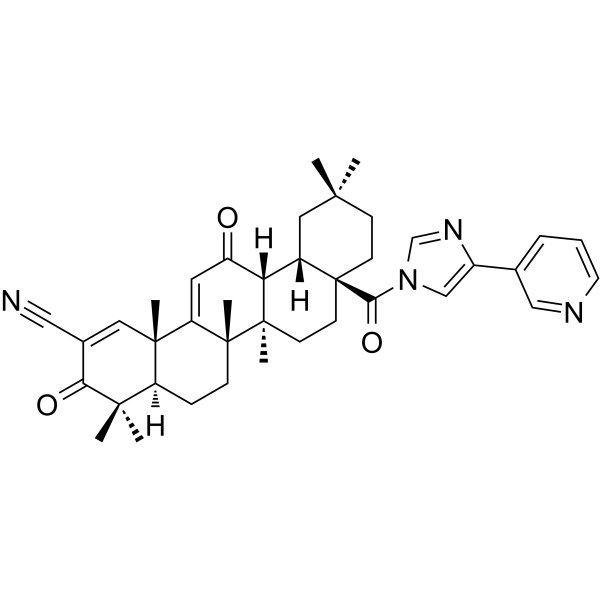
Physicochemical Properties
| Molecular Formula | C39H46N4O3 |
| Molecular Weight | 618.807549953461 |
| Exact Mass | 618.356 |
| CAS # | 1883650-95-0 |
| PubChem CID | 135397766 |
| Appearance | White to off-white solid powder |
| LogP | 7.1 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 46 |
| Complexity | 1480 |
| Defined Atom Stereocenter Count | 7 |
| SMILES | C[C@@]12CC[C@]3(CCC(C[C@H]3[C@H]1C(=O)C=C4[C@]2(CC[C@@H]5[C@@]4(C=C(C(=O)C5(C)C)C#N)C)C)(C)C)C(=O)N6C=C(N=C6)C7=CN=CC=C7 |
| InChi Key | GXKRPLQWYWDFQN-CXUPGZSSSA-N |
| InChi Code | InChI=1S/C39H46N4O3/c1-34(2)12-14-39(33(46)43-22-27(42-23-43)24-9-8-16-41-21-24)15-13-38(7)31(26(39)19-34)28(44)17-30-36(5)18-25(20-40)32(45)35(3,4)29(36)10-11-37(30,38)6/h8-9,16-18,21-23,26,29,31H,10-15,19H2,1-7H3/t26-,29-,31-,36-,37+,38+,39-/m0/s1 |
| Chemical Name | (4aR,6aR,6aS,6bR,8aS,12aS,14bS)-4,4,6a,6b,11,11,14b-heptamethyl-3,13-dioxo-8a-(4-pyridin-3-ylimidazole-1-carbonyl)-4a,5,6,6a,7,8,9,10,12,12a-decahydropicene-2-carbonitrile |
| Synonyms | CDDO3PIm; CDDO 3P Im |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | U937 cells are induced to differentiate at 30 nM by CDDO-3P-Im (30-100 nM; 4 days) [1]. With an IC50 of 4.3 nM, CDDO-3P-Im suppresses the generation of NO in RAW264.7 cells[1]. |
| ln Vivo | CDDO-3P-Im was found to be more stable than CDDO-Im in pharmacokinetic investigations [1]. In a variety of animal tissues in vivo, CDDO-3P-Im dramatically raises the levels of quinone reductase (NQO1) and heme oxygenase-1 (HO-1) mRNA and protein [1]. In A/J mice, CDDO-3P-Im (50–200 mg/kg; diet; 16 weeks) decreases the quantity, size, and severity of tumors [1]. |
| Cell Assay |
Apoptosis analysis [1] Cell Types: U937 Cell Tested Concentrations: 30 nM, 100 nM Incubation Duration: 4 days Experimental Results: 30 nM induced U937 cell differentiation. |
| Animal Protocol |
Animal/Disease Models: Sevenweeks old female A/J mice [1] Doses: 50 mg/kg, 200 mg/kg Route of Administration: diet; continued for 16 weeks Experimental Results: The number, size and severity of tumors were diminished. |
| References |
[1]. Cao M , et al. Novel synthetic pyridyl analogues of CDDO-Imidazolide are useful new tools in cancer prevention. Pharmacol Res. 2015 Oct;100:135-47. [2]. Yuanyuan Wang, et al. Discovery of bardoxolone derivatives as novel orally active necroptosis inhibitors. Eur J Med Chem. 2020 Nov 21;113030. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~404.00 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.36 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6160 mL | 8.0800 mL | 16.1600 mL | |
| 5 mM | 0.3232 mL | 1.6160 mL | 3.2320 mL | |
| 10 mM | 0.1616 mL | 0.8080 mL | 1.6160 mL |