Physicochemical Properties
| Molecular Formula | CH3*Y |
| Molecular Weight | 103.94042 |
| Exact Mass | 328.105 |
| Elemental Analysis | C, 62.19; H, 4.91; N, 8.53; O, 24.36 |
| CAS # | 78028-01-0 |
| PubChem CID | 2601 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 578.0±50.0 °C at 760 mmHg |
| Flash Point | 303.4±30.1 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.599 |
| LogP | 3.41 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 24 |
| Complexity | 460 |
| Defined Atom Stereocenter Count | 0 |
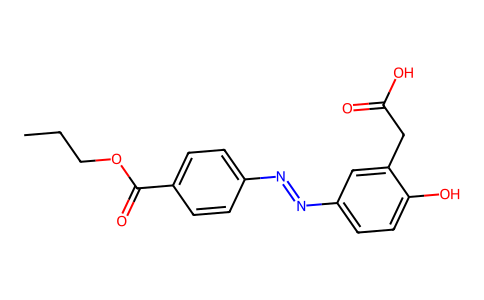
| SMILES | CCCOC(=O)C1=CC=C(C=C1)N=NC2=CC=C(C(=C2)CC(=O)O)O |
| InChi Key | YYBSDOZYJSGLTH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H16N2O5/c1-2-24-17(23)11-3-5-13(6-4-11)18-19-14-7-8-15(20)12(9-14)10-16(21)22/h3-9,20H,2,10H2,1H3,(H,21,22) |
| Chemical Name | 2-[5-[(4-ethoxycarbonylphenyl)diazenyl]-2-hydroxyphenyl]acetic acid |
| Synonyms | 78028-01-0; 5-[2-[4-(Ethoxycarbonyl)phenyl]diazenyl]-2-hydroxybenzeneacetic acid; 5-(2-(4-(Ethoxycarbonyl)phenyl)diazenyl)-2-hydroxybenzeneacetic acid; cay10397; CK47A; 2-[3-[(4-ethoxycarbonylphenyl)hydrazinylidene]-6-oxo-1-cyclohexa-1,4-dienyl]acetic Acid; 2-[5-[(4-ethoxycarbonylphenyl)diazenyl]-2-hydroxyphenyl]acetic acid; SCHEMBL10834750; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 15-hydroxyprostaglandin dehydrogenase |
| ln Vitro |
Inhibition of 15-PGDH by CAY10397 in LoVo and HCA-7 Cell Lysates [1] In contrast to LoVo cells, the HCA-7 cell line is known to express COX-2 and only trace amounts of COX-1 and 15-PGDH. This was confirmed by Western blot analysis (data not shown). LoVo (Figure 6a) or HCA-7 (Figure 6b) cell lysates were incubated with 500 mM NAD+, with or without the 15-PGDH inhibitor, CAY10397 (50 μM), for 10 min. Inhibition of 15-PGDH significantly abolished the formation of endogenous 11-oxo-ETE (Figure 6a) by 92%, along with the diminished formation of 15-oxo-ETE as well as 13,14-dihydro-15-oxo-PGE2 (Figure 6a) in the LoVo cell lysate. However, since the oxo-ETEs and 13,14-dihydro-15-oxo-PGE2 were undetectable in HCA-7 cell lysate, their precursors were quantified instead, and CAY10397 had no effect on their levels (Figure 6b). Inhibition of 15-PGDH enzyme by CAY10397 significantly diminished the formation of endogenous 11-oxo-ETE in LoVo cell lysates (Figure 6a). In addition, the formation of 15-oxo-ETE as well as 13,14-dihydro-15-oxo-PGE2, the other two 15-PGDH-dependent metabolites, was also significantly reduced in LoVo cell lysates treated with CAY10397 (Figure 6a). In contrast, the three 15-PGDH products, namely, 11- and 15-oxo-ETE and 13,14-dihydro-15-oxo-PGE2, were undetectable in HCA-7 cells as these cells do not express any 15-PGDH.10,28 Instead, the corresponding precursors for these three metabolites were observed in the lysates. Furthermore, treatment with CAY10397 had no effect on levels of 11(R)-HETE, 15(S)-HETE or PGE2 (Figure 6b). |
| Cell Assay |
15-PGDH Inhibition in LoVo or HCA-7 Cell Lysates by CAY10397 [1] LoVo or HCA-7 cells were grown to 90% confluence, washed with 10 mL of phosphate-buffered saline (PBS) buffer (2 times), and then gently scraped in 600 μL of lysis buffer containing 0.1 M Tris-HCl (pH 7.9) and the protease inhibitor. Cell suspension was transferred to 2 mL Eppendorf tubes and sonicated for 60 s on ice (power 5). Cell lysate was then incubated with or without the selective 15-PGDH inhibitor (CAY10397, 50 μM) and its cofactor (NAD+, 500 μM) for 10 min at 37 °C. The pH was then adjusted to 4 with 10% aqueous acetic acid (10 μL) followed by addition of the internal standard mix, [13C20]-15-oxo-ETE, [2H8]-15(S)-HETE and [2H4]-PGE2 (50 pg/μL, 20 μL). Diethyl ether (600 μL) was added, and samples were vortex-mixed and centrifuged (15000 rpm × 2 min). The organic layer was evaporated under nitrogen, and then the eicosanoids were derivatized with PFB bromide as mentioned above. Finally, samples were redissolved in hexane/ethanol (95:5; v/v, 100 μL) and analyzed (20 μL) by normal phase LC-ECAPCI/MS. The amounts of eicosanoids were normalized by protein concentrations of each lysate, which were determined by BCA assay. |
| References | [1]. 11-Oxoeicosatetraenoic acid is a cyclooxygenase-2/15-hydroxyprostaglandin dehydrogenase-derived antiproliferative eicosanoid. Chem Res Toxicol. 2011 Dec 19;24(12):2227-36. |
| Additional Infomation |
CAY10397 is a member of the class of azobenzenes that is azobenzene in which one of the phenyl groups is substituted at position 4 by an ethoxycarbonyl group, while the other is substituted by a carboxymethyl group at position 3 and a hydroxy group at position 4. It is a potent and selective inhibitor of 15-hydroxyprostaglandin dehydrogenase (15-hydroxy-PGDH). It has a role as an EC 1.1.1.141 [15-hydroxyprostaglandin dehydrogenase (NAD(+))] inhibitor. It is a dicarboxylic acid monoester, an ethyl ester, a member of azobenzenes and a member of phenols. Previously, we established that 11(R)-hydroxy-5,8,12,14-(Z,Z,E,Z)-eicosatetraenoic acid (HETE) was a significant cyclooxygenase (COX)-2-derived arachidonic acid (AA) metabolite in epithelial cells. Stable isotope dilution chiral liquid chromatography (LC)-electron capture atmospheric pressure chemical ionization (ECAPCI)/mass spectrometry (MS) was used to quantify COX-2-derived eicosanoids in the human colorectal adenocarcinoma (LoVo) epithelial cell line, which expresses both COX-2 and 15-hydroxyprostaglandin dehydrogenase (15-PGDH). 11(R)-HETE secretion reached peak concentrations within minutes after AA addition before rapidly diminishing, suggesting further metabolism had occurred. Surprisingly, recombinant 15-PGDH, which is normally specific for oxidation of eicosanoid 15(S)-hydroxyl groups, was found to convert 11(R)-HETE to 11-oxo-5,8,12,14-(Z,Z,E,Z)-eicosatetraenoic acid (ETE). Furthermore, LoVo cell lysates converted 11(R)-HETE to 11-oxo-ETE and inhibition of 15-PGDH with 5-[[4-(ethoxycarbonyl)phenyl]azo]-2-hydroxy-benzeneacetic acid (CAY10397) (50 μM) significantly suppressed endogenous 11-oxo-ETE production with a corresponding increase in 11(R)-HETE. These data confirmed COX-2 and 15-PGDH as enzymes responsible for 11-oxo-ETE biosynthesis. Finally, addition of AA to the LoVo cells resulted in rapid secretion of 11-oxo-ETE into the media, reaching peak levels within 20 min of starting the incubation. This was followed by a sharp decrease in 11-oxo-ETE levels. Glutathione (GSH) S-transferase (GST) was found to metabolize 11-oxo-ETE to the 11-oxo-ETE-GSH (OEG)-adduct in LoVo cells, as confirmed by LC–MS/MS analysis. Bromodeoxyuridine (BrdU)-based cell proliferation assays in human umbilical vein endothelial cells (HUVECs) revealed that the half-maximal inhibitory concentration (IC50) of 11-oxo-ETE for inhibition of HUVEC proliferation was 2.1 μM. These results show that 11-oxo-ETE is a novel COX-2/15-PGDH-derived eicosanoid, which inhibits endothelial cell proliferation with a potency that is similar to that observed for 15d-PGJ2. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 9.6209 mL | 48.1047 mL | 96.2094 mL | |
| 5 mM | 1.9242 mL | 9.6209 mL | 19.2419 mL | |
| 10 mM | 0.9621 mL | 4.8105 mL | 9.6209 mL |