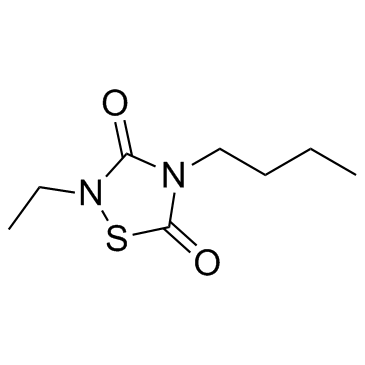
CCG 203769 is a novel, potent and selective G protein signaling (RGS4) inhibitor with an IC50 of 17 nM for blocking the RGS4-Gαo protein-protein interaction in vitro.
Physicochemical Properties
| Molecular Formula | C8H14N2O2S |
| Molecular Weight | 202.274 |
| Exact Mass | 202.077 |
| CAS # | 410074-60-1 |
| PubChem CID | 6539137 |
| Appearance | Colorless to light yellow liquid |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 264.3±23.0 °C at 760 mmHg |
| Flash Point | 113.6±22.6 °C |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.527 |
| LogP | 0.65 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 13 |
| Complexity | 220 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(N1CCCC)N(CC)SC1=O |
| InChi Key | WTFFYZGCISALRI-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H14N2O2S/c1-3-5-6-9-7(11)10(4-2)13-8(9)12/h3-6H2,1-2H3 |
| Chemical Name | 4-butyl-2-ethyl-1,2,4-thiadiazolidine-3,5-dione |
| Synonyms | CCG-203769; CCG203769; CCG 203769 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Moreover, CCG 203769 shows notable selectivity for RGS4 relative to other RGS proteins (8 to >5000-fold). With an IC50 of 140 nM (eight times more selective for RGS4), CCG 203769 inhibits RGS19, and at 6 μM (three hundred fifty times more selective for RGS4), it inhibits RGS16. There was very little inhibition of the similarly related RGS8 (IC50>60 μM), giving RGS4 a selectivity >4500-fold. GSK-3β is inhibited by CCG 203769, with an IC50 value of 5 μM. At 100 μM, CCG 203769 does not inhibit papain, a cysteine protease. Since cysteine is absent from the RGS domain of RGS7, CCG 203769 does not inhibit RGS7. RGS-selective inhibition of RGS/Gαo binding is achieved by CCG 203769. In an RGS4-dependent manner, CCG 203769 improves cellular Ca2+ signaling that is dependent on Gαq. Additionally, CCG 203769 inhibits RGS4's GTPase Accelerating Protein (GAP) function. RGS4 significantly increased the rate of GTP hydrolysis in single-turnover and steady-state GTPase tests utilizing Gαo and Gαi1, and this effect was blocked by CCG 203769 with IC50<1 μM [1]. |
| ln Vivo | In order to ascertain whether the pharmacological replication of this genetic disruption of RGS4 function was possible, we examined the impact of CCG 203769 on bradycardia mediated by carbamylcholine chloride in rats that were awake and unrestrained. In contrast to saline vehicle controls, carbamylcholine chloride (0.1 mg/kg, IP) moderately lowered heart rate. When given alone, CCG 203769 (10 mg/kg IV) had no discernible effect on heart rate. CCG 203769, on the other hand, greatly increased the bradycardia effect when it was given right before carbamylcholine chloride (p < 0.05). In a pharmacological model of D2 antagonist-induced bradykinesia, CCG 203769 was tested in light of RGS4's functional role in Parkinson's disease models. In the rod test, rats given raclopride had longer suspension times; however, CCG 203769, administered at doses ranging from 0.1 to 10 mg/kg, quickly corrected this effect. A submaximal effect was produced by 0.1 mg/kg, while the lowest dose of 0.01 mg/kg had no effect at all. Increased dosages of 1 mg/kg and 10 mg/kg have the same result. Similarly, mice's paw drag caused by raclopride is reversed by 0.1–10 mg/kg CCG 203769 [1]. |
| References |
[1]. Selectivity and anti-Parkinson's potential of thiadiazolidinone RGS4 inhibitors. ACS Chem Neurosci. 2015 Jun 17;6(6):911-9. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~62.5 mg/mL (~308.99 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (10.28 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (10.28 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (10.28 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.9439 mL | 24.7194 mL | 49.4389 mL | |
| 5 mM | 0.9888 mL | 4.9439 mL | 9.8878 mL | |
| 10 mM | 0.4944 mL | 2.4719 mL | 4.9439 mL |