Physicochemical Properties
| Molecular Formula | C35H43N7O2 |
| Molecular Weight | 593.7616 |
| Exact Mass | 593.347 |
| Elemental Analysis | C, 70.80; H, 7.30; N, 16.51; O, 5.39 |
| CAS # | 351351-75-2 |
| Related CAS # | Braco-19 trihydrochloride;1177798-88-7 |
| PubChem CID | 9808666 |
| Appearance | Brown to orange solid powder |
| Density | 1.275g/cm3 |
| Boiling Point | 854.95ºC at 760 mmHg |
| Flash Point | 470.857ºC |
| Index of Refraction | 1.708 |
| LogP | 4.5 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 44 |
| Complexity | 870 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(C([H])([H])C([H])([H])N1C([H])([H])C([H])([H])C([H])([H])C1([H])[H])N([H])C1C([H])=C([H])C2C(C=1[H])=NC1C([H])=C(C([H])=C([H])C=1C=2N([H])C1C([H])=C([H])C(=C([H])C=1[H])N(C([H])([H])[H])C([H])([H])[H])N([H])C(C([H])([H])C([H])([H])N1C([H])([H])C([H])([H])C([H])([H])C1([H])[H])=O |
| InChi Key | RKPYSYRMIXRZJT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C35H43N7O2/c1-40(2)28-11-7-25(8-12-28)38-35-29-13-9-26(36-33(43)15-21-41-17-3-4-18-41)23-31(29)39-32-24-27(10-14-30(32)35)37-34(44)16-22-42-19-5-6-20-42/h7-14,23-24H,3-6,15-22H2,1-2H3,(H,36,43)(H,37,44)(H,38,39) |
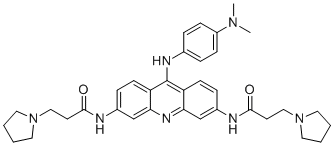
| Chemical Name | N-[9-[4-(dimethylamino)anilino]-6-(3-pyrrolidin-1-ylpropanoylamino)acridin-3-yl]-3-pyrrolidin-1-ylpropanamide |
| Synonyms | BRACO-19; 351351-75-2; BRACO19; Z7C5CD91WI; 1-Pyrrolidinepropanamide, N,N'-(9-((4-(dimethylamino)phenyl)amino)-3,6-acridinediyl)bis-; 9-[4-(N,N-dimethylamino)phenylamino]-3,6-bis(3-pyrrolodinopropionamido) acridine; N,N'-(9-((4-(Dimethylamino)phenyl)amino)-3,6-acridinediyl)bis(1-pyrrolidinepropanamide); 9-[4-(N,N-dimethylamino)phenylamino]-3,6-bis (3-pyrrolodino-propionamido) acridine; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Human telomeric G-quadruplex [1]; telomerase/telomere; BRACO-19 selectively binds to conserved G-quadruplex (GQ) motifs in the Human Adenovirus (HAdV) genome, specifically PGQ-AV-C3 (Kd = 0.28 × 10⁻⁶ M), PGQ-AV-D2 (Kd = 0.34 × 10⁻⁶ M), and PGQ-AV-E3 (Kd = 0.28 × 10⁻⁶ M) [2] |
| ln Vitro |
Well-known GQ-binding ligand Braco-19 selectively binds with HAdV GQ, enhancing its stability and inhibiting HAdV proliferation [2]. (1.0-10 μM; 5 days) BRACO-19 It was discovered that in UXF1138L cells, 1 μM resulted in no growth inhibition, with an IC50 of 2.5 μM and an IC100 of 5 μM [1]. Nuclear hTERT expression was significantly reduced by BRACO-19 (1 μM; 24 h). However, the formation of atypical mitoses was found to be accompanied by persisting cytoplasmic hTERT staining [1]. In eGFP-transfected HEK 293 cells, BRACO-19 (0–40 μM; 24 hours) decreases AdV virus proliferation in a dose-dependent manner [2]. Band intensity gradually decreases in a concentration-dependent manner for BRACO-19 (0-150 μM; 24 h) [2].
BRACO-19 inhibits proliferation in UXF1138L uterine carcinoma cells (IC50 = 2.5 μM, IC100 = 5 μM) in 5-day SRB assays. At subcytotoxic concentrations (1 μM), it induces telomere shortening (0.4 kb after 15 days) and replicative senescence, evidenced by morphological changes and β-galactosidase-positive cells. Immunohistochemistry reveals nuclear hTERT expression loss within 24 hours at 1 μM, with residual cytoplasmic staining. Atypical mitoses and enlarged nuclei indicate telomere dysfunction. TRAP assays confirm telomerase inhibition. Triple staining shows cytoplasmic colocalization of hTERT and ubiquitin after 24-hour treatment (1–10 μM), suggesting ubiquitin-mediated degradation of displaced hTERT.[1] BRACO-19 stabilizes HAdV G-quadruplex structures, increasing their melting temperature (ΔTm) by 13.1°C (PGQ-AV-C3), 27.77°C (PGQ-AV-D2), and 11.7°C (PGQ-AV-E3) in circular dichroism (CD) melting assays. Stabilization occurs via π-π stacking interactions, confirmed by NMR proton broadening.[2] In Taq polymerase stop assays, BRACO-19 (0–150 μM) dose-dependently inhibits DNA synthesis at GQ sites (e.g., PGQ-AV-C3/D2/E3), evidenced by reduced full-length product intensity on gels. Mutant controls show no inhibition.[2] BRACO-19 (20–40 μM) reduces adenovirus proliferation in HEK 293 cells by 65% (20 μM) and 70% (40 μM) via GFP/mTFP reporter assays, confirming GQ-mediated antiviral activity.[2] |
| ln Vivo |
Chronic, intraperitoneal BRACO-19 After administration, qdx5 was effective in inhibiting tumor growth in early-stage xenografts but not in late-stage xenografts [1]. BRACO-19 (ip; 2 mg/kg; 3 weeks; starting 6 days after UXF1138LX fragment transplantation) significantly inhibited tumor growth. Under these conditions, significant single-agent antitumor activity was observed, with some in the group showing complete antitumor activity [1].
In early-stage UXF1138LX xenografts (median volume 68 mm³), chronic i.p. administration of BRACO-19 (2 mg/kg/day, 5 days/week for 3 weeks) achieves 95.9% tumor growth inhibition (T/C = 4.1%, P = 0.018) with partial regressions (5/12 tumors regressed completely). Oral dosing (5 mg/kg twice weekly) is ineffective (T/C = 73%). Immunohistochemistry of treated xenografts shows reduced nuclear hTERT expression, increased cytoplasmic hTERT, and atypical mitotic figures (anaphase bridges, tripolar divisions), indicating telomere uncapping.[1] |
| Enzyme Assay |
TRAP telomerase assay: UXF1138L cell extracts incubated with TS primer for elongation, followed by PCR amplification with ACX primer. Products separated by PAGE and detected via SYBR green staining.[1] Isothermal Titration Calorimetry (ITC) was performed in 80 mM KCl phosphate buffer at 25°C. BRACO-19 (200 μM) was titrated into GQ DNA (PGQ-AV-C3/D2/E3; 20 μM). Data fitted to a two-site model revealed exothermic binding (Kd = 0.28–0.34 × 10⁻⁶ M), while mutants showed negligible affinity.[2] Taq polymerase stop assays used ³²P-labeled primer and GQ templates (10 nM) in Tris-HCl (pH 7.4). Reactions with 0–125 mM KCl showed polymerase arrest at GQ sites. BRACO-19 addition (0–150 μM) intensified arrest, visualized via 15% denaturing PAGE.[2] |
| Cell Assay |
Cell viability assay [1] Cell Types: HEK 293 Cell Tested Concentrations: 20 μM; 40 μM Incubation Duration: 24 hrs (hours) Experimental Results: demonstrated low cytotoxicity and diminished eGFP fluorescence. Cell proliferation: UXF1138L cells (2,000/well) treated with 0.1–10 μM BRACO-19 for 5 days. Viability assessed by sulforhodamine B (SRB) staining (515 nm absorbance). Senescence: Cells treated with 0.1–1 μM BRACO-19 for 15 days, fixed, and stained for β-galactosidase activity (blue pigmentation). hTERT/ubiquitin colocalization: Cells treated with 1–10 μM BRACO-19 for 24 hours, triple-stained with anti-hTERT (TRITC), anti-ubiquitin (FITC), and DAPI. Fluorescence microscopy analyzed colocalization (yellow-orange in merged images). Telomere length: Genomic DNA isolated after 15-day treatment (1 μM), digested with HinfI/RsaI, and analyzed by Southern blot using telomere-specific probes.[1] HEK 293 cells infected with AdV-eGFP (MOI = 3) were treated with BRACO-19 (0–40 μM) in DMEM + 2% FBS. Fluorescence (GFP/mTFP) quantified at 12–24 h post-infection via microscopy/flow cytometry showed dose-dependent viral suppression (30–70% at 20–40 μM).[2] Cytotoxicity assays confirmed BRACO-19 had low toxicity in HEK 293 cells at effective concentrations (≤40 μM).[2] |
| Animal Protocol |
Animal/Disease Models: Establishment of UXF1138LX nude mouse xenograft tumors [1] Doses: 2 mg/kg Route of Administration: intraperitoneal (ip) injection; 3 weeks; Results starting 6 days after UXF1138LX fragment transplantation: showing partial tumor regression, with optimal T/ on day 28 C was 4.1%, equivalent to 95.9% tumor growth inhibition compared to the control. Xenograft efficacy: UXF1138LX tumor fragments implanted s.c. in nude mice. Treatment initiated at early-stage (median 68 mm³) or advanced-stage (130–200 mm³). BRACO-19 dissolved in PBS, administered: - i.p.: 2 mg/kg/day, 5 consecutive days/week for 3 weeks. - p.o.: 5 mg/kg, twice weekly for 2 weeks. Tumor volumes measured twice weekly; %T/C calculated from relative tumor volumes.[1] |
| References |
[1]. The G-quadruplex-interactive Molecule BRACO-19 Inhibits Tumor Growth, Consistent With Telomere Targeting and Interference With Telomerase Function. Cancer Res. 2005 Feb 15;65(4):1489-96. [2]. Genome-wide Analysis Reveals a Regulatory Role for G-quadruplexes During Adenovirus Multiplication. Virus Res. 2020 Jul 2;283:197960. |
| Additional Infomation |
N,N'-(9-{[4-(dimethylamino)phenyl]amino}acridine-3,6-diyl)bis(3-pyrrolidin-1-ylpropanamide) is a member of acridines and a N-alkylpyrrolidine.
BRACO-19 is a trisubstituted acridine G-quadruplex ligand that displaces telomerase from telomeric ends, inducing rapid uncapping and ubiquitin-mediated hTERT degradation. Effective only in tumors with short telomeres (e.g., UXF1138L: 2.7 kb). Oral bioavailability is low; i.p. delivery required for efficacy. Mechanism involves G-quadruplex stabilization → hTERT displacement → telomere dysfunction (atypical mitoses) → senescence/apoptosis. No ADME/toxicity data reported. [1] BRACO-19 is a trisubstituted acridine derivative that stabilizes viral G-quadruplexes, disrupting replication. It previously showed antiviral activity against HIV-1 and HSV by similar mechanisms.[2] This study identifies HAdV GQs as druggable targets; BRACO-19 represents a prototype for anti-adenoviral therapeutics targeting genomic secondary structures.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6842 mL | 8.4209 mL | 16.8418 mL | |
| 5 mM | 0.3368 mL | 1.6842 mL | 3.3684 mL | |
| 10 mM | 0.1684 mL | 0.8421 mL | 1.6842 mL |