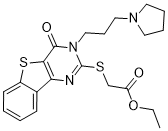
CM037 is a novel, potent and selective aldehyde dehydrogenase 1A1 (ALDH1A1) inhibitor which acts by binding within the aldehyde binding pocket of ALDH1A1 in a competitive mode of inhibition.
Physicochemical Properties
| Molecular Formula | C21H25N3O3S2 |
| Molecular Weight | 431.571502447128 |
| Exact Mass | 431.133 |
| CAS # | 896795-60-1 |
| PubChem CID | 5049161 |
| Appearance | Light brown to brown solid powder |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 29 |
| Complexity | 639 |
| Defined Atom Stereocenter Count | 0 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
CM-037 is a selective inhibitor of human aldehyde dehydrogenase 1A1 (ALDH1A1). It exhibits an IC50 of 4.6 ± 0.8 µM against ALDH1A1 using propionaldehyde as the substrate and a competitive inhibition constant (Kd) of 0.23 ± 0.06 µM against acetaldehyde. [1] |
| ln Vitro |
CM037 was also selective for ALDH1A1 at 20 µM versus eight other ALDH isoenzymes tested. While 20 µM CM037 had little effect on most ALDH isoenzymes tested, ALDH1A3 was inhibited approximately 20% at this concentration. Higher concentrations were not tested due to solubility limits of CM037 under these assay conditions. CM037 exhibits an IC50 = 4.6 ± 0.8 µM toward ALDH1A1 versus the substrate propionaldehyde and a competitive mode of inhibition with respect to varied substrate acetaldehyde and an average Ki of 0.23 ± 0.06 µM from three independent inhibition experiments42.[1] When Gly458 is mutated to asparagine, 20 µM CM037 no longer inhibited the G458N enzyme.[1] CM-037 selectively inhibits the aldehyde dehydrogenase activity of human ALDH1A1. At a concentration of 20 µM, it inhibits ALDH1A1 activity by >80% while showing only minimal inhibition (~20%) against ALDH1A3 and has little to no effect on ALDH1A2, ALDH1B1, ALDH2, ALDH3A1, ALDH4A1, ALDH5A1, and the carboxyl-terminal ALDH domain of rat ALDH1L1. Its selectivity is determined by its binding within the aldehyde binding pocket of ALDH1A1, exploiting the presence of a unique Glycine residue (Gly458). Mutation of Gly458 to Asn (G458N) in ALDH1A1 abolishes inhibition by CM-037 at 20 µM. [1] |
| Enzyme Assay |
The aldehyde dehydrogenase activity assays for characterizing CM-037 were performed spectrophotometrically by monitoring NADH formation at 340 nm. Assays were conducted at 25°C in buffer (e.g., 50 mM sodium BES, pH 7.5). For selectivity screening, reactions contained enzyme (concentration varied by isoform), 200 µM NAD+, 1% DMSO, and a non-saturating concentration of aldehyde substrate (e.g., 100 µM propionaldehyde for ALDH1A1, 1A2, 1A3, 1B1, and ALDH2; 300 µM benzaldehyde for ALDH3A1). Compounds were pre-incubated with enzyme and cofactor for 2 minutes before reaction initiation by aldehyde addition. IC50 values were determined by varying compound concentration (0-200 µM). Steady-state kinetics for inhibition mode determination involved co-varying inhibitor and substrate (acetaldehyde or propionaldehyde) concentrations at a fixed concentration of the second substrate (NAD+). Data were fitted to appropriate inhibition models using analysis software. [1] |
| References |
[1]. Morgan CA, Hurley TD. Characterization of two distinct structural classes of selective aldehyde dehydrogenase 1A1 inhibitors. J Med Chem. 2015 Feb 26;58(4):1964-75. |
| Additional Infomation |
CM-037 (molecular weight 431.6 Da) was identified from a high-throughput screen based on ALDH1A1 esterase activity. X-ray crystallography (resolution 1.85 Å) revealed that CM-037 binds in the aldehyde binding pocket of ALDH1A1, with its long axis oriented almost orthogonally to another inhibitor class (CM026). Binding induces a conformational movement of Trp178 to accommodate its benzyl ring. Its selectivity among ALDH isoenzymes is primarily due to the unique space provided by Gly458 in ALDH1A1; bulkier amino acid side chains at the equivalent position in other isoforms (e.g., Asn in ALDH1A2/1A3/1B1, Asp in ALDH2, Ile in ALDH3A1) sterically hinder binding. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~83.33 mg/mL (~193.09 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3171 mL | 11.5856 mL | 23.1712 mL | |
| 5 mM | 0.4634 mL | 2.3171 mL | 4.6342 mL | |
| 10 mM | 0.2317 mL | 1.1586 mL | 2.3171 mL |