Physicochemical Properties
| Molecular Formula | C18H12N3OF3CL2 |
| Molecular Weight | 414.20858 |
| Exact Mass | 413.031 |
| Elemental Analysis | C, 52.20; H, 2.92; Cl, 17.12; F, 13.76; N, 10.14; O, 3.86 |
| CAS # | 581809-46-3 |
| PubChem CID | 11434448 |
| Appearance | Typically exists as solid at room temperature |
| Boiling Point | 474.653 ℃ at 760mmHg |
| Melting Point | 146 ℃ |
| Flash Point | 240.862 ℃ |
| LogP | 6.106 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 27 |
| Complexity | 530 |
| Defined Atom Stereocenter Count | 0 |
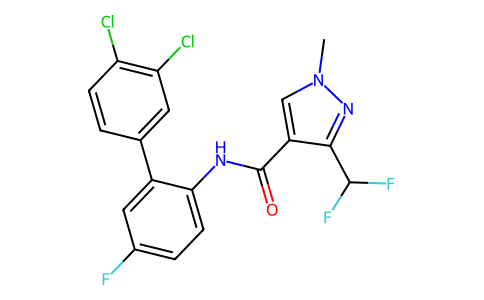
| SMILES | CN1C=C(C(NC2=C(C3=CC(Cl)=C(Cl)C=C3)C=C(F)C=C2)=O)C(C(F)F)=N1 |
| InChi Key | LDLMOOXUCMHBMZ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H12Cl2F3N3O/c1-26-8-12(16(25-26)17(22)23)18(27)24-15-5-3-10(21)7-11(15)9-2-4-13(19)14(20)6-9/h2-8,17H,1H3,(H,24,27) |
| Chemical Name | N-[2-(3,4-dichlorophenyl)-4-fluorophenyl]-3-(difluoromethyl)-1-methylpyrazole-4-carboxamide |
| Synonyms | Bixafen; 581809-46-3; Bixafen [ISO]; bixafene; UNII-28XK2L8M3B; 28XK2L8M3B; AVIATOR XPRO; BOOGIE XPRO; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Succinate dehydrogenase |
| ln Vivo | Bixafen (BIX), a member of the succinate dehydrogenase inhibitor (SDHI) class of fungicides, has seen a surge in interest due to its expanding market presence and positive development outlook. However, there is a growing concern about its potential harm to aquatic life, largely due to its resistance to breaking down in the environment. In this study, we thoroughly examined the toxicological impact of BIX on zebrafish as a model organism. Our results revealed that BIX significantly hindered the development of zebrafish embryos, leading to increased mortality, hatching failures, and oxidative stress. Additionally, we observed cardiovascular abnormalities, including dilated cardiac chambers, reduced heart rate, sluggish blood circulation, and impaired vascular function. Notably, BIX also altered the expression of key genes involved in cardiovascular development, such as myl7, vmhc, nkx2.5, tbx5, and flt1. In summary, BIX was found to induce developmental and cardiovascular toxicity in zebrafish, underscoring the risks associated with SDHI pesticides and emphasizing the need for a reassessment of their impact on human health. These findings are crucial for the responsible use of BIX [1]. |
| Animal Protocol |
Bixafen exposure [1] Following the Organization for Economic Co-operation and Development (OECD) test guideline No. 236, zebrafish embryos were exposed to a range of BIX concentrations (0.05, 0.1, 0.2, 0.4, and 0.8 μM) to evaluate their survival rates (OECD, 2013). Each treatment group contained 30 post-fertilized eggs in 10 mL of solution within Petri dishes. The DMSO solution at the same concentration served as the solvent control. Embryos were exposed to various BIX concentrations for 72 h post-fertilization (hpf). To ensure a constant BIX concentration, the solution was replaced every 24 h. Each treatment group was replicated three times to confirm reproducibility. The highest concentration of BIX that did not result in significant embryonic lethality by 72hpf was designated as the exposure concentration. Survival rates were measured at 24, 48, and 72 hpf, while hatching rates were noted at 48 and 72 hpf. Furthermore, measurements of eye length, trunk pigmentation area, body length, and deformity rates were conducted at 72 hpf, and the observation of autonomous movement was made at 24 hpf. |
| Toxicity/Toxicokinetics |
RAIS Toxicity Values Oral Acute Reference Dose (RfDoa)(mg/kg-day) 2.5 Oral Acute Reference Dose Reference OPP Oral Chronic Reference Dose (RfDoc) (mg/kg-day) 0.03 Oral Chronic Reference Dose Reference OPP |
| References | [1]. Insights into the developmental and cardiovascular toxicity of bixafen using zebrafish embryos and larvae. Environ Res. 2024 Dec 1;262(Pt 2):119916. |
| Additional Infomation | Bixafen is an aromatic amide obtained by formal condensation of the carboxy group of 3-(difluoromethyl)-1-methylpyrazole-4-carboxylic acid with the amino group of 3',4'-dichloro-5-fluorobiphenyl-2-amine. A fungicide for use in cereals for key stem and leaf disease control including strobilurin-resistant septoria. It has a role as an EC 1.3.5.1 [succinate dehydrogenase (quinone)] inhibitor and an antifungal agrochemical. It is an aromatic amide, an organofluorine compound, a member of pyrazoles, a member of biphenyls, a dichlorobenzene and an anilide fungicide. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4142 mL | 12.0712 mL | 24.1423 mL | |
| 5 mM | 0.4828 mL | 2.4142 mL | 4.8285 mL | |
| 10 mM | 0.2414 mL | 1.2071 mL | 2.4142 mL |