Physicochemical Properties
| Molecular Formula | C24H22N2O |
| Molecular Weight | 354.444 |
| Exact Mass | 354.173 |
| CAS # | 883561-04-4 |
| PubChem CID | 11610059 |
| Appearance | White to off-white solid powder |
| LogP | 5.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 27 |
| Complexity | 479 |
| Defined Atom Stereocenter Count | 0 |
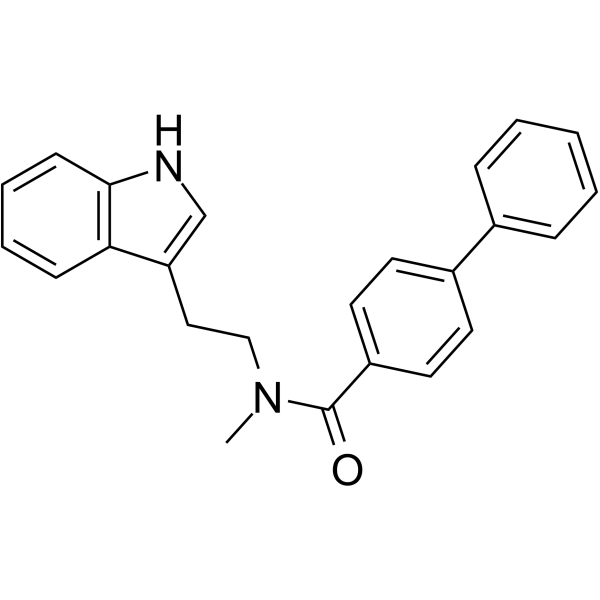
| SMILES | N1C2=C(C=CC=C2)C(CCN(C)C(C2=CC=C(C3=CC=CC=C3)C=C2)=O)=C1 |
| InChi Key | CPKLVRIYXBROSG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H22N2O/c1-26(16-15-21-17-25-23-10-6-5-9-22(21)23)24(27)20-13-11-19(12-14-20)18-7-3-2-4-8-18/h2-14,17,25H,15-16H2,1H3 |
| Chemical Name | N-[2-(1H-indol-3-yl)ethyl]-N-methyl-4-phenylbenzamide |
| Synonyms | CA-224; CA 224; CA224 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | CA224 (Compound 1) (48 hours) treatment against human cancer cell lines demonstrated antiproliferative efficacy [1]. CA224 (18–48 hours) selectively destroys SV40 large T antigen-transformed normal mouse embryonic liver cells (BNL S A.8) and inhibits the growth of cancer cells in the G0/G1 and G2/M phases of the cell cycle [1][2]. Tubulin polymerization is inhibited and stable tubulin depolymerization is enhanced by CA224 (0–4 µM, 30 min) [1]. Cancer cells undergo apoptosis when exposed to CA224 (0-72 h) [1]. Inhibiting CYP3A4, CYP2D6, CYP2C9, and CYP2C19 by 50%, 14%, 51%, and 19%, respectively, is the effect of CA224 (10 μM) [1]. |
| ln Vivo | Significant tumor growth inhibition was demonstrated by CA224 (Compound 1) (100 mg/kg; intraperitoneal injection; once daily for 9 days) without overt toxicity [1]. |
| Cell Assay |
Cell Proliferation Assay[1] Cell Types: LS174T, PC-3, MiaPaCa, A549, Calu-1, NCI-H460, NCI-H1299, NCI-H358, BNL CL2 and BNL SV A.8 Tested Concentrations: Incubation Duration: 48 hrs (hours) Experimental Results: demonstrated anti-proliferative activity against LS174T, PC-3, MiaPaCa, A549, Calu-1, NCI-H460, NCI- with IC50 values of 3.5, 6.2, 4.0, 3.5, 11.5, 2.0, 2.5, 2.2, 2.6 and 3.8 uM are H1299, NCI-H358, BNL CL2 and BNL SV A.8 respectively. Cell cycle analysis[1][2] Cell Types: A549, NCI-H1299, NCI-H358, BNL CL2, BNL SV A.8 and Calu-1 Tested Concentrations: IC50 concentration (IC70 for Calu-1) Incubation Duration: 24 hrs (hours) 18 hrs (hours) for NCI-H358 and 48 hrs (hours) for BNL CL2 and BNL SV for A549, NCI-H1299 and Calu-1 A.8 Experimental Results: Deep blockade was induced at G2/M in A549 and NCI-H1299 cells. Maintenance of nocodazole- and paclitaxel-induced G2/M arrest in NCI-H358 cells. BNL CL2 cells exhibit significant G2/M phase arrest. In BNL S A.8 cells 31% of cells were detected in sub-G1 phase (control: 0%). G0/G1 blockade is preserved in serum- |
| Animal Protocol |
Animal/Disease Models: Severe combined immunodeficiency (SCID) mice, which lack T and B immune cells. Male mice weighing 18~25 g and 6~8 weeks old were injected subcutaneously (sc) (sc) with HCT-116, and female mice weighing 15~24 g and 6~8 weeks old were injected subcutaneously (sc) (sc) with NCI-H460[1]. Doses: 100 mg/kg Route of Administration: intraperitoneal (ip) injection, one time/day for 9 days. Experimental Results: It demonstrated significant tumor growth inhibition in both HCT-116 and NCI-H460 tumor models, and there was no significant decrease in body weight. Animal/Disease Models: balb/c (Bagg ALBino) mouse [1] Doses: 10 mg/kg (oral) or 1.0 mg/kg (iv) Route of Administration: Oral or intravenous (iv) (iv)(pharmacokinetic/PK/PK analysis) Experimental Results: After IV and PO Determination of administrative pharmacokinetic/PK/PK parameters of CA224 [1]. Parameter IV (1 mg/kg) Oral (10 mg/kg) t1/2,β (h) 0.33 1.16 AUC0-t (ng·h/mL) 187 172 AUC0-∞ (ng·h/mL) 189 182 Cmax (ng/mL) 371 190 Vd (L/Kg) 2.52 nd Vdss (L/Kg) 1.76 nd CL (mL/min/kg) 88.3 nd Bioavailability - 9.6% t1/2,β Time point 0.5 consid |
| References |
[1]. Biphenyl-4-carboxylic acid [2-(1H-indol-3-yl)-ethyl]-methylamide (CA224), a nonplanar analogue of fascaplysin, inhibits Cdk4 and tubulin polymerization: evaluation of in vitro and in vivo anticancer activity. J Med Chem. 2014 Nov 26;. [2]. CA224, a non-planar analogue of fascaplysin, inhibits Cdk4 but not Cdk2 and arrests cells at G0/G1 inhibiting pRB phosphorylation. Bioorg Med Chem Lett. 2006 Aug 15;16(16):4272-8. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~282.14 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.05 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8214 mL | 14.1068 mL | 28.2135 mL | |
| 5 mM | 0.5643 mL | 2.8214 mL | 5.6427 mL | |
| 10 mM | 0.2821 mL | 1.4107 mL | 2.8214 mL |