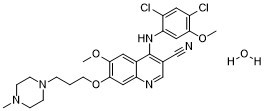
Bosutinib hydrate (SKI606; KIN001160; SKI-606; KIN-001160; Bosulif), the mono-hydrated form of bosutinib, is a synthetic quinolone derivative and dual kinase inhibitor targeting both Abl and Src kinases with potential antineoplastic activity. In 2012, bosutinib was licensed for the treatment of adult patients with chronic myelogenous leukemia (CML), which is chromosome-positive (Ph+).
Physicochemical Properties
| Molecular Formula | C26H31CL2N5O4 |
| Molecular Weight | 548.465 |
| Exact Mass | 547.175 |
| Elemental Analysis | C, 56.94; H, 5.70; Cl, 12.93; N, 12.77; O, 11.67 |
| CAS # | 918639-08-4 |
| Related CAS # | Bosutinib;380843-75-4;Bosutinib-d8 |
| PubChem CID | 11990828 |
| Appearance | White to off-white solid powder |
| LogP | 4.423 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 37 |
| Complexity | 734 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | N#CC1C(NC2C(Cl)=CC(Cl)=C(OC)C=2)=C2C(C=C(C(=C2)OC)OCCCN2CCN(C)CC2)=NC=1.O |
| InChi Key | BXPOSPOKHGNMEP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H29Cl2N5O3.H2O/c1-32-6-8-33(9-7-32)5-4-10-36-25-13-21-18(11-24(25)35-3)26(17(15-29)16-30-21)31-22-14-23(34-2)20(28)12-19(22)27;/h11-14,16H,4-10H2,1-3H3,(H,30,31);1H2 |
| Chemical Name | 4-(2,4-dichloro-5-methoxyanilino)-6-methoxy-7-[3-(4-methylpiperazin-1-yl)propoxy]quinoline-3-carbonitrile;hydrate |
| Synonyms | KIN-001160; SK606; KIN001-160; SKI 606; KIN 001160; KIN001160; SKI606; SK-I606; SK-606; SK 606; KIN 001-160; KIN-001-160; trade name: Bosulif. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Csk (IC50 = 314 nmol/L); Abl kinase (IC50 = 2.4 nmol/L)
Bosutinib hydrate targets BCR-ABL (Ki = 1.2 nM; IC50 = 3.7 nM for ABL kinase activity); Src family kinases including Src (Ki = 1.5 nM), Lyn (Ki = 1.8 nM), Hck (Ki = 2.4 nM), Fyn (Ki = 4.3 nM) [1] Bosutinib hydrate inhibits BCR-ABL T315I mutant (IC50 = 240 nM) and wild-type BCR-ABL (IC50 = 45 nM) in kinase assays [3] Bosutinib hydrate shows activity against c-Kit (IC50 = 62 nM) and PDGFRβ (IC50 = 68 nM) [1] |
| ln Vitro |
Bosutinib has an IC50 of 1.2 nM for Src family kinases and an IC50 of 100 nM for potently inhibiting Src-dependent cell proliferation. [1] With IC50 values of 5 nM, 20 nM, and 20 nM, respectively, bosutinib more potently inhibits the proliferation of Bcr-Abl-positive leukemia cell lines KU812, K562, and MEG-01 but not Molt-4, HL-60, Ramos, or other leukemia cell lines. Bosutinib exhibits antiproliferative activity against Abl-MLV-transformed fibroblasts with an IC50 of 90 nM, which is comparable to STI-571. At concentrations of approximately 50 nM, 10–25 nM, and 200 nM, respectively, bosutinib inhibits the tyrosine phosphorylation of Bcr-Abl and STAT5 in CML cells as well as v-Abl expressed in fibroblasts. This results in the inhibition of Lyn/Hck phosphorylation by Bcr-Abl downstream signaling.[2] With an IC50 of approximately 250 nM, bosutinib significantly reduces the motility and invasion of breast cancer cells, albeit it is unable to inhibit the proliferation and survival of these cells. This effect is linked to an increase in cell-to-cell adhesion and β-catenin membrane localization.[3] In wild-type BCR-ABL-positive CML cell lines (K562, KU812), Bosutinib hydrate inhibits cell proliferation with IC50 values of 25 nM and 32 nM, respectively; induces G2/M cell cycle arrest and apoptosis (Annexin V staining shows 35–40% apoptotic cells at 100 nM after 48 h) [1] - In imatinib-resistant CML cell lines (K562-R, expressing BCR-ABL T315I), Bosutinib hydrate exhibits antiproliferative activity with IC50 = 180 nM, significantly lower than imatinib (IC50 > 1 μM) [3] - Inhibits phosphorylation of BCR-ABL and downstream signaling molecules (STAT5, CrkL, ERK1/2) in K562 cells, as detected by Western blot; maximum inhibition of phospho-ABL is achieved at 50 nM [2] - In breast cancer cell lines (MDA-MB-231, BT-474) with high Src expression, Bosutinib hydrate inhibits cell migration and invasion (transwell assay shows 60–70% reduction in migrated cells at 100 nM) and proliferation (IC50 = 75–90 nM) [6] - In acute lymphoblastic leukemia (ALL) cell lines (SUP-B15, BV173), Bosutinib hydrate suppresses cell growth (IC50 = 42–58 nM) and induces caspase-dependent apoptosis [5] |
| ln Vivo |
Bosutinib (60 mg/kg/day) is effective against HT29 xenografts and Src-transformed fibroblast xenografts in nude mice with T/C values of 18% and 30%, respectively.[1] When mice are given Bosutinib orally for five days, the growth of K562 tumors is significantly suppressed. This effect is dose-dependent, with large tumors being completely removed at a dose of 100 mg/kg and tumor free at 150 mg/kg without causing overt toxicity.[2] Whereas bosutinib's significant dose-dependent ability against HT29 xenografts contrasts with its inactive nature against Colo205 xenografts in nude mice at 50 mg/kg twice daily, bosutinib's dosing at 75 mg/kg twice daily is necessary against Colo205 xenografts, and increasing the dose has no additional benefit.[4] In K562 xenograft nude mouse model, oral administration of Bosutinib hydrate (50 mg/kg, twice daily for 21 days) inhibits tumor growth by 82% compared to vehicle control; tumor tissue analysis shows reduced phospho-ABL and STAT5 expression [2] - In imatinib-resistant K562-R xenograft model, Bosutinib hydrate (100 mg/kg, oral, twice daily) achieves 65% tumor growth inhibition, with no significant tumor regrowth during treatment [3] - In CML bone marrow transplantation mouse model (expressing wild-type BCR-ABL), Bosutinib hydrate (75 mg/kg, oral, daily for 28 days) reduces leukemic cell burden in peripheral blood and bone marrow by 70–75% [4] - In MDA-MB-231 breast cancer xenograft model, Bosutinib hydrate (80 mg/kg, oral, daily) inhibits tumor growth by 58% and reduces lung metastasis (number of metastatic nodules decreased by 62%) [6] |
| Enzyme Assay |
ELISA is used to measure Src kinase activity. Reaction buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl2, 0.1 mM EGTA, 0.5 mM Na3VO4), Src (3 units/reaction), and cdc2 substrate peptide are added to varying concentrations of bosutinib and incubated for 10 minutes at 30 °C. A final concentration of 100 μM of ATP is added to initiate the reaction, which is then incubated for an hour at 30 °C before being stopped with the addition of EDTA. The next steps are carried out in accordance with the manufacturer's instructions. Using a DELFIA solid phase europium-based detection assay format, the Abl kinase assay is carried out. For 1.5 hours, 1 mg/mL ovalbumin in PBS is used to bind biotinylated peptide (2 μM) to streptavidin-coated microtitration plates. A PBS wash is performed after an hour of washing the plates with PBS/0.1% Tween 80. One hour at 30°C is spent heating the kinase reaction. The following mixture contains 10 units of Abl kinase: 50 mM Tris-HCl (pH 7.5), 10 mM MgCl2, 80 μM EGTA, 100 μM ATP, 0.5 mM Na3VO4, 1% DMSO, 1 mM HEPES (pH 7.0), 200 μg/mL ovalbumin, and different concentrations of bosutinib. EDTA, at a final concentration of 50 mM, is used to stop the reaction. Equine phosphotyrosine antibody labeled with Eu and DELFIA enhancement solution are used to track the reaction. ABL kinase activity assay: Recombinant ABL kinase domain is incubated with ATP (10 μM) and a biotinylated peptide substrate in the presence of serial dilutions of Bosutinib hydrate. After 60 min incubation at 30°C, the reaction is terminated, and phosphorylated substrate is detected using a streptavidin-conjugated detection system. The IC50 value is calculated by nonlinear regression of kinase activity vs. drug concentration [1] - Src family kinase activity assay: Recombinant Src, Lyn, or Hck kinase is mixed with a fluorescent peptide substrate and ATP, followed by addition of Bosutinib hydrate (0.1–1000 nM). Kinase activity is measured by detecting fluorescence polarization of phosphorylated substrate after 45 min incubation at 37°C. Ki values are determined using Michaelis-Menten kinetics [1] - BCR-ABL mutant (T315I) kinase assay: Recombinant T315I mutant ABL kinase is incubated with substrate and ATP in the presence of Bosutinib hydrate. Phosphorylated product is quantified via ELISA, and IC50 is calculated by plotting inhibition percentage against drug concentration [3] |
| Cell Assay |
Bosutinib is applied to cells at different concentrations for a duration of 72 hours. In 96-well ultra-low binding plates treated with Sigmacote to prevent residual cell attachment, the anchorage-independent proliferation of Abl-MLV-transformed fibroblasts is assessed. Cell-Glo or MTS are used to measure cell proliferation. Using the CycleTest Plus DNA reagent kit, cells are prepared for FACS analysis and examined on a fluorescence-activated cell sorter flow cytometer to determine whether the cells are in the cell cycle or have died. Cell proliferation assay: CML or breast cancer cells are seeded in 96-well plates (4 × 103 cells/well) and treated with Bosutinib hydrate (0.1–1000 nM) for 72 h. Cell viability is assessed by adding a colorimetric reagent, incubating for 3 h, and reading absorbance at 570 nm. IC50 values are derived from dose-response curves [1] - Western blot for signaling pathways: K562 cells are treated with Bosutinib hydrate (10–100 nM) for 2 h, then lysed in ice-cold lysis buffer. Lysates are separated by SDS-PAGE, transferred to membranes, and probed with antibodies against phospho-ABL, total ABL, phospho-STAT5, phospho-ERK1/2, and GAPDH. Band intensity is quantified using densitometry [2] - Apoptosis assay: SUP-B15 cells are treated with Bosutinib hydrate (50–200 nM) for 48 h, harvested, and stained with Annexin V-FITC and PI. Apoptotic cells are analyzed by flow cytometry, with early (Annexin V+/PI-) and late (Annexin V+/PI+) apoptosis combined for quantification [5] - Migration and invasion assay: MDA-MB-231 cells are seeded in transwell inserts (upper chamber) coated with (invasion) or without (migration) Matrigel. Bosutinib hydrate (50–100 nM) is added to the upper chamber, and cells are incubated for 24 h. Migrated/invaded cells on the lower membrane are stained and counted under a microscope [6] |
| Animal Protocol |
Nude female mice injected with K562 cells ~150 mg/kg/day Oral gavage K562 xenograft model: Female nude mice (6–7 weeks old) are subcutaneously injected with 2 × 106 K562 cells into the right flank. When tumors reach 100–150 mm3, mice are randomly divided into vehicle and treatment groups (n = 8 per group). Bosutinib hydrate is suspended in 0.5% methylcellulose and administered orally at 50 mg/kg twice daily for 21 days. Tumor volume is measured every 3 days, and mice are euthanized at study end for tumor collection [2] - Imatinib-resistant K562-R xenograft model: Nude mice are implanted with 2 × 106 K562-R cells. Once tumors reach 120–180 mm3, Bosutinib hydrate (100 mg/kg, oral, twice daily) or vehicle is administered for 28 days. Body weight and tumor volume are monitored weekly, and tumor lysates are analyzed for phospho-ABL expression [3] - CML bone marrow transplantation model: Syngeneic mice are transplanted with bone marrow cells expressing wild-type BCR-ABL. Two weeks post-transplantation, mice are treated with Bosutinib hydrate (75 mg/kg, oral, daily) for 28 days. Peripheral blood and bone marrow are collected to quantify leukemic cell burden via flow cytometry [4] - Breast cancer metastasis model: Nude mice are injected with 5 × 105 MDA-MB-231 cells via tail vein. One day later, Bosutinib hydrate (80 mg/kg, oral, daily) is administered for 4 weeks. Mice are euthanized, and lungs are harvested to count metastatic nodules [6] |
| ADME/Pharmacokinetics |
In rats, oral administration of Bosutinib hydrate (25 mg/kg) shows bioavailability of 34 ± 5%, with Cmax = 1.2 ± 0.2 μM achieved at 2 h post-dosing [1] - Plasma half-life (t1/2) in rats is 6.8 ± 1.1 h, and AUC0–24h is 9.7 ± 1.5 μM·h [1] - In mice, oral Bosutinib hydrate (50 mg/kg) exhibits tissue distribution with high levels in liver (tissue/plasma ratio = 4.2 ± 0.6), moderate in kidney (2.8 ± 0.4), and low in brain (0.3 ± 0.1) [4] - Metabolic studies in human liver microsomes show Bosutinib hydrate is metabolized primarily via CYP3A4, with minor contributions from CYP2C9 and CYP2C19 [7] |
| Toxicity/Toxicokinetics |
In 28-day repeated-dose toxicity study in rats (oral doses of 25, 50, 100 mg/kg/day), Bosutinib hydrate causes mild weight loss (≤8%) at 100 mg/kg, with no significant changes in ALT, AST, BUN, or creatinine levels. Histopathological examination shows mild hepatocellular vacuolation at 100 mg/kg [4] - Plasma protein binding rate of Bosutinib hydrate is 94 ± 2% in human plasma and 92 ± 3% in rat plasma, determined by equilibrium dialysis [1] - No significant drug-drug interaction with warfarin (CYP2C9 substrate) or omeprazole (CYP2C19 substrate) in in vitro microsomal studies; moderate interaction with midazolam (CYP3A4 substrate) due to competitive inhibition [7] - In mice, the maximum tolerated dose (MTD) of oral Bosutinib hydrate is 150 mg/kg/day (twice daily dosing), with lethal dose (LD50) > 200 mg/kg [3] |
| References |
[1]. J Med Chem . 2001 Nov 8;44(23):3965-77. [2]. Cancer Res . 2003 Jan 15;63(2):375-81. [3]. Mol Cancer Ther . 2008 May;7(5):1185-94. [4]. Cancer Res . 2005 Jun 15;65(12):5358-64. [5]. Leukemia . 2010 Jun;24(6):1223-7. [6]. Oncol Rep . 2011 Mar;25(3):661-7. [7]. Oncotarget . 2017 Jan 3;8(1):1469-1480. |
| Additional Infomation |
Bosutinib hydrate is a hydrate that is the monohydrate form of anhydrous bosutinib. It has a role as an antineoplastic agent and a tyrosine kinase inhibitor. It contains a bosutinib. Bosutinib Monohydrate is the monohydrate form of bosutinib, a synthetic quinolone derivative and dual kinase inhibitor that targets both Abl and Src kinases with potential antineoplastic activity. Unlike imatinib, bosutinib inhibits the autophosphorylation of both Abl and Src kinases, resulting in inhibition of cell growth and apoptosis. Because of the dual mechanism of action, this agent may have activity in resistant CML disease, other myeloid malignancies and solid tumors. Abl kinase is upregulated in the presence of the abnormal Bcr-abl fusion protein which is commonly associated with chronic myeloid leukemia (CML). Overexpression of specific Src kinases is also associated with the imatinib-resistant CML phenotype. See also: Bosutinib (has active moiety). Drug Indication Bosulif is indicated for the treatment of adult patients with: newlyâdiagnosed chronic phase (CP) Philadelphia chromosome-positive chronic myelogenous leukaemia (Ph+ CML). CP, accelerated phase (AP), and blast phase (BP) Ph+ CML previously treated with one or more tyrosine kinase inhibitor(s) [TKI(s)] and for whom imatinib, nilotinib and dasatinib are not considered appropriate treatment options. Bosutinib hydrate is a dual BCR-ABL/Src family tyrosine kinase inhibitor, designed to overcome imatinib resistance in CML by targeting both BCR-ABL and Src-mediated signaling pathways [1] - Resistance to Bosutinib hydrate in CML cells is associated with mutations in BCR-ABL (e.g., E255K, Y253H) or increased expression of ABCG2 efflux pump [3] - Bosutinib hydrate exhibits synergistic antiproliferative activity with dasatinib in imatinib-resistant CML cell lines, with combination index (CI) < 1 [7] - In preclinical studies, Bosutinib hydrate shows efficacy in both chronic phase and blast crisis CML, as well as Src-driven solid tumors (breast, prostate cancer) [6] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8233 mL | 9.1163 mL | 18.2325 mL | |
| 5 mM | 0.3647 mL | 1.8233 mL | 3.6465 mL | |
| 10 mM | 0.1823 mL | 0.9116 mL | 1.8233 mL |