Physicochemical Properties
| Molecular Formula | C23H27FN4O3 |
| Molecular Weight | 426.483888864517 |
| Exact Mass | 426.206 |
| Elemental Analysis | C, 64.77; H, 6.38; F, 4.45; N, 13.14; O, 11.25 |
| CAS # | 1350296-29-5 |
| PubChem CID | 58511893 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.8 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 31 |
| Complexity | 642 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | FYRMUNUDXHUNNW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H27FN4O3/c1-16(29)17-4-6-21-22(14-17)31-23(30)28(21)9-3-2-8-26-10-12-27(13-11-26)20-7-5-18(24)15-19(20)25/h4-7,14-15H,2-3,8-13,25H2,1H3 |
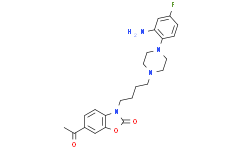
| Chemical Name | 6-acetyl-3-[4-[4-(2-amino-4-fluorophenyl)piperazin-1-yl]butyl]-1,3-benzoxazol-2-one |
| Synonyms | CM 764; CM-764; CM764 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Sigma 2 Receptor (Ki = 3.5 nM); Sigma 1 Receptor (Ki = 86.6 nM) |
| ln Vitro | In this study, researchers report a novel metabolic regulatory function for sigma-2 receptors. CM764 [6-acetyl-3-(4-(4-(2-amino-4-fluorophenyl)piperazin-1-yl)butyl)benzo[d]oxazol-2(3H)-one] binds with Ki values of 86.6 ± 2.8 and 3.5 ± 0.9 nM at the sigma-1 and sigma-2 receptors, respectively. CM764 increased reduction of MTT [3-[4,5 dimethylthiazol-2-yl]-2,5 diphenyltetrazolium bromide] in human SK-N-SH neuroblastoma compared with untreated cells, an effect not due to proliferation. This effect was attenuated by five different sigma antagonists, including CM572 [3-(4-(4-(4-fluorophenyl)piperazin-1-yl)butyl)-6-isothiocyanatobenzo[d]oxazol-2(3H)-one], which has no significant affinity for sigma-1 receptors. This effect was also observed in MG-63 osteosarcoma and HEK293T cells, indicating that this function is not exclusive to neuroblastoma or to cancer cells. CM764 produced an immediate, robust, and transient increase in cytosolic calcium, consistent with sigma-2 receptor activation. Additionally, we observed an increase in the total NAD(+)/NADH level and the ATP level in CM764-treated SK-N-SH cells compared with untreated cells. After only 4 hours of treatment, basal levels of reactive oxygen species were reduced by 90% in cells treated with CM764 over untreated cells, and HIF1α and VEGF levels were increased after 3-24 hours of treatment. These data indicate that sigma-2 receptors may play a role in induction of glycolysis, representing a possible prosurvival function for the sigma-2 receptor that is consistent with its upregulation in cancer cells compared with healthy tissue.[1] |
| Enzyme Assay |
Radioligand Binding Assay. [1] Rat liver membrane homogenates were used for receptor binding assays with slight modification to the previously reported procedure (Hellewell et al., 1994). Frozen rat livers were thawed and homogenized for membrane preparation as previously described (Hellewell et al., 1994). Membrane protein (150 μg) was incubated with 3 nM [3H](+)-pentazocine (sigma-1 receptor condition) or 5 nM [3H]-1,3-di-o-tolylguanidine and 100 nM unlabeled (+)-pentazocine to mask sigma-1 receptors (sigma-2 receptor condition) with various concentrations of CM764 for 120 minutes at 25°C in 20 mM HEPES pH 7.4 with gentle shaking. Nonspecific binding was measured in the presence of 10 μM haloperidol. Membranes were collected by filtration using a Brandel Cell Harvester onto glass fiber filters that were presoaked in 0.5% polyethyleneimine for 30 minutes at room temperature (RT). Ice-cold 10 mM Tris-HCl, pH 7.4 was used to terminate reactions using 5 ml buffer followed by two 5 ml buffer washes. The Ki values were determined by competition binding assay and data analyzed using GraphPad Prism 6 with Kd = 17.9 nM for [3H]-1,3-di-o-tolylguanidine at sigma-2 receptors and Kd = 7.5 nM for [3H](+)-pentazocine at sigma-1 receptors in rat liver membrane homogenates. |
| Cell Assay |
CyQUANT Cell Proliferation Assay.[1] Cellular proliferation was measured using the CyQUANT Cell Proliferation Assay Kit according to manufacturer's specifications. For the experiments cells were plated in 96-well plates at 15,000 cells/well for 24–48 hours or 10,000 cells/well for 72–96 hours. Cells were allowed to attach overnight prior to treatment with 10 µM CM764 or fresh media without ligand (control). Media with or without ligand was replaced every 24 hours. After the indicated time, cells were washed once with phosphate-buffered saline, and then the plate was inverted and blotted dry onto a Kimwipe (Kimberly-Clark Professional, Roswell, GA) by gentle tapping and frozen at −80°C for at least 24 hours. Plates were then thawed to RT and stained with 1X CyQUANT GR dye (which exhibits enhanced fluorescence when bound to nucleic acids) in cell lysis buffer for 5 minutes and protected from light prior to measurement of fluorescence at 480-nm excitation/520-nm emission. NAD+/NADH Assay. [1] NAD+ and NADH were measured using the NAD+/NADH Quantification Colorimetric Kit, which allows for determination of total NAD, NAD+, and NADH from plated cells without the need for purification steps. Cells were plated in 35-mm petri dishes at 200,000 cells/dish and allowed to attach overnight prior to treatment with 10 µM CM764 or fresh media without ligand (control) for 24 hours. The NAD+/NADH assay was then performed according to the manufacturer’s specifications. Cells were washed once with cold phosphate-buffered saline, dissociated with 2.5 mM EDTA, pelleted by centrifugation at 223g for 5 minutes, and extracted with 400-µl extraction buffer by two freeze/thaw cycles of 20 minutes on dry ice and 10 minutes at RT. The extract was then pelleted by centrifugation at 37,500g for 5 minutes and supernatant was collected. For NADH determination, NAD+ was degraded by heating at 60°C for 30 minutes. Samples were incubated with reaction mix for 5 minutes prior to the addition of 10 µl developer, and then allowed to cycle for 4 hours. Absorbance was measured every hour at 450 nm. NADt (NADH + NAD+) and NADH levels were measured directly; the NAD+ level was inferred to be the difference between NADt and NADH. Protein concentration was measured by BCA Assay and NADt/NADH/NAD+ levels were adjusted for protein levels. ATP Assay. [1] Cellular ATP was measured using an ATP Colorimetric/Fluorometric Assay Kit according to manufacturer’s specifications. Cells were plated in 35-mm petri dishes at 600,000 cells/well and allowed to attach overnight prior to treatment with 10 µM CM764 or fresh media without ligand (control). After 24 hours, cells were washed once with phosphate-buffered saline, lysed, and immediately deproteinized with perchloric acid and neutralized with potassium hydroxide. A 50-µl aliquot of deproteinized supernatant was incubated with reaction mix for 30 minutes at RT and protected from light. Fluorescence of the samples was read at 535-nm excitation/587-nm emission. Protein concentration was measured by BCA Assay and ATP levels were adjusted for protein levels. |
| References |
[1]. Sigma-2 Receptors Play a Role in Cellular Metabolism: Stimulation of Glycolytic Hallmarks by CM764 in Human SK-N-SH Neuroblastoma. J Pharmacol Exp Ther. 2016 Feb;356(2):232-43. |
| Additional Infomation |
It has been proposed that the sigma-2 receptor binding site resides within the PGRMC1 protein complex (Xu et al., 2011). However, whether the sigma-2 receptor and PGRMC1 are one in the same molecule remains controversial (Abate et al., 2015; Chu et al., 2015). Furthermore, the physiologic effects of PGRMC1 activation have largely been shown to promote cell survival and inhibit apoptosis, which is in direct contrast to classic proapoptotic models of sigma-2 receptor activation (Lösel et al., 2008; Neubauer et al., 2009; Ahmed et al., 2010; Peluso et al., 2010). Interestingly, activation of PGRMC1 by cell-impermeable progesterone was shown to significantly stimulate VEGF gene expression in MCF-7 cells (Neubauer et al., 2009). The lack of (+)-pentazocine activity and the data shown in Fig. 5 indicate that the pharmacological profile of CM764-induced stimulation of MTT reduction is consistent with mediation by sigma-2 receptors. However, stimulation of VEGF expression by CM764 resembles an effect of PGMRC1 activation (Neubauer et al., 2009). In view of the controversy over the identity of the sigma-2 receptor, there are three possible explanations for the data reported here: 1) the sigma-2 receptor is the binding site of PGRMC1, with bifurcating apoptotic and nonapoptotic pathways being initiated from a single sigma-2/PGRMC1 receptor entity, depending on the specific ligand involved; 2) PGRMC1 and sigma-2 receptors are distinct molecules, but have overlapping pharmacological profiles allowing some sigma-2 ligands to have effects at PGRMC1; or 3) PGRMC1 and sigma-2 receptors are distinct entities with distinct pharmacological profiles, but binding of some compounds to sigma-2 receptors results in complexation with PGRMC1 and activation of nontoxic PGRMC1 signaling events. Distinguishing these possibilities will require further investigation. It should be mentioned here that several other SN79 analogs with high sigma-2 affinity are also able to stimulate MTT reduction (Nicholson, et al.; manuscript in preparation). In deference to the observed pharmacological profile, we have referred to the effects described herein as being sigma-2 receptor mediated.
In conclusion, this study unveils a novel metabolically stimulative, nontoxic sigma-2 receptor function. It is consistent with an evolutionary benefit to upregulation of sigma-2 receptors in cancer cells and rapidly proliferating noncancerous cells, suggesting potential for protection against oxidative damage, hypoxic conditions, and stimulation of angiogenesis via VEGF production.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3448 mL | 11.7239 mL | 23.4478 mL | |
| 5 mM | 0.4690 mL | 2.3448 mL | 4.6896 mL | |
| 10 mM | 0.2345 mL | 1.1724 mL | 2.3448 mL |