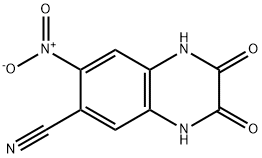
CNQX (FG9065; FG-9065; 6-cyano-7-nitroquinoxaline-2,3-dione) is an AMPA/kainate antagonist that inhibits AMPA and kainate receptors with IC50 values of 0.3 μM, 1.5 μM, respectively. CNQX is also an antagonist at the glycine modulatory site on the NMDA receptor complex with an IC50 of 25 μM). CNQX can be used to isolate GABAA receptor mediated spontaneous inhibitory postsynaptic currents and antagonizes non-NMDA receptor-mediated responses in cultured cerebellar granule cells. CNQX shows neuroprotective effects in models of ischemia and inhibits seizure-like activity in hippocampal neurons.
Physicochemical Properties
| Molecular Formula | C₉H₄N₄O₄ |
| Molecular Weight | 232.15 |
| Exact Mass | 232.023 |
| CAS # | 115066-14-3 |
| Related CAS # | CNQX disodium;479347-85-8 |
| PubChem CID | 3721046 |
| Appearance | White to yellow solid powder |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 659.3ºC at 760 mmHg |
| Melting Point | 300 °C |
| Flash Point | 352.6ºC |
| Vapour Pressure | 0.001mmHg at 25°C |
| Index of Refraction | 1.699 |
| LogP | 0.64 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 17 |
| Complexity | 434 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | RPXVIAFEQBNEAX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C9H4N4O4/c10-3-4-1-5-6(2-7(4)13(16)17)12-9(15)8(14)11-5/h1-2H,(H,11,14)(H,12,15) |
| Chemical Name | 7-nitro-2,3-dioxo-1,4-dihydroquinoxaline-6-carbonitrile |
| Synonyms | FG9065; 6-CYANO-7-NITROQUINOXALINE-2,3-DIONE; 7-Nitro-2,3-dioxo-1,2,3,4-tetrahydroquinoxaline-6-carbonitrile; 7-nitro-2,3-dioxo-1,4-dihydroquinoxaline-6-carbonitrile; 6-Quinoxalinecarbonitrile, 1,2,3,4-tetrahydro-7-nitro-2,3-dioxo-; FG 9065; FG-9065;FG 9065 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
AMPA (IC50 = 0.3 μM); kainate receptor (IC50 = 1.5 μM)[1] AMPA/Kainate receptors (non-NMDA subtype of glutamate receptors). The study demonstrates a clear preference of CNQX for non-NMDA receptors. [1] Non-NMDA type glutamate receptors, with higher affinity for the quisqualate receptor subtype compared to NMDA and kainate receptor subtypes (as referenced from binding studies). [2] Non-NMDA subtype of excitatory amino acid receptors (specifically kainate and quisqualate/AMPA receptors). The study confirms its selectivity against NMDA receptors. [3] CNQX is an antagonist of the AMPA/kainate subtype of ionotropic glutamate receptors. The text also notes that compounds in the quinoxalinedione series, including CNQX, have inhibitory activity at kainate receptors and have been reported to show affinity for the glycine site of the NMDA receptor. [4] |
| ln Vitro |
CNQX (FG9065; 2–5 μM) avoids both fast and slow GABA-mediated inhibition of hippocampal slice perfusion while reversibly dilating Schaffer collaterals and filamentous excitatory postsynaptic potentials (EPSPs) [2]. The amplitude of the monosynaptic component of DR-VRR recorded from lumbar segments with dancing excitation is reduced when CNQX (1–5 μM) is grounded [3]. In electrophysiological recordings from rat spinal neurons in vivo, CNQX (ejected by electrophoresis from a 1 mM solution) reduced the excitatory responses induced by the agonists quisqualate and kainate approximately equally. For example, in one neuron, CNQX (20 nA) reduced the increase in firing rate caused by both quisqualate and kainate. The effect had a rapid onset and recovery. In contrast, CNQX had little to no effect on the excitatory response induced by N-methyl-DL-aspartate (NMA, an NMDA receptor agonist). The study notes that CNQX was considerably more potent than another compound, GAMS, in reducing quisqualate and kainate responses.[1] In receptor binding experiments referenced in the discussion, CNQX was noted to be a relatively weak inhibitor of [3H]kainate binding (specifically at the Ca2+-sensitive high-affinity site) compared to its inhibitory activity in the [3H]AMPA binding assay. This suggests its functional antagonism under physiological conditions may primarily involve quisqualate (AMPA) receptors.[1] In the rat hippocampal slice preparation, superfusion with CNQX (2-5 µM) completely and reversibly blocked the excitatory postsynaptic potential (EPSP) evoked by stimulation of the Schaffer collateral pathway in the CA1 region and the mossy fiber pathway in the CA3 region. The blockade was rapid (approximately 2 minutes with 4 µM CNQX) and recovery required up to 30 minutes of washout. The synaptic blockade by CNQX was not associated with changes in membrane potential, input resistance, or spike accommodation in the recorded neurons.[2] CNQX (2-4 µM) abolished synaptic excitation without altering the amplitude of the fast and slow inhibitory postsynaptic potentials (IPSPs) mediated by GABA, suggesting it does not affect feedforward inhibitory pathways.[2] In voltage-clamp experiments on CA3 neurons, CNQX demonstrated selectivity in reducing agonist-induced inward currents. The current induced by quisqualate (10 µM) was reduced to 56 ± 17% of control by 4 µM CNQX and to 32 ± 4% by 10 µM CNQX. The current induced by NMDA (20 µM) was reduced to a lesser extent (77 ± 14% and 66 ± 11% of control by 4 µM and 10 µM CNQX, respectively, after 5 minutes exposure). The inward current induced by kainate (200 nM) was slightly reduced by 10 µM CNQX (78 ± 17% of control). In contrast, the NMDA receptor antagonists AP-5 or AP-7 (50 µM) reduced NMDA-induced currents but not those induced by quisqualate or kainate. The order of antagonistic potency of CNQX against the tested agonists was quisqualate > kainate = NMDA.[2] In the isolated lamprey spinal cord preparation, bath application of CNQX (5 µM) blocked the fast, non-NMDA receptor-mediated component of compound monosynaptic excitatory postsynaptic potentials (EPSPs) recorded in motoneurons. When applied in normal Ringer's solution containing physiological Mg²⁺ concentration, CNQX left a smaller, slower component of the EPSP, which was subsequently blocked by the NMDA receptor antagonist AP5, indicating CNQX's selectivity for non-NMDA receptors.[3] CNQX (at doses up to 15 µM) had no effect on the rhythm or organization of fictive locomotion (a motor pattern) induced by bath application of the NMDA receptor agonist N-methyl-D,L-aspartate (NMA, 100 µM).[3] |
| ln Vivo |
CNQX (FG9065; 2–5 μM) avoids both fast and slow GABA-mediated inhibition of hippocampal slice perfusion while reversibly dilating Schaffer collaterals and filamentous excitatory postsynaptic potentials (EPSPs) [2]. The amplitude of the monosynaptic component of DR-VRR recorded from lumbar segments with dubbing is approximately decreased by CNQX (1 -5 μM) [3]. In the first 15 minutes of the cocaine-free phase, CNQX disodium (FG9065 disodium; 0.75–3 mg/kg; i.p.; 20 minutes before to testing) dose-dependently decreases the number of cocaine reactions [4]. Ten minutes prior to the retention test, CNQX disodium (0.5 or 1.25 μg) can be infused bilaterally into the amygdala or dorsal hippocampus to partially decrease the expression of hypotensive inhibitory avoidance in rats 24 hours post-training. At 0.5 μg, CNQX disodium completely blocks the channel [5]. The motor pattern underlying locomotion in the lamprey is activated and maintained by excitatory amino acid neurotransmission. The quinoxalinediones 6,7-dinitroquinoxaline-2,3-dione (DNQX) and 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) are potent and selective antagonists of non-N-methyl-D-aspartate (NMDA) receptors in the mammalian central nervous system. In the lamprey, these compounds are now shown to block fast excitatory synaptic potentials elicited in neurones of the spinal ventral horn. They selectively antagonise responses to the application of selective kainate and quisqualate receptor agonists (kainate and alpha-amino-3-hydroxy-5-methyl-4-isoxalone (AMPA)) but do not influence NMDA receptor-mediated responses. Additionally, it is shown that the activation of NMDA receptors is sufficient to elicit and maintain fictive locomotion after blockade of non-NMDA receptors with either DNQX or CNQX. Conversely, activation of quisqualate receptors with AMPA, but not quisqualate leads to fictive locomotion with properties much like that activated by kainate[3]. Increasing the cocaine unit dose increased responding during the first and second intervals, with a decrease in the latency to the first CS. CNQX decreased the number of cocaine responses in a dose-dependent manner during the first 15-min cocaine-free interval, but did not affect cocaine responding during either the second interval or the latter part of the session under the FR4(FR7:S) schedule. In the locomotor activity test, reductions in rearing were produced by higher CNQX doses than those that attenuated significantly responding during the first fixed interval. Conclusions: These results suggest that AMPA/kainate receptors are involved in mediation of cocaine-seeking behaviour controlled partly by cocaine-associated cues[4]. The involvement of non-N-methyl-D-aspartate receptors in the amygdala in the expression of conditioned fear was examined using the fear-potentiated startle paradigm. Rats implanted with bilateral cannulae in the basolateral amygdaloid nuclei received 10 pairings of either a visual or auditory conditioned stimulus with footshock on each of 2 days. The next day, they were tested by eliciting the acoustic startle reflex in the presence or absence of the conditioned stimulus and divided into groups with equivalent levels of potentiation. One or two days later, rats were tested again following intra-amygdala infusion of vehicle or 0.025, 0.25, or 2.5 micrograms of 6-cyano-7-nitroquinoxaline-2,3-dione. The drug dose-dependently blocked the expression of potentiated startle in both sensory modalities, indicating that activation of non-NMDA receptors in the amygdala is necessary for the expression of conditioned fear[5]. 1. Systemic intraperitoneal (IP) administration of the AMPA/kainate receptor antagonist CNQX (0.75, 1.5, and 3 mg/kg) dose-dependently decreased the number of active lever responses during the first cocaine-free, fixed interval (FI15 min) under a second-order schedule of cocaine reinforcement in rats. This attenuation was specific to the initial "cocaine-seeking" phase, as responding during the second interval (after the first cocaine infusion) and the total number of cocaine infusions self-administered later in the session were not affected.[4] 2. In a separate locomotor activity test, CNQX at 3 mg/kg IP significantly suppressed vertical activity (rearing) but did not decrease horizontal activity; in fact, it increased horizontal activity during a specific time period. The attenuation of cocaine-seeking by the 3 mg/kg dose may be partially related to motor-impairing effects, but lower doses that attenuated seeking did not significantly affect rearing.[4] |
| Enzyme Assay | Superfusion of hippocampal slices with 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX, 2-5 microM) reversibly blocked the Schaffer collateral and mossy fibre excitatory postsynaptic potential (EPSP), while sparing the fast and slow gamma-aminobutyric acid (GABA)-mediated inhibition. Membrane potential, input resistance and spike accommodation were not altered. Inward currents induced by quisqualate were reduced to a greater extent by CNQX than those induced by kainate or N-methyl-D-aspartate. We suggest that CNQX may be a useful antagonist to study excitatory amino acid-mediated synaptic transmission[2]. |
| Animal Protocol |
Animal/Disease Models: Male Wistar rats, body weight 180-200 g[4] Doses: 0.75, 1.5 and 3 mg/kg Route of Administration: IP; 20 minutes before test Experimental Results: During the first 15 minutes cocaine-free interval, Reduces the number of cocaine (IV; 0.25 mg/infusion) responses in a dose-dependent manner Electrophysiological experiments were performed on pentobarbital-anesthetized rats. Action potentials from single neurons in the spinal cord (dorsal horn) were recorded using the center barrel of seven-barreled microelectrodes filled with 3.6M NaCl. The outer barrels of the microelectrode contained drug solutions for ejection by electrophoresis (iontophoresis). Once a neuron was located, baseline excitatory responses were established by cyclical ejection of the agonists quisqualate, kainate, and NMA. CNQX was dissolved at a concentration of 1 mM in a 200 mM CaCl2 solution (pH adjusted to 9.8) for loading into a microelectrode barrel. It was then ejected near the recorded neuron using electrophoretic current (e.g., 20 nA) to study its effects on the agonist-induced neuronal firing rates.[1] Transverse hippocampal slices (500 µm thick) were prepared from male Wistar rats (90-125 g). The slices were transferred to a submerged-type recording chamber and continuously superfused (2 ml/min) with oxygenated artificial cerebrospinal fluid (ACSF) at 33-34°C. The ACSF composition was (in mM): NaCl 126, KCl 3.5, MgCl2 1.3, NaH2PO4 1.2, CaCl2 2, NaHCO3 25, and glucose 11, gassed with 95% O2 / 5% CO2 to a pH of 7.3. For intracellular recording and voltage-clamp experiments, microelectrodes were used. CNQX was applied by bath superfusion at concentrations ranging from 2 to 10 µM. In some voltage-clamp experiments, tetrodotoxin (TTX, 1 µM) was used to block voltage-gated sodium channels, and microelectrodes were filled with 3 M CsCl to reduce potassium currents.[2] Experiments were performed on adult river lampreys (Lampetra fluviatilis). For intracellular recordings and synaptic transmission studies, the spinal cord was completely isolated. For studies of fictive locomotion, pieces of spinal cord were left attached to the underlying notochord. Preparations were bathed in cooled (8-10°C) lamprey Ringer's solution. For some intracellular recording experiments, Mg²⁺ was omitted from the solution. In experiments involving agonist ejection, drugs (e.g., NMDA, kainate, AMPA) were pressure-ejected from a pipette positioned over the spinal cord. For bath application, drugs like CNQX, DNQX, NMA, or AMPA were added directly to the bathing Ringer's solution at specified concentrations (e.g., CNQX 5-15 µM). Ventral root activity was recorded with suction electrodes to monitor fictive locomotion.[3] 1. Cocaine Self-Administration and CNQX Testing: Male Wistar rats were trained to self-administer intravenous cocaine under a complex second-order schedule [FI15 min(FR7:S)] followed by a period under an FR4(FR7:S) schedule. After stable responding was established at a cocaine dose of 0.50 mg/infusion, the effects of CNQX were tested. CNQX (0, 0.75, 1.5, 3 mg/kg) was dissolved in distilled water and administered intraperitoneally (IP) at a volume of 1 ml/kg, 20 minutes before the start of the self-administration session. Doses were administered in a counterbalanced order (Latin square design) once per week.[4] 2. Locomotor Activity Test: Rats were habituated to test cages after vehicle injections. The effects of CNQX (0, 0.75, 1.5, 3 mg/kg IP) were examined 20 minutes after injection. Locomotor activity (horizontal and vertical movements detected by photocell beams) was recorded for 60 minutes. Testing followed a within-subjects Latin square design with at least two vehicle injection days separating different CNQX dose tests.[4] 3. Surgical Procedure: Rats were implanted with chronic jugular catheters under isoflurane anesthesia for intravenous drug delivery. Catheters were flushed daily with an antibiotic and heparinized saline to maintain patency and prevent infection.[4] |
| References |
[1]. Quinoxalinediones: Potent Competitive non-NMDA Glutamate Receptor Antagonists. Science. 1988 Aug 5;241(4866):701-3. [2]. Blockade of excitatory synaptic transmission by 6-cyano-7-nitroquinoxaline-2,3-dione(CNQX) in the hippocampus in vitro. Neurosci Lett. 1988 Sep 23;92(1):64-8. [3]. CNQX and DNQX block non-NMDA synaptic transmission but not NMDA-evoked locomotion in lamprey spinal cord. Brain Res. 1990 Jan 8;506(2):297-302. [4]. Attenuation of Cocaine-Seeking Behaviour by the AMPA/kainate Receptor Antagonist CNQX in Rats. Psychopharmacology (Berl). 2003 Feb;166(1):69-76. [5]. Infusion of the non-NMDA receptor antagonist CNQX into the amygdala blocks the expression of fear-potentiated startle. Behav Neural Biol. 1993 Jan;59(1):5-8. |
| Additional Infomation |
6-Cyano-7-nitroquinoxaline-2,3-dione is a quinoxaline derivative. A potent excitatory amino acid antagonist with a preference for non-NMDA iontropic receptors. It is used primarily as a research tool. CNQX (6-cyano-7-nitroquinoxaline-2,3-dione) is identified in this study as a potent and selective antagonist for non-NMDA type glutamate receptors (specifically quisqualate/AMPA and kainate receptors), with minimal effect on NMDA receptor-mediated responses. It was used as a pharmacological tool to dissect the contributions of different excitatory amino acid receptor subtypes in synaptic transmission in the mammalian spinal cord. The development of CNQX and DNQX opened possibilities for studying quisqualate and kainate receptor subtypes.[1] This study characterizes CNQX (6-cyano-7-nitroquinoxaline-2,3-dione) as a potent, selective, and reversible antagonist of excitatory synaptic transmission in the hippocampus, which is mediated by non-NMDA glutamate receptors. Its ability to block synaptic EPSPs while sparing GABAergic inhibition and its preferential antagonism of quisqualate-induced currents over NMDA-induced currents suggest it is a useful pharmacological tool for studying excitatory amino acid-mediated synaptic transmission. The findings support the hypothesis that the transmitter for the Schaffer collateral and mossy fiber EPSPs acts primarily on quisqualate-type receptors.[2] This study in the lamprey spinal cord confirms that CNQX is a potent and selective antagonist of synaptic transmission mediated by non-NMDA (kainate and quisqualate/AMPA) excitatory amino acid receptors, consistent with its actions in the mammalian CNS. A key finding is that blockade of non-NMDA receptors with CNQX does not disrupt "fictive locomotion" (the coordinated motor pattern for swimming) induced solely by NMDA receptor activation, indicating that NMDA receptor-mediated synaptic transmission is sufficient to coordinate this rhythmic motor activity. The study also notes that, unlike DNQX at very high concentrations (>10 µM), CNQX (up to 15 µM) did not disrupt NMDA-evoked locomotion, suggesting a cleaner pharmacological profile at the doses used, potentially due to less action at the glycine site of the NMDA receptor complex.[3] 1. CNQX (6-cyano-7-nitroquinoxaline-2,3-dione disodium) is a water-soluble AMPA/kainate receptor antagonist.[4] 2. The study suggests that AMPA/kainate receptors are involved in mediating cocaine-seeking behavior that is controlled, at least partly, by cocaine-associated environmental cues (conditioned stimuli). The effect of systemic CNQX was specific to the cue-controlled "seeking" phase before cocaine intake, not the "taking" phase after cocaine infusion.[4] 3. The article discusses that the neural circuit potentially involved includes glutamatergic projections from the anterior cingulate cortex and basolateral amygdala to the nucleus accumbens core.[4] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~20 mg/mL (~86.15 mM) H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.3076 mL | 21.5378 mL | 43.0756 mL | |
| 5 mM | 0.8615 mL | 4.3076 mL | 8.6151 mL | |
| 10 mM | 0.4308 mL | 2.1538 mL | 4.3076 mL |