Physicochemical Properties
| Molecular Formula | C22H24CLFN2O2 |
| Molecular Weight | 402.89 |
| Exact Mass | 402.15103 |
| CAS # | 3106363-91-8 |
| Related CAS # | 3106363-91-8 ; Bfl-1-IN-6 trifluoroacetate |
| Appearance | Typically exists as solids at room temperature |
| LogP | 4.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 28 |
| Complexity | 541 |
| Defined Atom Stereocenter Count | 3 |
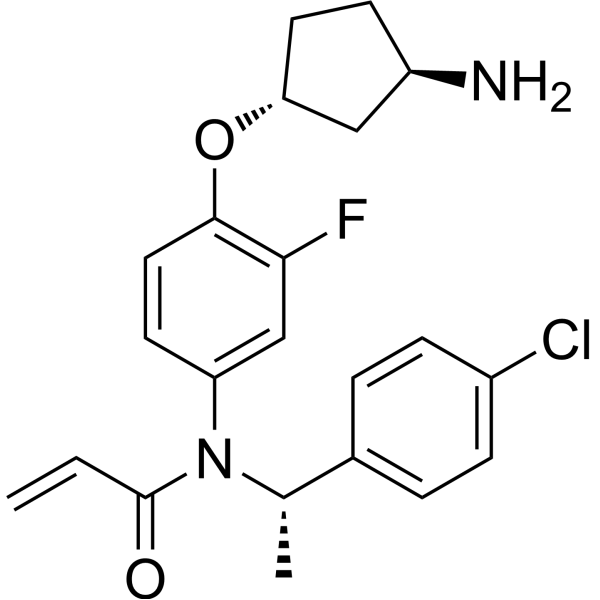
| SMILES | C[C@@H](C1=CC=C(C=C1)Cl)N(C2=CC(=C(C=C2)O[C@@H]3CC[C@H](C3)N)F)C(=O)C=C |
| InChi Key | PDKKJSNSPGZTJL-POZUXBRTSA-N |
| InChi Code | InChI=1S/C22H24ClFN2O2/c1-3-22(27)26(14(2)15-4-6-16(23)7-5-15)18-9-11-21(20(24)13-18)28-19-10-8-17(25)12-19/h3-7,9,11,13-14,17,19H,1,8,10,12,25H2,2H3/t14-,17+,19+/m0/s1 |
| Chemical Name | N-[4-[(1R,3R)-3-aminocyclopentyl]oxy-3-fluorophenyl]-N-[(1S)-1-(4-chlorophenyl)ethyl]prop-2-enamide |
| Synonyms | Bfl-1-IN-6; Bfl-1-IN6; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | BFL1 (Bcl-2-related protein A1). The compound is a covalent inhibitor selectively targeting BFL1 over other BCL2 family members (BCL-xl, BCL2, MCL1 all had IC50 >30 μM in FRET assays). [1] |
| ln Vitro |
- Biochemical Activity: Compound 20 demonstrated potent inhibition in a BFL1 TR-FRET assay with an IC50 of 19 nM. Its second-order rate constant (kinact/KI) for covalent binding was determined to be 11,000 M⁻¹·s⁻¹. [1]
- Cellular Activity (Caspase Activation): In SUDHL1 cells (which are codependent on BFL1 and MCL1), treatment with 20 in combination with the MCL1 inhibitor AZD5991 (0.5 μM) resulted in caspase 3/7 activation with an EC50 of 0.35 μM. [1] - Cellular Activity (Cell Viability): In a cell viability assay using SUDHL1 cells in combination with 0.5 μM AZD5991, 20 showed an EC50 of 0.25 μM. In the BFL1-null Karpas-422 cell line, no loss of cell viability was observed with 20 treatment, suggesting minimal off-target toxicity. [1] - Mechanism of Action (BIM Displacement): 20 was shown to displace the pro-apoptotic protein BIM from BFL1, which is consistent with the mechanism of action of BH3-mimetic antagonists. This displacement leads to the re-association of BIM with MCL1, which can be mitigated by combination with an MCL1 inhibitor. [1] - Mechanism of Action (Protein Stabilization): In a pulsed-SILAC experiment using OCILY10 cells, treatment with 3.2 μM of 20 significantly extended the half-life of the BFL1 protein from 2.6 hours (basal) to 10.8 hours, indicating a stabilizing effect due to high target occupancy. [1] - Target Occupancy: In SUDHL1 cells, targeted proteomics showed that 20 decreased the levels of the unbound (apo) BFL1 peptide and increased the levels of the drug-bound (adduct) peptide in a dose- and time-dependent manner. At 1 μM and 24 hours, 20 showed slightly improved occupancy compared to the earlier compound 7. [1] - Selectivity Profiling: In a panel of 24 distinct molecular targets (receptors, ion channels, transporters, enzymes), 20 showed binding activity below 1 μM only for the CB1 receptor, but it did not show functional activity (IC50 >100 μM). In a panel of 48 kinases, no activity >50% inhibition was observed at a concentration of 1 μM. [1] |
| ln Vivo | Pharmacodynamics (PD) in Tumor Model: In mice bearing OCILY10 tumors, a single oral dose of 20 at 100 mg/kg and 300 mg/kg resulted in dose-dependent BFL1 stabilization and cleaved caspase-3 activation in tumor tissue 6 hours post-treatment. These effects were consistent with the plasma exposures predicted from pharmacokinetic studies. [1] |
| Enzyme Assay |
- BFL1 TR-FRET Assay: To screen compounds for their ability to block the interaction between BFL1 and the BH3-only protein BIM, a TR-FRET assay was established. The assay was performed at a 2-hour time point. Compounds were incubated with BFL1 protein and a labeled BIM peptide. Inhibition of the BFL1-BIM interaction by the test compound results in a decreased FRET signal. IC50 values were calculated from normalized dose-response FRET data using curve fitting software. For compound 20, this assay yielded an IC50 of 19 nM. [1] - kinact/KI Determination (Covalent Binding Efficiency): The second-order rate constant, which describes the efficacy of covalent bond formation, was determined for key compounds. In this assay, BFL1 protein (600 nM in 20 mM Tris pH 7.4, 150 mM NaCl) was incubated with a log10 concentration range of the test compound (0.5 to 50 μM) for seven different incubation times (1 to 90 seconds). The reaction was stopped by the addition of a quench solution (1.6% formic acid). The fraction of protein modified by the compound was measured using intact protein mass spectrometry. The observed rate constants (kobs) were plotted against inhibitor concentration and fitted to a kinetic model to derive the kinact/KI value. For compound 20, the kinact/KI was determined to be 11,000 M⁻¹·s⁻¹. [1] - Glutathione (GSH) Reactivity Assay: To monitor the reactivity of the acrylamide warhead and avoid promiscuous compounds, a GSH reactivity assay was performed. Compounds were incubated with an excess of GSH, and the half-life (t1/2) of compound consumption was measured by mass spectrometry. The target was a GSH t1/2 >100 min. Compound 20 had a GSH t1/2 of 140 min. [1] |
| Cell Assay |
- Caspase Activation Assay in SUDHL1 Cells: To measure the potency of compounds in an efficacy assay, a Caspase 3/7 assay was used. SUDHL1 cells (3000 cells/well) were seeded in 384-well white plates containing pre-dispensed compound 20 at a 10-point dilution range (0.3 nM to 10 μM) in combination with a fixed dose of 0.5 μM AZD5991 (MCL1 inhibitor). The final DMSO concentration was 0.1%. Neutral controls were treated with 0.5 μM AZD5991 alone, and positive controls were treated with a combination of 0.5 μM AZD5991 and 0.5 μM AZD4320 (BCL-xL inhibitor). After a 6-hour incubation at 37°C, 5% CO2, Caspase-Glo 3/7 reagent was added. The luminescent signal, proportional to caspase 3/7 activity, was measured after 30 minutes. The EC50 for 20 in this assay was 0.35 μM. [1] - Cell Viability Assay in SUDHL1 and Karpas-422 Cells: To profile potency and selectivity, a cell viability assay measuring total ATP content (CellTiter-Glo) was used. SUDHL1 (BFL1-expressing) and Karpas-422 (BFL1-null) cells (4000 cells/well) were seeded in 384-well plates containing pre-dispensed compound 20 at a 10-point dilution range (0.3 nM to 10 μM). For combination studies in SUDHL1 cells, a fixed dose of 0.5 μM AZD5991 was included. After 24 hours of incubation at 37°C, 5% CO2, CellTiter-Glo 2.0 reagent was added, and luminescence was measured. In SUDHL1 cells with AZD5991 combination, 20 had a cell viability EC50 of 0.25 μM. No effect on viability was seen in Karpas-422 cells. [1] - BFL1 Target Occupancy Assay in SUDHL1 Cells: To measure target occupancy, SUDHL1 cells were treated in duplicate with DMSO or 20 at 10, 1, or 0.1 μM for 2, 6, or 24 hours. Cells were collected, washed, and lysed. Heavy standard peptides were spiked into the samples for absolute quantitation. An unaffected BFL1 peptide, the apo-peptide (containing the unbound Cys55), and the adduct-peptide (with 20 covalently bound) were monitored by mass spectrometry. Occupancy was calculated from the ratio of apo-BFL1 to total BFL1. 20 showed dose- and time-dependent increases in occupancy. [1] - BFL1 Protein Turnover (pulsed-SILAC) Assay in OCILY10 Cells: OCILY10 cells were fully labeled in "Heavy" media. They were then pretreated with DMSO or 3.2 μM 20 for 1 hour. At the start of the experiment (t=0), cells were switched to "Light" media containing the same treatment. Cells were collected at various time points (0 min to 24 h). BFL1 peptides were monitored by mass spectrometry to track the degradation of the pre-existing "Heavy" protein and the synthesis of new "Light" protein. The half-life of BFL1 was calculated from the decay of the "Heavy" signal. Treatment with 20 extended the BFL1 half-life from 2.6 hours (DMSO) to 10.8 hours. [1] - MSD Assay for BFL1 Stabilization and Cleaved Caspase-3 in OCILY10 Cells: To evaluate target engagement, an MSD (Meso Scale Discovery) assay was developed. OCILY10 cells were pre-treated with Q-VD-OPH (50 μM) overnight, then treated with 20 (0.3, 1, 3, 9 μM) or DMSO for 6 hours. Cell lysates were collected and protein concentration was normalized. For the BFL1 stabilization assay, plates were coated with a capture antibody for BFL1. After blocking and washing, lysates were added, followed by a primary detection antibody and a SULFO-TAG labeled secondary antibody. For the cleaved caspase-3 assay, a commercial kit was used per the manufacturer's protocol. Both assays showed dose-dependent increases in BFL1 stabilization and cleaved caspase-3 signal upon treatment with 20. [1] |
| Animal Protocol |
- Pharmacokinetic (PK) Study in Mice: Male CD-1 mice (n=2 per route) were dosed with compound 20. The intravenous (iv) dose was 0.5 mg/kg administered via tail vein bolus. The oral (po) dose was 1 mg/kg administered by gavage. The formulation for both routes was a solution in 5% DMSO and 95% SBE-β-CD (30% w/v) in water. Blood samples were collected from the dorsal metatarsal vein at 2, 5, 30 min, 1, 2, 4, 8, and 24 h (iv) and at 15, 30 min, 1, 2, 4, 8, and 24 h (po). Plasma was isolated and analyzed by LC-MS/MS. [1] - Pharmacokinetic (PK) Study at Higher Dose: Female SCID mice were dosed orally with 20 at 30 mg/kg to determine plasma concentration at the 6-hour time point. The formulation used was a suspension in 0.5% hydroxypropylmethylcellulose/0.1% Tween in water. [1] - In Vivo Pharmacodynamics (PD) Study: Female C.B.-17 SCID mice were subcutaneously implanted with 5 million OCILY10 cells in the right flank. When tumors were established, mice were given a single oral dose of 20 at 100 mg/kg or 300 mg/kg. The formulation was in water with the pH adjusted to 2.3-2.5. Six hours post-dose, tumors were collected, homogenized, and analyzed for BFL1 stabilization and cleaved caspase-3 activation using MSD assays. [1] |
| ADME/Pharmacokinetics |
- Physicochemical Properties: Compound 20 has a measured LogD at pH 7.4 of 2.1. It shows good solubility (182 μM) and high passive permeability (Caco-2 Papp: 9.1 × 10⁻⁶ cm/s). [1] - In Vitro Metabolic Stability: 20 shows moderate intrinsic clearance in rat hepatocytes (24 μL/min/10⁶ cells) and low intrinsic clearance in human liver microsomes (<7 μL/min/mg). In mouse hepatocytes, the intrinsic clearance was 26 μL/min/10⁶ cells. [1] - In Vivo Pharmacokinetics in Mice (iv 0.5 mg/kg, po 1 mg/kg): Following iv administration, compound 20 had an observed plasma clearance (CLobs) of 41 mL/min/kg, a volume of distribution at steady state (Vss) of 2.1 L/kg, and a terminal half-life (T1/2) of 1.8 hours. After oral administration, the bioavailability (F) was 55%, with a Cmax of 605 nM and an AUC of 270 nM·h. The mouse plasma protein binding was 5.9% free. [1] - In Vivo Pharmacokinetics in Mice (po 30 mg/kg): Following a single oral dose of 30 mg/kg in SCID mice, the plasma concentration of 20 at the 6-hour time point was approximately 150 nM, suggesting linear PK scaling. [1] |
| Toxicity/Toxicokinetics |
- Cellular Selectivity/Toxicity: In the BFL1-null Karpas-422 cell line, compound 20 did not affect cell viability, suggesting a minimal off-target cellular phenotype. [1] - Off-Target Pharmacology: In a broad panel of 24 molecular targets, 20 showed significant binding (IC50 <1 μM) only to the CB1 receptor, but it did not exhibit functional activity (IC50 >100 μM). In a panel of 48 kinases, no significant inhibition (>50%) was observed at 1 μM. [1] |
| References |
[1]. Structure-Based Discovery of a Series of Covalent, Orally Bioavailable, and Selective BFL1 Inhibitors. J Med Chem. 2024 Dec 26;67(24):22055-22079. |
| Additional Infomation |
- Background and Rationale: BFL1 is an understudied anti-apoptotic member of the BCL2 family, overexpressed in various hematological malignancies. Targeting BFL1 is challenging due to the shallow protein-protein interaction (PPI) interface. The unique cysteine residue Cys55 in BFL1's BH3 groove was targeted for covalent inhibition. Compound 20 was developed through structure-based optimization from a covalent fragment hit (compound 6) to achieve improved potency, selectivity, and oral bioavailability. [1] - Mechanism of Action (Cell Death): In SUDHL1 and OCILY10 cell lines, treatment with 20 (in combination with an MCL1 inhibitor in codependent lines) induced a cell death phenotype, evidenced by caspase 3 activation. In vivo, this was confirmed by the dose-dependent activation of cleaved caspase-3 in OCILY10 tumor xenografts. [1] - Protein Stabilization: 20 was shown to bind to BFL1 with high occupancy, stabilizing the protein and extending its half-life from 2.6 hours to 10.8 hours. This stabilization effect served as a marker for target engagement both in vitro and in vivo. [1] - Lead Selection: Based on its balanced profile of potent cellular activity (EC50 = 0.35 μM in caspase assay), moderate in vitro intrinsic clearance, high permeability, good solubility, and favorable oral bioavailability (55%) in mice, compound 20 was selected as an in vitro and in vivo tool compound for further studies. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4821 mL | 12.4103 mL | 24.8207 mL | |
| 5 mM | 0.4964 mL | 2.4821 mL | 4.9641 mL | |
| 10 mM | 0.2482 mL | 1.2410 mL | 2.4821 mL |