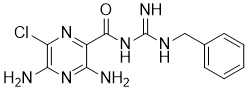
Benzamil is an Amiloride analogue acting as a Na+/Ca2+ exchanger (NCX) inhibitor (IC50~100 nM) with the potential to treat cystic fibrosis.. Benzamil is also a non-selective Deg/epithelial sodium channels (ENaC) blocker, and can potentiate myogenic vasoconstriction. Benzamil inhibits TRPP3-mediated Ca2+-activated currents, with an IC50 of 1.1 μM.
Physicochemical Properties
| Molecular Formula | C13H14N7OCL |
| Molecular Weight | 319.74956 |
| Exact Mass | 355.072 |
| CAS # | 2898-76-2 |
| Related CAS # | Benzamil hydrochloride;161804-20-2 |
| PubChem CID | 108107 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.594 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 22 |
| Complexity | 413 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | N=C(NC(C1=NC(Cl)=C(N=C1N)N)=O)NCC2=CC=CC=C2 |
| InChi Key | KXDROGADUISDGY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C13H14ClN7O/c14-9-11(16)20-10(15)8(19-9)12(22)21-13(17)18-6-7-4-2-1-3-5-7/h1-5H,6H2,(H4,15,16,20)(H3,17,18,21,22) |
| Chemical Name | 3,5-diamino-N-(N'-benzylcarbamimidoyl)-6-chloropyrazine-2-carboxamide |
| Synonyms | Benzamil; 2898-76-2; Benzylamiloride; 3,5-Diamino-N-(N-benzylcarbamimidoyl)-6-chloropyrazine-2-carboxamide; N-(N-Benzylamidino)-3,5-diamino-6-chloropyrazine carboxamide; UNII-04659UUJ94; CHEBI:34558; 3,5-diamino-N-(N'-benzylcarbamimidoyl)-6-chloropyrazine-2-carboxamide; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Na+/Ca2+ exchanger (NCX) (IC50 = 100 nM) |
| ln Vitro | Benzamil (Benzylamiloride) blocks Ca2+-activated small conductance K2+ channels in neurons and heterologously produced cells [4]. |
| ln Vivo | Stroke-prone spontaneously hypertensive rats (SHRSP) treated with benaamil (benzylamiloride) (0.7 mg/kg/day; sc) survived, on average, until 16.1 weeks of age[5]. |
| Enzyme Assay |
Small conductance Ca2+-activated K+ (SK) channels are expressed throughout the soma and dendrites of pyramidal neurons in the neocortex and hippocampal formation, where they participate in the local regulation of membrane excitability and synaptic signals. Through their inter-play with Ca2+ channels, SK channels regulate Ca2+ influx triggered by back-propagating action potentials in dendrites. Inhibition of SK channels affects both the amplitude and duration of Ca2+ transients, but the role of Ca2+ clearance mechanisms and their link to SK channel activity has not been established. Here we report the effect of the Na+/Ca2+ exchanger (NCX) inhibitor benzamil on Ca2+ extrusion and SK channels in the regulation of dendritic Ca2+ signals. Benzamil increased the duration and amplitude of dendritic Ca2+ transients elicited by back-propagating action potentials in hippocampal pyramidal neurons. This data is consistent with previous studies with SK channel blockers and suggests that benzamil inhibits SK channels in addition to the Na+/Ca2+ exchanger. Here we show that indeed both the neuronal SK-mediated IAHP current and the currents mediated by heterologously expressed SK channels were inhibited by benzamil. The inhibition of recombinant SK channels was seen with different K+ concentration gradients, and was stronger at negative voltages. The suppression of SK channels by benzamil is consistent with previous findings on the modulation of Ca2+ signals by SK channels in neurons. We additionally show that benzamil inhibits neuronal voltage-gated calcium currents. The results prompt a careful reassessment of the effects of benzamil on Ca2+ transients in native systems, given the spectrum of ion channels and exchangers this compound targets within a similar range of concentrations.[4]
In this study, researchers found that amiloride and its analogs inhibit TRPP3 channel activities with different affinities. Radiolabeled (45)Ca2+ uptake showed that TRPP3-mediated Ca2+ transport was inhibited by amiloride, phenamil, Benzamil, and 5-(N-ethyl-N-isopropyl)amiloride (EIPA). Two-microelectrode voltage clamp experiments revealed that TRPP3-mediated Ca2+-activated currents are substantially inhibited by amiloride analogs, in an order of potency of phenamil > Benzamil > EIPA > amiloride, with IC50 values of 0.14, 1.1, 10.5, and 143 microM, respectively. The inhibition potency positively correlated with the size of inhibitors. Using cell-attached patch clamping, we showed that the amiloride analogs decrease the open probability and mean open time but have no effect on single-channel conductance. Study of inhibition by phenamil in the presence of previously reported inhibitor tetrapentylammonium indicates that amiloride and organic cation inhibitors compete for binding the same site on TRPP3. TRPP3 may contribute to previously reported in vivo amiloride-sensitive cation transport[3]. |
| Cell Assay | Benzamil, an inhibitor of ENaC that also blocks Na+/Ca2+ exchange (NCX), potentiated myogenic vasoconstriction. Benzamil and low [Na+]o elicited vasoconstriction; however, these responses were attenuated by diltiazem and were associated with significant membrane depolarization, suggesting a contribution of mechanisms other than a reduction in NCX. Na+ repletion induced a vasodilation in pressurized afferent arterioles preequilibrated in low [Na+]o, a hallmark of NCX, and this response was reduced by 10 micromol/l benzamil. The dilation was eliminated, however, by a combination of benzamil plus ouabain, suggesting an involvement of the electrogenic Na+-K+-ATPase. In concert, these findings refute the premise that ENaC plays a significant role in the rat afferent arteriole and instead suggest that reducing [Na+](o) and/or Na+ entry is coupled to membrane depolarization. The mechanisms underlying these unexpected and paradoxical effects of Na+ are not resolved at the present time[2]. |
| Animal Protocol | Kidneys were allowed to equilibrate for at least 1 h following the establishment of in vitro perfusion. Ibuprofen (10 μmol/l) was added to eliminate the effects of endogenous prostaglandins. Basal renal perfusion pressure was held at 80 mmHg during the equilibration period. In some experiments, myogenic responses were evoked by raising renal arterial pressure and holding pressures at each step for at least 1 min. In these studies, stepped responses were assessed before and after the administration of amiloride or Benzamil. In other experiments, perfusion pressure was maintained at a constant level (60, 80, or 140 mmHg) to assess vasoconstrictor and/or vasodilatory responses. In the studies assessing the impact of low [Na+]o media, the media Na+ concentration ([Na+]) was lowered from 140 to 100 mmol/l by the isosmotic substitution of choline chloride for NaCl. Benzamil, amiloride, and ouabain were obtained commercially. Fresh stock solutions of Benzamil and amiloride were prepared in dimethyl sulfoxide. Ouabain (3 mmol/l in DMEM) was prepared fresh for each experiment. Phentolamine and propranolol (10 μmol/l; Sigma) were added during the ouabain experiments to avoid any effects mediated by neurotransmitter release.[2] |
| References |
[1]. Characterization of a Na(+)-Ca(2+) exchanger in podocytes. Nephrol Dial Transplant. 2002 Oct;17(10):1742-50. [2]. Effects of amiloride, benzamil, and alterations in extracellular Na+ on the rat afferent arteriole and its myogenic response. Am J Physiol Renal Physiol. 2008 Jul;295(1):F272-82. [3]. Inhibition of TRPP3 channel by amiloride and analogs. Mol Pharmacol. 2007 Dec;72(6):1576-85. [4]. Benzamil inhibits neuronal and heterologously expressed small conductance Ca2+-activated K+channels. Neuropharmacology. 2019 Nov 1;158:107738. [5]. Epithelial sodium channel inhibition in cardiovascular disease. A potential role for amiloride. Am J Hypertens. 2007 Jan;20(1):109-17. |
| Additional Infomation | Benzamil is a member of pyrazines and a member of guanidines. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1274 mL | 15.6372 mL | 31.2744 mL | |
| 5 mM | 0.6255 mL | 3.1274 mL | 6.2549 mL | |
| 10 mM | 0.3127 mL | 1.5637 mL | 3.1274 mL |