Physicochemical Properties
| Molecular Formula | C26H28N4O2 |
| Molecular Weight | 428.5261 |
| Exact Mass | 428.221 |
| Elemental Analysis | C, 72.87; H, 6.59; N, 13.07; O, 7.47 |
| CAS # | 1130067-34-3 |
| PubChem CID | 25105715 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 756.6±60.0 °C at 760 mmHg |
| Flash Point | 411.4±32.9 °C |
| Vapour Pressure | 0.0±2.5 mmHg at 25°C |
| Index of Refraction | 1.691 |
| LogP | 1.63 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 32 |
| Complexity | 669 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C1C2(C([H])([H])C([H])([H])N(C([H])([H])C([H])([H])N([H])C(C3C([H])=C([H])C4=C([H])C([H])=C([H])C([H])=C4C=3[H])=O)C([H])([H])C2([H])[H])N(C2C([H])=C([H])C([H])=C([H])C=2[H])C([H])([H])N1[H] |
| InChi Key | FAIFAFUXFFWVNQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H28N4O2/c31-24(22-11-10-20-6-4-5-7-21(20)18-22)27-14-17-29-15-12-26(13-16-29)25(32)28-19-30(26)23-8-2-1-3-9-23/h1-11,18H,12-17,19H2,(H,27,31)(H,28,32) |
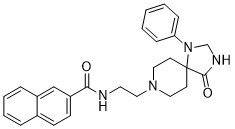
| Chemical Name | N-[2-(4-oxo-1-phenyl-1,3,8-triazaspiro[4,5]dec-8-yl)ethyl]-2-naphthalenecarboxamide |
| Synonyms | CAY10594 CAY-10594 CAY 10594 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
CAY10594 targets phospholipase D1 (PLD1, IC50 = 3.7 μM) and phospholipase D2 (PLD2, IC50 = 0.16 μM), with high selectivity for PLD2 [1] CAY10594 is identified as a specific phospholipase D2 (PLD2) inhibitor [2] |
| ln Vitro |
In MDA-MB-231 breast cancer cells, CAY10594 inhibited cell invasiveness by approximately 50% at 1 μM and 80% at 5 μM, without significant effect on cell proliferation at concentrations up to 10 μM [1] - CAY10594 selectively suppressed PLD2 enzyme activity, with minimal inhibition of PLD1 even at high concentrations [1] - In HepG2 human hepatoma cells, CAY10594 (10 μM) attenuated acetaminophen-induced cytotoxicity, increasing cell viability from ~40% (acetaminophen alone) to ~75% [2] - Western blot analysis showed that CAY10594 (10 μM, 24 h) reduced acetaminophen-induced phosphorylation of JNK (p-JNK) and GSK-3β (Ser9 site, p-GSK-3β), while upregulating total GSK-3β protein expression and inhibiting caspase-3 activation [2] |
| ln Vivo |
We examined the role of phospholipase D2 (PLD2) on acetaminophen (APAP)-induced acute liver injury using a PLD2 inhibitor (CAY10594). 500 mg/kg of APAP challenge caused acute liver damage. CAY10594 administration markedly blocked the acute liver injury in a dose-dependent manner, showing almost complete inhibition with 8 mg/kg of CAY10594. During the pathological progress of acute liver injury, GSH levels are decreased, and this is significantly recovered upon the administration of CAY10594 at 6 hours post APAP challenge. GSK-3β (Serine 9)/JNK phosphorylation is mainly involved in APAPinduced liver injury. CAY10594 administration strongly blocked GSK-3β (Serine 9)/JNK phosphorylation in the APAP-induced acute liver injury model. Consistently, sustained JNK activation in the cytosol and
mitochondria from hepatocytes were also decreased in CAY10594-treated mice. Many types of immune
cells are also implicated in APAP-induced liver injury. However, neutrophil and monocyte populations were not different between vehicle- and CAY10594-administered mice which are challenged with APAP. Therapeutic administration of CAY10594 also significantly attenuated liver damage caused by the APAP challenge, eliciting an enhanced survival rate. Taken together, these results indicate that PLD2 is involved in the intrinsic response pathway of hepatocytes driving the pathogenesis of APAP-induced acute liver injury, and PLD2 may therefore represent an important therapeutic target for patients with
drug-induced liver injury.[2] In acetaminophen-induced acute liver injury (ALI) model in C57BL/6 mice, intraperitoneal administration of CAY10594 (10 mg/kg, twice: 1 h before and 6 h after acetaminophen challenge) significantly reduced serum alanine transaminase (ALT) levels from ~2500 U/L (model group) to ~800 U/L and aspartate transaminase (AST) levels from ~1800 U/L to ~600 U/L [2] - Histopathological examination of liver tissues revealed that CAY10594 treatment reduced the area of hepatic necrosis and infiltration of inflammatory cells compared to the model group [2] - In liver tissues, CAY10594 decreased the phosphorylation levels of JNK and GSK-3β (Ser9), restored GSK-3β activity, and inhibited acetaminophen-induced caspase-3 activation [2] |
| Enzyme Assay |
PLD enzyme activity assay was performed using a radiolabeled substrate method. The reaction mixture contained recombinant PLD1/PLD2, [3H]-phosphatidylcholine as the substrate, buffer, and serial dilutions of CAY10594. After incubation at 37°C for 30 minutes, the radiolabeled product (phosphatidic acid) was extracted, and radioactivity was measured by liquid scintillation counting. IC50 values were calculated by fitting the dose-response curves [1] |
| Cell Assay |
Cancer cell invasion assay: MDA-MB-231 cells (5×104 cells/chamber) were seeded into the upper chamber of Transwell inserts. CAY10594 at different concentrations was added to the serum-free medium in the upper chamber, and medium containing 10% fetal bovine serum was added to the lower chamber as a chemoattractant. After incubation at 37°C with 5% CO2 for 24 hours, non-invading cells in the upper chamber were removed. Invading cells were fixed, stained, and counted to calculate the invasion inhibition rate [1] - Hepatocyte cytotoxicity assay: HepG2 cells were seeded in 96-well plates at 1×105 cells/well and incubated overnight. Cells were pretreated with serial dilutions of CAY10594 for 1 hour, followed by incubation with acetaminophen (20 mM) for 24 hours. Cell viability was determined using a tetrazolium salt-based colorimetric assay [2] - Western blot assay: HepG2 cells treated with CAY10594 and acetaminophen were lysed to extract total proteins. Proteins were separated by SDS-PAGE, transferred to PVDF membranes, and probed with primary antibodies against p-JNK, JNK, p-GSK-3β (Ser9), GSK-3β, caspase-3, and β-actin. After incubation with HRP-conjugated secondary antibodies, chemiluminescent signals were detected [2] |
| Animal Protocol |
Mice were fasted for 16 hours
before APAP injection. APAP (500 mg/kg) was administered with oral gavage in mice. CAY10594 (N-[2-(4-oxo1-phenyl-1,3,8-triazaspiro[4,5]dec-8-yl)ethyl]-2-naphthalene carboxamide)23 was dissolved in 1% DMSO and intraperitoneally administered to mice 30 minutes prior to APAP injection for examining protective effects or after 3 hours from APAP challenge for investigating therapeutic effects of CAY10594.[2] Acetaminophen-induced acute liver injury model: Male C57BL/6 mice (6-8 weeks old) were randomly divided into three groups (n=6 per group): control group, acetaminophen model group, and CAY10594 treatment group. CAY10594 was dissolved in a mixture of DMSO and normal saline (final DMSO concentration ≤5%) and administered via intraperitoneal injection at a dose of 10 mg/kg. Two doses were given: 1 hour before and 6 hours after acetaminophen challenge (300 mg/kg, intraperitoneal injection). At 24 hours after acetaminophen induction, mouse serum was collected to measure ALT and AST levels. Liver tissues were harvested: part was used for pathological sectioning (HE staining), and part was used for protein extraction for western blot analysis [2] |
| References |
[1]. Design of isoform-selective phospholipase D inhibitors that modulate cancer cell invasiveness. [2]. A phospholipase D2 inhibitor, CAY10594, ameliorates acetaminophen-induced acute liver injury by regulating the phosphorylated-GSK-3β/JNK axis. Sci Rep. 2019 May 10;9(1):7242. |
| Additional Infomation |
N-[2-(4-oxo-1-phenyl-1,3,8-triazaspiro[4.5]decan-8-yl)ethyl]-2-naphthalenecarboxamide is a naphthalenecarboxamide. CAY10594 is an isoform-selective PLD inhibitor with higher inhibitory activity against PLD2 than PLD1. It reduces the invasiveness of breast cancer cells (e.g., MDA-MB-231) by inhibiting PLD-mediated signaling pathways, representing a potential candidate for treating cancer metastasis [1] - CAY10594 alleviates acetaminophen-induced acute liver injury by selectively inhibiting PLD2 and regulating the phosphorylated-GSK-3β/JNK signaling axis. Its mechanism involves suppressing hepatocyte apoptosis and inflammatory responses, providing a new therapeutic option for acute liver injury [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~11.36 mg/mL (~26.51 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.14 mg/mL (2.66 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 11.4 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.14 mg/mL (2.66 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 11.4 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3336 mL | 11.6678 mL | 23.3356 mL | |
| 5 mM | 0.4667 mL | 2.3336 mL | 4.6671 mL | |
| 10 mM | 0.2334 mL | 1.1668 mL | 2.3336 mL |