Physicochemical Properties
| Molecular Formula | C11H10BR2FNO |
| Molecular Weight | 351.01 |
| Exact Mass | 348.911 |
| CAS # | 143703-25-7 |
| Related CAS # | (R)-CE3F4;1593478-56-8 |
| PubChem CID | 21781066 |
| Appearance | White to off-white solid powder |
| Density | 1.8±0.1 g/cm3 |
| Boiling Point | 431.1±45.0 °C at 760 mmHg |
| Flash Point | 214.5±28.7 °C |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.641 |
| LogP | 3.7 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 16 |
| Complexity | 279 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | ZZLQPWXVZCPUGC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H10Br2FNO/c1-6-2-3-7-9(15(6)5-16)4-8(12)11(14)10(7)13/h4-6H,2-3H2,1H3 |
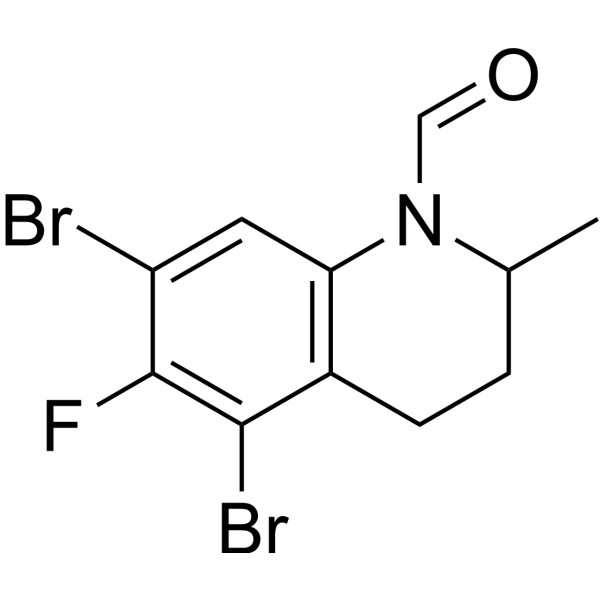
| Chemical Name | 5,7-dibromo-6-fluoro-2-methyl-3,4-dihydro-2H-quinoline-1-carbaldehyde |
| Synonyms | CE 3 F 4; CE-3-F-4; CE3F4 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | IC50s for Epac1 and Epac2(B) are 10.7 μM and 66 μM, respectively, indicating that CE3F4 is a selective antagonist of Epac1. While (S)-CE3F4 (IC50: 56 μM) is less active than (R)-CE3F4 (IC50: 5.8 μM) against Epac1, CE3F4 is more active than that of the latter. When it comes to Epac1's GEF activity, CE3F4 (50 μM) has a greater inhibitory effect than either Epac2(AB) or Epac2(B). An IC50 of 23 ± 3 μM is achieved by CE3F4 in reducing 007-induced Epac1 exchange activity. Without disrupting Rap1 activity or the Epac1-Rap1 connection, CE3F4 (40 μM) selectively inhibits the guanine nucleotide exchange activity of Epac1. On PKA activity, CE3F4 has no effect. Cultured HEK293 cells that have been exposed to Epac are not able to activate Rap1 due to CE3F4 (20 μM). The late phase of glucose-stimulated ERK activation in INS-1 cells is greatly inhibited by CE3F4 (20 μM) [3–4]. |
| ln Vivo | Atrial fibrillation (AF) is inhibited by CE3F4 (1-3 mg/kg; via internal jugular vein catheter), while ventricular arrhythmias are inhibited by CE3F4 (3 mg/kg; intravenously) [4]. After a myocardial infarction, mice's hearts can perform better when given an intravenous infusion of CE3F4 (10 mg/kg) [5]. |
| Animal Protocol |
Animal/Disease Models: Wild-type (WT) mice (AF is induced 20 minutes after CE3F4 administration) [4] Doses: 3 mg/kg and 1mg/kg Route of Administration: via internal jugular vein catheter Experimental Results: Shortened pacing duration - 3mg /kg induces AF. Animal/Disease Models: Casq2-KO mice (isoproterenol is injected 20 minutes after CE3F4 administration to induce premature ventricular contractions) [4] Doses: 3 mg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results:Reduce ventricular contractions caused by sympathetic nerve excitation Incidence of cardiac arrhythmias. |
| References |
[1]. The (R)-enantiomer of CE3F4 is a preferential inhibitor of human exchange protein directly activated by cyclic AMP isoform 1 (Epac1). Biochem Biophys Res Commun. 2013 Oct 25;440(3):443-8. [2]. Identification of a tetrahydroquinoline analog as a pharmacological inhibitor of the cAMP-binding protein Epac. J Biol Chem. 2012 Dec 28;287(53):44192-202. [3]. Ca2+ influx through L-type Ca2+ channels and Ca2+-induced Ca2+ release regulate cAMP accumulation and Epac1-dependent ERK 1/2 activation in INS-1 cells. Mol Cell Endocrinol. 2016 Jan 5;419:60-71. [4]. Usefulness of Exchanged Protein Directly Activated by cAMP (Epac)1-Inhibiting Therapy for Prevention of Atrial and Ventricular Arrhythmias in Mice. Circ J. 2019;83(2):295-303. [5]. Abstract 17548: Inhibition of Exchange Protein 1 Directly Activated by cAMP (Epac1) is Cardioprotective Against Ischemia-reperfusion Injury. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~142.45 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.12 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.12 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8489 mL | 14.2446 mL | 28.4892 mL | |
| 5 mM | 0.5698 mL | 2.8489 mL | 5.6978 mL | |
| 10 mM | 0.2849 mL | 1.4245 mL | 2.8489 mL |