Physicochemical Properties
| Molecular Formula | C14H9N3O |
| Molecular Weight | 235.240762472153 |
| Exact Mass | 235.074 |
| CAS # | 2100864-57-9 |
| PubChem CID | 129216797 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 2.5 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 18 |
| Complexity | 350 |
| Defined Atom Stereocenter Count | 0 |
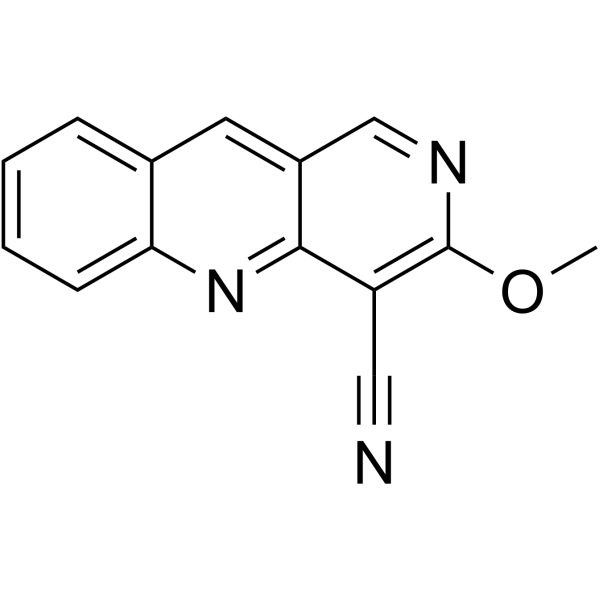
| SMILES | O(C)C1=C(C#N)C2C(C=N1)=CC1C=CC=CC=1N=2 |
| InChi Key | QNJYUHRGCPRPQS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H9N3O/c1-18-14-11(7-15)13-10(8-16-14)6-9-4-2-3-5-12(9)17-13/h2-6,8H,1H3 |
| Chemical Name | 3-methoxybenzo[b][1,6]naphthyridine-4-carbonitrile |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | CCB02 inhibits the CPAP PN2-3-tubulin interaction in the PN2-3 CPAP-GST pull-down test, with an IC50 of 0.441 μM [1]. The phosphorylation status of Aurora A, Plk1, Plk2, CDK2, and CHK1 as well as centrosome-associated kinases are not inhibited by CCB02 [1]. Cancer cells with additional centrosomes are inhibited from proliferating by CCB02 (0.1-15 μM, 72 hours) with an IC50 of 0.86-2.9 μM [1]. CCB02 stimulates the recruitment of PCM proteins to the centrosome, activates the spindle assembly checkpoint, and increases the centrosome's microtubule nucleation activity [1]. |
| ln Vivo | CCB02 (30 mg/kg orally daily for 24 days) demonstrated strong anticancer effects in tumor xenografts of the human lung (H1975T790M cells) subcutaneously implanted in nude mice [1]. In mouse xenografts, CCB02 also results in multipolar mitosis and inhibits MDA-MB-231 cell migration [1]. |
| Cell Assay |
Cell Viability Test[1] Cell Types: BT549, MDA-MB-231, Pop10, SCC13, SW1271p53/pRb/CDKN2Adel, KYSE30p53/MYC/CyclinD1, A549G12S, PC-9EGFR-Exon19del, HCC827-GR, HCC1833-GR, H1975T790M Cell Tested Concentrations: 0.1-15 μM Incubation Duration: 72 hrs (hours) Experimental Results: IC50 is 0.86 μM (Pop10), 1.2 μM (HCC827-GR), 1.5 μM (H1975T790Mp53/MYC/CyclinD1), 1.15 μM (HCC1833-GR), 1.61 μM (SW1271p53/pRb/CDKN2Adel), 2.41 μM (SCC13) and 2.94 μM (PC-9EGFR-Exon19del). |
| Animal Protocol |
Animal/Disease Models: Nude mice carrying subcutaneoushuman lung (H1975T790M) tumor xenografts [1] Doses: 30 mg/kg Route of Administration: daily oral administration for 24 days Experimental Results: Tumor volume was Dramatically diminished on day 24. |
| References |
[1]. Inhibition of CPAP-tubulin interaction prevents proliferation of centrosome-amplified cancer cells. EMBO J. 2019 Jan 15;38(2). pii: e99876. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~106.27 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (10.63 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (10.63 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.2510 mL | 21.2549 mL | 42.5098 mL | |
| 5 mM | 0.8502 mL | 4.2510 mL | 8.5020 mL | |
| 10 mM | 0.4251 mL | 2.1255 mL | 4.2510 mL |