Physicochemical Properties
| Molecular Formula | C28H28O4 |
| Molecular Weight | 428.51952 |
| Exact Mass | 428.198 |
| CAS # | 158081-99-3 |
| PubChem CID | 9867257 |
| Appearance | White to light yellow solid powder |
| LogP | 5.5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 32 |
| Complexity | 627 |
| Defined Atom Stereocenter Count | 2 |
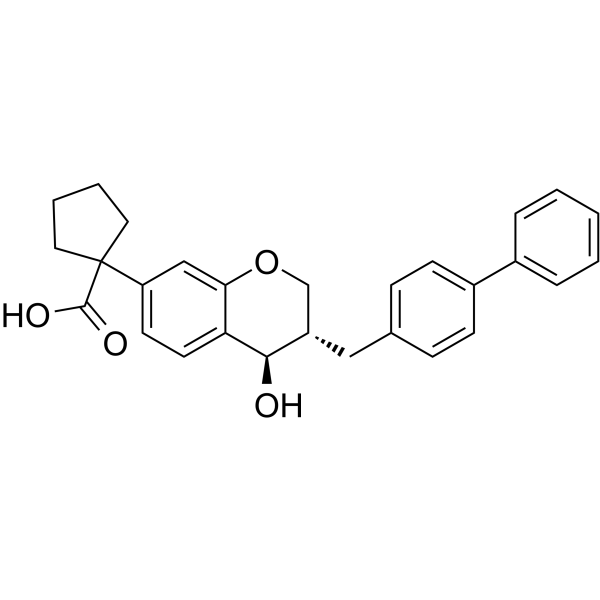
| SMILES | O=C(C1(C2=CC=C3[C@H](O)[C@@H](CC4=CC=C(C5=CC=CC=C5)C=C4)COC3=C2)CCCC1)O |
| InChi Key | KMNLXCBYBKHKSK-BKMJKUGQSA-N |
| InChi Code | InChI=1S/C28H28O4/c29-26-22(16-19-8-10-21(11-9-19)20-6-2-1-3-7-20)18-32-25-17-23(12-13-24(25)26)28(27(30)31)14-4-5-15-28/h1-3,6-13,17,22,26,29H,4-5,14-16,18H2,(H,30,31)/t22-,26+/m0/s1 |
| Chemical Name | 1-[(3S,4R)-4-hydroxy-3-[(4-phenylphenyl)methyl]-3,4-dihydro-2H-chromen-7-yl]cyclopentane-1-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | CP-105696 is an extremely selective and structurally unique LTB4 receptor antagonist. With an IC50 value of 8.42±0.26 nM, CP-105696 inhibits the binding of [3H]LTB4 (0.3 nM) to the high-affinity LTB4 receptor on human neutrophils in vitro. When [3H]LTB4 binds to these high-affinity receptors, CP-105696 binds to the receptors noncompetitively, as revealed by Scatchard analysis. With an IC50 value of 5.0±2.0 nM, CP-105696 inhibits human neutrophil chemotaxis mediated by LTB4 (5 nM) in a non-competitive manner. By using Scatchard analysis, it is possible to determine that CP-105696 functions as a competitive antagonist for [3H]LTB4 binding to low-affinity receptors on neutrophils and that it competitively inhibits LTB4-mediated inhibition of human neutrophil CD11b was increased (pA2= 8.03±0.19). At 10 μM, CP-105696 does not prevent CD11b overexpression or human neutrophil chemotaxis that is mediated by G protein-coupled chemokine receptors other than C5a, IL-8, or PAF. CP-105696, with an IC50 value of 940±70 nM, inhibits LTB4 (5 nM)-mediated Ca2+ mobilization in isolated human monocytes[1]. |
| ln Vivo | B10.BR (H2k) allografts transplanted into C57Bl/6 (H2b) recipients were considerably protected as compared with control grafts at a dose of 50 mg/kg/day (28 days) Reflected by mean survival time (27 ± 20 days) [n = 10] vs. 12 ± 6 days [n = 14]; P = 0.0146). Using the induction regimen (days -1 to 3), CP-105696 at 100 mg/kg/day significantly increased allograft survival (33±23 days [n=9]; P=0.0026) , but not 10 mg of CP-105696/kg/day (18±16 days [n=8]; P=0.1433). Syngeneic grafts survived indefinitely (n=11). Immunohistological assessment of allograft rejection revealed a mononuclear cell infiltrate comprised predominantly of CD3+ and CD11b+ (Mac-1+) cells, which is rarely found in syngeneic grafts. Allografts from mice treated with 50 or 100 mg/kg/day CP-105696 revealed preferential reduction in β2-integrin (Mac-1) expression on monocytes/macrophages, consistent with allogeneic This was verified by the density of CD11b staining relative to allograft controls [2]. |
| References |
[1]. The in vitro and in vivo pharmacologic activity of the potent and selective leukotriene B4 receptor antagonist CP-105696. J Pharmacol Exp Ther. 1995 Apr;273(1):176-84. [2]. Antagonizing leukotriene B4 receptors delays cardiac allograft rejection in mice. Transplantation. 1999 Mar 27;67(6):808-15. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~233.36 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3336 mL | 11.6681 mL | 23.3361 mL | |
| 5 mM | 0.4667 mL | 2.3336 mL | 4.6672 mL | |
| 10 mM | 0.2334 mL | 1.1668 mL | 2.3336 mL |