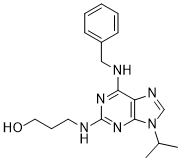
Bohemine, structurally similar to Olomoucine and Roscovitine, is a novel, potent, selective, and cell-permeable cyclin-dependent kinase (CDK) with IC50s of 4.6, 83, and 2.7 μM for Cdk2/cyclin E, Cdk2/cyclin A, and Cdk9/cyclin T1, respectively. Bohemine exhibits a 52 μM IC50 for ERK2 inhibition, while its impact on CDK1, CDK4, and CDK6 is lessened. The anti-cancer effects of bohemine are wide-ranging.
Physicochemical Properties
| Molecular Formula | C18H24N6O |
| Molecular Weight | 340.42276 |
| Exact Mass | 340.201 |
| Elemental Analysis | C, 63.51; H, 7.11; N, 24.69; O, 4.70 |
| CAS # | 189232-42-6 |
| Related CAS # | 189232-42-6 |
| PubChem CID | 2422 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 589.4±60.0 °C at 760 mmHg |
| Flash Point | 310.3±32.9 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.653 |
| LogP | 1.02 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 25 |
| Complexity | 390 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC(C)N1C=NC2=C(NCC3=CC=CC=C3)NC(=NCCCO)N=C21 |
| InChi Key | OPQGFIAVPSXOBO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H24N6O/c1-13(2)24-12-21-15-16(20-11-14-7-4-3-5-8-14)22-18(23-17(15)24)19-9-6-10-25/h3-5,7-8,12-13,25H,6,9-11H2,1-2H3,(H2,19,20,22,23) |
| Chemical Name | 3-[[6-(benzylamino)-9-propan-2-ylpurin-2-yl]amino]propan-1-ol |
| Synonyms | Bohemine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CDK2/cyclinE (IC50 = 4.6 μM); cdk2/cyclin A (IC50 = 83 μM); CDK9/cyclinT1 (IC50 = 2.7 μM); ERK2 (IC50 = 52 μM) |
| ln Vitro |
Bohemine treatment inhibits cell growth. When bohemine is added at concentrations between 1 and 10 µM, growth and the production of monoclonal antibodies are temporarily stopped. Following the brief suppression of cell functions, there is a notable, transient increase in both the rate of specific growth and production[1]. The G1/S and G2/M boundaries of hybridoma cells are retarded, based on the concentration of bohemine (0-30 µM)[1]. T-cell lymphoblastic line Five proteins are shown to be downregulated when bohemine is used to treat CEM: α-enolase, triosephosphate isomerase, initiation factor 5A, and the α- and β-subunits of Rho GDP-dissociation inhibitor 1. These proteins are important for the processes of glycolysis, proteosynthesis, and cytoskeleton rearrangement[1]. With an IC50 of 27 µM, bohemine inhibits the growth of human tumor cell lines[2]. |
| ln Vivo | Bohemine (50 mg/kg; intravenous injection; BALB/c mice) treatment reveals a T1/2 of 1.39 hours, an observed clearance of 0.23 L/h, and a Cmax of 72,308 nM[2]. |
| Animal Protocol |
BALB/c mice bearing the colon 26 murine tumor[2] 50 mg/kg Intravenous injection (Pharmacokinetic Analysis) |
| References |
[1]. Diverse effects of the cyclin-dependent kinase inhibitor bohemine: Concentration- and time-dependent suppression or stimulation of hybridoma culture. Cytotechnology. 2001 Jul;36(1-3):117-23. [2]. In vitro and in vivo pharmacokinetic-pharmacodynamic relationships for the trisubstituted aminopurine cyclin-dependent kinase inhibitors olomoucine, bohemine and CYC202. Clin Cancer Res. 2005 Jul 1;11(13):4875-87. |
| Additional Infomation | Bohemine is purine substituted on C-2, C-6 and N-9 with (3-hydroxypropyl)amino, benzylamino and isopropyl groups respectively; a synthetic, cell-permeable, cyclin-dependent kinase (CDK) inhibitor that is structurally similar to olomoucine and roscovitine. It has a role as an EC 2.7.11.22 (cyclin-dependent kinase) inhibitor. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 68~100 mg/mL (199.8~293.8 mM) Ethanol: ~68 mg/mL (199.8 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9375 mL | 14.6877 mL | 29.3755 mL | |
| 5 mM | 0.5875 mL | 2.9375 mL | 5.8751 mL | |
| 10 mM | 0.2938 mL | 1.4688 mL | 2.9375 mL |