Physicochemical Properties
| Molecular Formula | C19H32N2O5 |
| Molecular Weight | 368.474 |
| Exact Mass | 368.231 |
| Elemental Analysis | C, 61.93; H, 8.75; N, 7.60; O, 21.71 |
| CAS # | 72814-32-5 |
| Related CAS # | (8-epi)-BW 245C;65705-83-1 |
| PubChem CID | 119304 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Index of Refraction | 1.521 |
| LogP | 2.12 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 26 |
| Complexity | 490 |
| Defined Atom Stereocenter Count | 0 |
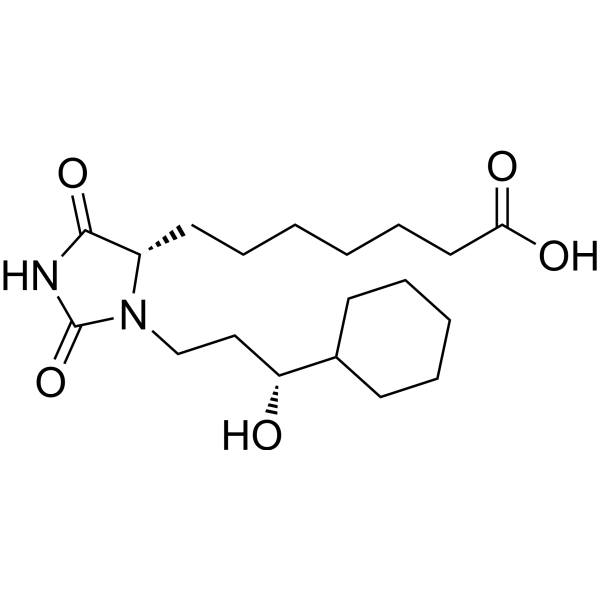
| SMILES | O([H])C([H])(C([H])([H])C([H])([H])N1C(N([H])C(C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C(=O)O[H])=O)=O)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] |
| InChi Key | ZIDQIOZJEJFMOH-JKSUJKDBSA-N |
| InChi Code | InChI=1S/C19H32N2O5/c22-16(14-8-4-3-5-9-14)12-13-21-15(18(25)20-19(21)26)10-6-1-2-7-11-17(23)24/h14-16,22H,1-13H2,(H,23,24)(H,20,25,26)/t15-,16+/m0/s1 |
| Chemical Name | 7-[(4S)-3-[(3R)-3-cyclohexyl-3-hydroxypropyl]-2,5-dioxoimidazolidin-4-yl]heptanoic acid |
| Synonyms | BW-245C; BW 245C; BW 245C; 72814-32-5; BW245C; 72880-75-2; (R-(R*,S*))-3-(3-Cyclohexyl-3-hydroxypropyl)-2,5-dioxoimidazolidine-4-heptanoic acid; 5B26TJM1L8; (3R,4S)-BW 245C; 4-Imidazolidineheptanoic acid, 3-((3R)-3-cyclohexyl-3-hydroxypropyl)-2,5-dioxo-, (4S)-rel-; BW245C |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | DP1/prostanoid DP-receptor |
| ln Vitro | In Th2 cells, BW245C (0.01-1 μM) suppresses TGF-β collagen in a dose-dependent manner. Lung fibroblasts’ intracellular cAMP is likewise increased by BW245C (0.01-1 μM) [3]. The levels needed to raise transendothelial resistance and reduce FITC-dextran permeability in human venous endothelial cells are 0.1-3 μMol/L. Intracellular cAMP levels and necessary protein structure A (PKA) activity are increased by BW245C (0.3 μMol/L) [4]. |
| ln Vivo | In WT mice, BW245C (0.02, 0.2, and 2.0 mg/kg) significantly increased CBF; however, what was the impact of this therapy on DP1? Not present in mice. Following a stroke, functional deficits are lessened by BW245C. After a stroke in WT mice, BW245C greatly reduced the severe decline in locomotor activity and the significant rise in neurological impairments. After receiving a BW245C injection (0.2 mg/kg) one hour post-stroke, WT showed a substantial reduction in cerebral infarction; however, at DP1? //? In mice, this therapy had no discernible effects. CBF is enhanced during and after a stroke by BW245C. When BW245C was administered, the coagulation time was significantly longer than when the vehicle-treated group was given [1]. In guinea pig esophageal epithelial cells, OVA-induced MBP acidification is not substantially increased by BW 245C (100 nM) [2]. |
| Cell Assay | Isolated eosinophils (5 × 105/ml) were kept in RPMI supplemented with 1% FBS and PenStrep and stimulated with 1 μM DK‐PGD2 or BW245c for 18 h at 37°C. Cells were stained with mouse anti‐human CD193 (CCR3)–BV421 and mouse anti‐human CD49d (VLA‐4)‐PE, or the respective isotype controls. Human TruStain FcX (BioLegend) was used as Fc receptor blocking solution. Data were acquired in a BD FACS Canto II[5]. |
| Animal Protocol | To determine if BW245C affects basal cerebral blood flow (CBF), C57BL/6 WT and DP1(-/-) mice were given a single i.p. injection of vehicle or BW245C, and CBF was recorded for 2h. To test the effect of BW245C on stroke, WT and DP1(-/-) mice were subjected to middle cerebral artery occlusion followed by a single i.p. injection of vehicle or 0.02, 0.2, or 2.0-mg/kg BW245C immediately before reperfusion. Functional and anatomical outcomes were determined at 96h. We also determined the effect of BW245C on CBF in peri-infarct and core during occlusion and reperfusion. Furthermore, we tested the effect of BW245C on bleeding time and ex vivo coagulation. BW245C treatment increased the basal CBF significantly in WT but not in DP1(-/-) mice. The BW245C treatment also significantly improved functional outcome and lowered infarction volume. The multisite CBF monitoring by laser-Doppler flowmetry shows that BW245C significantly increased the CBF in peri-infarct, with a significant inverse correlation between infarction and CBF. The significantly higher infarction volume in DP1(-/-) mice remained unchanged with BW245C treatment. Moreover, BW245C preserves hemostasis in non-stroke conditions. Combined, these data suggest that the DP1 receptor is an endogenous target that can rescue the brain following stroke by regulating CBF and hemostasis[1]. |
| References |
[1]. Ahmad AS, et al. PGD2 DP1 receptor stimulation following stroke ameliorates cerebral blood flow and outcomes. Neuroscience. 2014 Oct 24;279:260-8. [2]. Zhang S, et al. Prostaglandin D2 receptor D-type prostanoid receptor 2 mediates eosinophil trafficking into the esophagus. Dis Esophagus. 2014 Aug;27(6):601-6. [3]. Ayabe S, et al. Prostaglandin D2 inhibits collagen secretion from lung fibroblasts by activating the DP receptor. J Pharmacol Sci. 2013;121(4):312-7. Epub 2013 Mar 29. [4]. Kobayashi K, et al. Prostaglandin D2-DP signaling promotes endothelial barrier function via the cAMP/PKA/Tiam1/Rac1 pathway. Arterioscler Thromb Vasc Biol. 2013 Mar;33(3):565-71. [5]. DP1 receptor signaling prevents the onset of intrinsic apoptosis in eosinophils and functions as a transcriptional modulator. J Leukoc Biol. 2018 Jul; 104(1): 159–171. |
| Additional Infomation | 7-[3-(3-cyclohexyl-3-hydroxypropyl)-2,5-dioxoimidazolidin-4-yl]heptanoic acid is a imidazolidine-2,4-dione that is 7-(2,5-dioxoimidazolidin-4-yl)heptanoic acid in which the imidazoline ring as substituted at position 3 by a 3-(3-cyclohexyl-3-hydroxypropyl) group. It is an imidazolidine-2,4-dione, a secondary alcohol and a monocarboxylic acid. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~135.70 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7139 mL | 13.5696 mL | 27.1393 mL | |
| 5 mM | 0.5428 mL | 2.7139 mL | 5.4279 mL | |
| 10 mM | 0.2714 mL | 1.3570 mL | 2.7139 mL |