Physicochemical Properties
| Molecular Formula | C₁₁H₁₂CLN₃O₂S |
| Molecular Weight | 285.75 |
| Exact Mass | 285.034 |
| CAS # | 120615-25-0 |
| Related CAS # | CKI-7;1177141-67-1 |
| PubChem CID | 129236 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.432g/cm3 |
| Boiling Point | 499.7ºC at 760mmHg |
| Melting Point | 188-190ºC |
| Flash Point | 256ºC |
| Vapour Pressure | 4.04E-10mmHg at 25°C |
| Index of Refraction | 1.644 |
| LogP | 3.297 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 18 |
| Complexity | 371 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | OGKYMFFYOWUTKV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H12ClN3O2S/c12-10-1-2-11(18(16,17)15-6-4-13)9-7-14-5-3-8(9)10/h1-3,5,7,15H,4,6,13H2 |
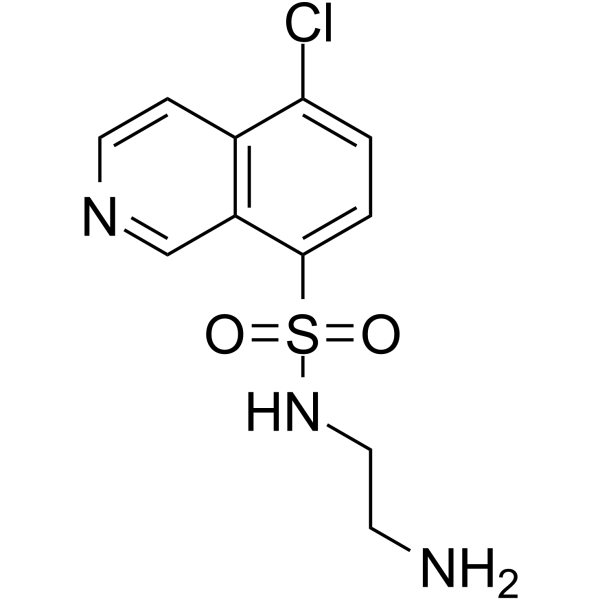
| Chemical Name | N-(2-aminoethyl)-5-chloroisoquinoline-8-sulfonamide |
| Synonyms | CKI7 free base; CKI 7 free base |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In ES cells, CKI-7 treatment (0.1-10 μM; 5 days) dramatically enhanced the number of cells positive for the neural markers βIII-tubulin and nestin, as well as the expression of the early neuroectoderm marker Sox1 in a concentration-dependent manner [1]. Day 5 β-catenin stabilization induced by SFEB was decreased by CKI-7 (5 μM; 5 days; ES cells) treatment, suggesting that CKI-7 suppresses Wnt signaling [1]. |
| ln Vivo | Using a newly isolated Philadelphia chromosome-positive acute lymphoblastic leukemia cell line, the in vivo dose-dependent anticancer efficacy of CKI-7 was established in a SCID-Beige mouse systemic tumor model. The activation of caspase 3 and subsequent cell cycle-dependent death are confirmed by standard cell cycle synchronization tests upon exposure to CKI-7 [2]. |
| Cell Assay |
RT-PCR[1] Cell Types: Mouse ES cells Tested Concentrations: 0.1-10 μM Incubation Duration: 5 days Experimental Results: The expression of early neuroectoderm marker Sox1 was Dramatically increased, and the neural markers nestin and βIII-tubulin were Dramatically increased. The number of positive cells increased Dramatically in a concentration-dependent manner. Western Blot Analysis[1] Cell Types: Mouse ES cells Tested Concentrations: 5 μM Incubation Duration: 5 days Experimental Results: Inhibition of SFEB-induced β-catenin stabilization on day 5. |
| References |
[1]. In vitro differentiation of retinal cells from human pluripotent stem cells by small-molecule induction. J Cell Sci. 2009 Sep 1;122(Pt 17):3169-79. [2]. Small Molecule Inhibition of Cdc7, a Key Cell Cycle Regulator and Novel Therapeutic Target, Successfully Inhibits Leukemia Cell Growth in Vitro and in Vivo. Blood (2008) 112 (11): 2668. [3]. A newly synthesized selective casein kinase I inhibitor, N-(2-aminoethyl)-5-chloroisoquinoline-8-sulfonamide, and affinity purification of casein kinase I from bovine testis. J Biol Chem. 1989 Mar 25;264(9):4924-7. [4]. D4476, a cell-permeant inhibitor of CK1, suppresses the site-specific phosphorylation and nuclear exclusion of FOXO1a. EMBO Rep. 2004 Jan;5(1):60-5. |
| Additional Infomation | N-(2-aminoethyl)-5-chloroisoquinoline-8-sulfonamide is a member of the class of isoquinolines that is isoquinoline-8-sulfonamide which is substituted by chlorine at position 5 and in which the sulfonamide nitrogen is substituted by a 2-aminoethyl group. It is an inhibitor of casein kinase I. It has a role as an EC 2.7.11.1 (non-specific serine/threonine protein kinase) inhibitor. It is a sulfonamide, a member of isoquinolines, an organochlorine compound and a primary amino compound. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~87.49 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.28 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (7.28 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (7.28 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4996 mL | 17.4978 mL | 34.9956 mL | |
| 5 mM | 0.6999 mL | 3.4996 mL | 6.9991 mL | |
| 10 mM | 0.3500 mL | 1.7498 mL | 3.4996 mL |