387 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | PONANXDRJJIGPG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H19NO4/c20-17(12-15-9-5-2-6-10-15)19(23)13-16(18(21)22)11-14-7-3-1-4-8-14/h1-10,16,23H,11-13H2,(H,21,22) |
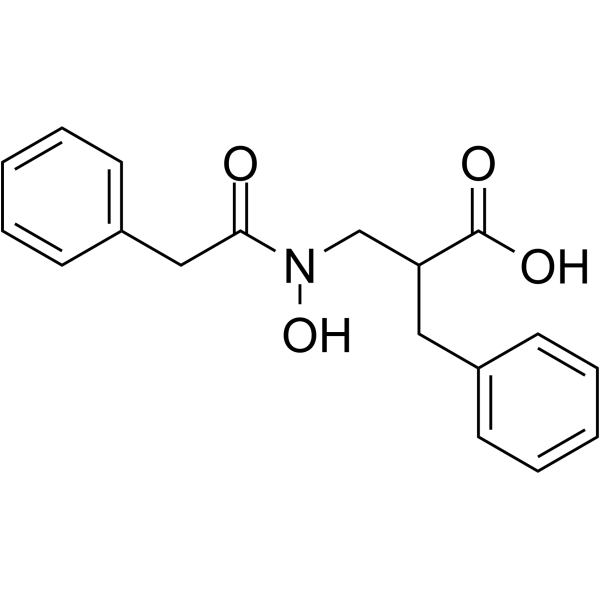
| Chemical Name | 2-benzyl-3-[hydroxy-(2-phenylacetyl)amino]propanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
CPA inhibitor (N-acyl-N-hydroxy-β-phenylalanines) targets carboxypeptidase A (CPA) with varying Ki values: compound 3a (Ki = 0.35 μM), compound 3b (Ki = 0.42 μM), compound 3c (Ki = 0.58 μM), compound 3d (Ki = 0.71 μM), compound 3e (Ki = 0.95 μM) [1] |
| ln Vitro |
CPA inhibitor (N-acyl-N-hydroxy-β-phenylalanines) exhibited potent in vitro inhibitory activity against carboxypeptidase A (CPA). All tested analogs showed dose-dependent inhibition of CPA, with compound 3a being the most active (Ki = 0.35 μM) and compound 3e the least active (Ki = 0.95 μM) among the series. The inhibition was determined to be competitive, as indicated by Lineweaver-Burk plots showing increased Km values without changes in Vmax, suggesting binding of the inhibitors to the active site of CPA [1] |
| Enzyme Assay |
The enzyme activity assay for CPA inhibition was performed using Hippuryl-L-phenylalanine as the substrate. The reaction mixture contained a suitable buffer, CPA enzyme, different concentrations of CPA inhibitor (N-acyl-N-hydroxy-β-phenylalanines), and the substrate. The mixture was incubated at 37°C for a specified period to allow the enzymatic reaction. The release of hippuric acid, a product of substrate hydrolysis, was measured spectrophotometrically at a specific wavelength. The initial reaction rates were calculated from the absorbance data, and Ki values were determined by Lineweaver-Burk linear regression analysis of the rate data obtained at multiple substrate and inhibitor concentrations [1] |
| References |
[1]. First hydroxamate inhibitors for carboxypeptidase A. N-acyl-N-hydroxy-beta-phenylalanines. Bioorg Med Chem Lett. 1999 Mar 8;9(5):691-6. |
| Additional Infomation |
CPA inhibitor (N-acyl-N-hydroxy-β-phenylalanines) represent the first reported hydroxamate-based inhibitors of carboxypeptidase A. The inhibitory activity is attributed to the hydroxamate moiety (-CONHOH) of the compounds, which is proposed to chelate the zinc ion in the active center of CPA, a metalloenzyme. The structure-activity relationship (SAR) analysis revealed that the length and substitution pattern of the N-acyl chain influence inhibitory potency, with shorter acyl chains (e.g., acetyl in compound 3a) conferring higher activity than longer or substituted chains [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~319.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.98 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.98 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.98 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1913 mL | 15.9566 mL | 31.9132 mL | |
| 5 mM | 0.6383 mL | 3.1913 mL | 6.3826 mL | |
| 10 mM | 0.3191 mL | 1.5957 mL | 3.1913 mL |