Physicochemical Properties
| Molecular Formula | C25H37N3O5 |
| Molecular Weight | 459.59 |
| Exact Mass | 459.273 |
| Elemental Analysis | C, 65.34; H, 8.12; N, 9.14; O, 17.41 |
| CAS # | 118675-50-6 |
| PubChem CID | 122021 |
| Appearance | Colorless to light yellow liquids |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 638.1±65.0 °C at 760 mmHg |
| Flash Point | 339.7±34.3 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.593 |
| LogP | 3.31 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 33 |
| Complexity | 646 |
| Defined Atom Stereocenter Count | 0 |
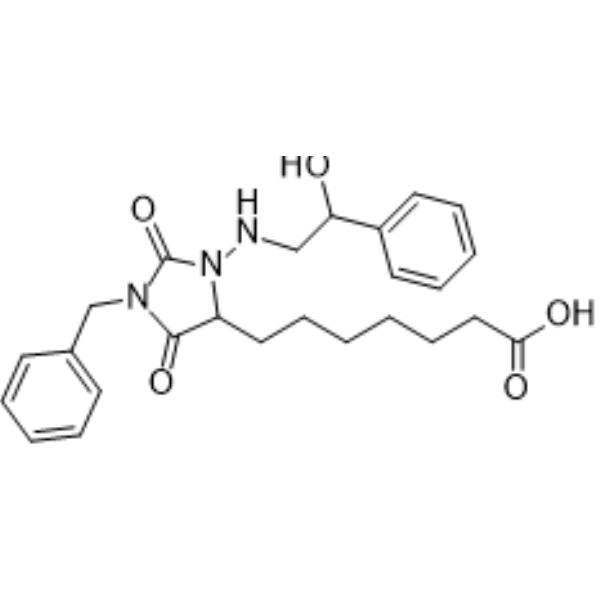
| SMILES | C(CCCC(=O)O)CCC1C(=O)N(CC2=CC=CC=C2)C(=O)N1NCC(C3CCCCC3)O |
| InChi Key | YZJVWSKJHGEIBL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C25H37N3O5/c29-22(20-13-7-4-8-14-20)17-26-28-21(15-9-1-2-10-16-23(30)31)24(32)27(25(28)33)18-19-11-5-3-6-12-19/h3,5-6,11-12,20-22,26,29H,1-2,4,7-10,13-18H2,(H,30,31) |
| Chemical Name | 7-[1-benzyl-3-[(2-cyclohexyl-2-hydroxyethyl)amino]-2,5-dioxoimidazolidin-4-yl]heptanoic acid |
| Synonyms | BWA 868C; BWA-868C; BW A868C; 118675-50-6; BWA868C; BW-A868C; 3-Benzyl-5-(6-carboxyhexyl)-1-(2-cyclohexyl-2-hydroxyethylamino)hydantoin; 4-Imidazolidineheptanoicacid, 3-[(2-cyclohexyl-2-hydroxyethyl)amino]-2,5-dioxo-1-(phenylmethyl)-; 7-[1-benzyl-3-[(2-cyclohexyl-2-hydroxyethyl)amino]-2,5-dioxoimidazolidin-4-yl]heptanoic acid; (+/-)-3-benzyl-5-(6-carboxyhexyl)-1-(2-cyclohexyl-2-hydroxyethylamino)hydantoin; BWA868C |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Prostaglandin D2 (PGD2) receptor |
| ln Vitro | Human platelet adenylyl cyclase is activated by prostaglandin D2 and BW 245C, which are both antagonistic to BW A868C [1]. |
| Enzyme Assay | 1. BW A868C, a novel compound, behaved as a simple competitive antagonist in a human washed platelet aggregation assay. Anti-aggregatory concentration-effect curves to BW 245C were displaced in a parallel manner. The shifts accorded with a Schild plot slope of unity and a pKB of 9.26. 2. Inhibition of platelet aggregation by prostaglandin D2 (PGD2) was antagonized with a similar potency, as were the relaxation effects of BW 245C and PGD2 in the rabbit jugular vein. BW A868C can, therefore, be classified as a DP-receptor antagonist. 3. Actions of BW A868C at other prostaglandin receptors (IP, EP1, EP2, TP and FP) were excluded at concentrations up to 1,000 times higher than the DP-receptor affinity. 4. Analyses of BW 245C- and PGD2-mediated effects were complicated by additional agonist receptor interactions which were revealed by BW A868C. In rabbit jugular vein a resistant phase of agonism was detectable, indicating that both agonists exerted effects through another receptor (possibly EP2). Also, PGD2, in addition to its anti-aggregatory effect on platelets, demonstrated a pro-aggregatory action in the presence of BW A868C. 5. The contractile effects of PGD2 in guinea-pig tracheal strips were resistant to 10 microM BW A868C indicating that they were not mediated through DP-receptors. 6. To our knowledge this is the first account of a well-classified competitive antagonist at the DP-receptor. Its potency and selectivity make it an important new tool in prostanoid receptor classification and identification[1]. |
| References |
[1]. The classification of prostaglandin DP-receptors in platelets and vasculature using BW A868C, a novel, selective and potent competitive antagonist. Br J Pharmacol. 1989 Feb;96(2):291-300. |
| Additional Infomation | BW A868C is an imidazolidine-2,4-dione. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1759 mL | 10.8793 mL | 21.7585 mL | |
| 5 mM | 0.4352 mL | 2.1759 mL | 4.3517 mL | |
| 10 mM | 0.2176 mL | 1.0879 mL | 2.1759 mL |