Physicochemical Properties
| Molecular Formula | C12H12CLN3 |
| Molecular Weight | 233.699 |
| Exact Mass | 233.072 |
| Elemental Analysis | C, 61.67; H, 5.18; Cl, 15.17; N, 17.98 |
| CAS # | 205437-64-5 |
| Related CAS # | 205437-64-5 (HCl);187173-05-3; |
| PubChem CID | 11957470 |
| Appearance | Brown to gray solid powder |
| LogP | 2.151 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 16 |
| Complexity | 262 |
| Defined Atom Stereocenter Count | 0 |
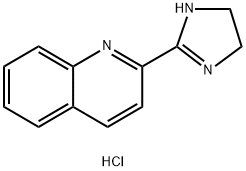
| SMILES | C1CN=C(N1)C2=NC3=CC=CC=C3C=C2.Cl |
| InChi Key | DDFHQXAQWZWRSQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C12H11N3.ClH/c1-2-4-10-9(3-1)5-6-11(15-10)12-13-7-8-14-12;/h1-6H,7-8H2,(H,13,14);1H |
| Chemical Name | 2-(4,5-dihydro-1H-imidazol-2-yl)quinoline;hydrochloride |
| Synonyms | BU-224 HCl; BU-224; BU 224; BU224; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
In mouse neuroblastoma N2a cells stably transfected with APP Swedish mutation (N2asw), incubation with BU-224 at concentrations of 10, 50, and 100 µM for 24 or 48 hours did not change the levels of secreted amyloid-β (Aβ) or sAPPα in the conditioned media, nor the levels of full-length APP or its C-terminal fragments (CTFs) in cell lysates.[1] In primary rat astrocyte cultures, treatment with BU-224 at 1, 3, and 10 µM for 48 hours led to a concentration-dependent increase (up to 30%) in the number of GFAP-positive astrocytes, suggesting a potential to promote astrocyte proliferation.[1] In calcium imaging experiments using N2a cells, pre-treatment with synthetic Aβ oligomers (2 µM) significantly increased the peak amplitude of intracellular calcium influx in response to NMDA (50 µM). Co-incubation with 5 µM BU-224 reversed this Aβ-induced increase in NMDA receptor-mediated calcium signaling.[1] In organotypic brain slice cultures from Thy-1-GFP/5XFAD double transgenic mice, treatment with 10 µM BU-224 for 24 hours increased the size of dendritic spines compared to vehicle-treated slices, although it did not change the reduced spine density present in the transgenic slices.[1] |
| ln Vivo |
BU224 hydrochloride (5 mg/kg, intraperitoneal injection, twice daily for 10 days) can improve behavioral performance and memory function in 5XFAD mice [1]. BU224 hydrochloride (5 mg/kg, IP, twice daily for 10 days) decreases the levels of the microglia marker Iba1 and the proinflammatory cytokines IL-1β and TNF-α in 5XFAD animals and enhances the Expression of glial cell marker GFAP[1]. BU224 hydrochloride (10 mg/kg, intraperitoneal injection, once) lowers the immobility of rats in the FST (forced swim test), demonstrating antidepressant-like effects [2]. Sub-chronic treatment (5 mg/kg, i.p., twice daily for 10 days) with BU-224 in 6-month-old female 5XFAD mice (a model of Alzheimer's disease amyloidosis) reversed spatial and recognition memory deficits. This was demonstrated by improved performance in the object location test (OLT) and novel object recognition (NOR) test, bringing performance to levels similar to wild-type controls.[1] In the same model, BU-224 treatment improved associative learning and fear memory, as assessed by trace fear-conditioning tests, showing increased freezing behavior during training, contextual (hippocampal-dependent), and cued (amygdala- and hippocampal-dependent) memory tests compared to vehicle-treated transgenic mice. No such effects were observed in treated wild-type mice.[1] BU-224 treatment did not alter amyloid-β plaque burden or levels of Aβ40 and Aβ42 in the cortex and hippocampus of 5XFAD mice, as assessed by immunohistochemistry, thioflavin S staining, ELISA, and Western blot.[1] The treatment significantly increased the area covered by astrocytes, as indicated by elevated levels of the astrocytic markers glial fibrillary acidic protein (GFAP) and glutamine synthase (GS) in the cortex and hippocampus of 5XFAD mice.[1] BU-224 treatment reduced microgliosis, shown by a significant decrease in the expression of the microglial marker Iba1 in the hippocampus of 5XFAD mice.[1] The treatment exerted anti-inflammatory effects, reducing protein levels of IL-1β (in cortex and hippocampus) and mRNA levels of TNF-α (in frontal cortex) in 5XFAD mouse brains. Levels of IL-6 and MCP-1 were unaffected.[1] BU-224 treatment did not affect neuronal density (NeuN-positive cells) in cortical layer V, dentate gyrus, or subiculum of 6-month-old 5XFAD mice, nor did it alter the expression of the apoptosis-related marker Fas-R, the mitochondrial marker COX-4, or markers of oxidative stress (8-OHdG/8-OHG) in these mice.[1] The treatment did not significantly change the total expression levels of synaptic markers synaptophysin and PSD-95 in cortical and hippocampal homogenates, although a non-significant increasing trend in synaptophysin immunofluorescence was noted in the hippocampus.[1] |
| Cell Assay |
N2a cell treatment for APP/Aβ analysis: Mouse neuroblastoma N2a cells stably expressing APP with the Swedish mutation (N2asw) were cultured. Cells were seeded and, 24 hours later, treated with BU-224 at concentrations of 10, 50, and 100 µM (diluted in DMSO) or vehicle (DMSO) in OptiMEM medium for 24 or 48 hours. Conditioned media were collected for analysis of secreted Aβ and sAPPα by ELISA or Western blot. Cell lysates were prepared for analysis of full-length APP and C-terminal fragments by Western blot.[1] Primary astrocyte culture and proliferation assay: Primary astrocyte cultures were prepared from cortices of 1-day-old Sprague-Dawley rats. Cells were cultured and maintained. After a medium change at 7 days in vitro, cells were treated with BU-224 at 1, 3, and 10 µM (in DMSO) or DMSO vehicle for 48 hours. Cells were then fixed and subjected to immunocytochemistry for the astrocyte marker GFAP and counterstained with DAPI. The number of GFAP-positive cells was counted using imaging software.[1] Calcium imaging in N2a cells: N2a cells were seeded on coated coverslips and treated overnight with 2 µM oligomeric Aβ and/or 5 µM BU-224. Cells were then loaded with the calcium indicator fura-2 AM and probenecid in buffer for 30 minutes at 37°C. Coverslips were placed in a perfusion chamber. After establishing baseline fluorescence, cells were stimulated with 50 µM NMDA dissolved in buffer. Fluorescence images were captured with alternating excitation wavelengths. The fluorescence intensity ratio (F355/F380) was calculated over time to reflect changes in intracellular calcium concentration.[1] |
| Animal Protocol |
Animal/Disease Models: 5XFAD mice and WT littermates (6 months old) [1] Doses: 0 mg/kg, 5 mg/kg Route of Administration: IP twice (two times) daily for 10 days Experimental Results: Significant increase immobility (54% increase in freezing) and reversed memory deficits secondary to Aβ deposition in the brain. Six-month-old female 5XFAD transgenic mice and age-matched wild-type (C57Bl/6) control mice were used. BU-224 was dissolved in saline (0.9% sodium chloride). Mice received intraperitoneal (i.p.) injections of BU-224 at a dose of 5 mg/kg body weight, or an equivalent volume of saline as vehicle control. Injections were administered twice per day (approximately every 12 hours) for a total duration of 10 days. Behavioral tests (open field, object location test, novel object recognition, fear conditioning) were conducted following the treatment period. Mice were transcardially perfused with ice-cold PBS under terminal anesthesia after behavioral assessment. Brains were collected, with one hemisphere fixed for histology and the other frozen for biochemical analysis.[1] |
| Toxicity/Toxicokinetics |
No adverse effects on body weight or general well-being were observed in mice treated with BU-224 (5 mg/kg, i.p., twice daily for 10 days) during the study.[1] BU-224 did not cause overt cytotoxicity in primary human cortical neurons at concentrations up to 1 µM over a seven-day treatment in a separate cited context (within the discussion, referencing the lack of cytotoxicity in human neurons for other compounds). This specific experiment on BU-224 in human neurons is not detailed in the methodology/results of this paper.[1] |
| References |
[1]. Imidazoline ligand BU224 reverses cognitive deficits, reduces microgliosis and enhances synaptic connectivity in a mouse model of Alzheimer's disease. Br J Pharmacol. 2021 Feb;178(3):654-671. [2]. Behavioral, neuroendocrine and neurochemical effects of the imidazoline I2 receptor selective ligand BU224 in naive rats and rats exposed to the stress of the forced swim test. Psychopharmacology (Berl). 2003 May;167(2):195-202. [3]. Discriminative stimulus effects of the imidazoline I2 receptor ligands BU224 and phenyzoline in rats. Eur J Pharmacol. 2015 Feb 15;749:133-41. |
| Additional Infomation |
See also: BU 224 (annotation moved to). BU-224 (2-(4,5-Dihydroimidazol-2-yl)quinoline hydrochloride) is a synthetic, high-affinity and highly selective ligand for the imidazoline I2 receptor subtype.[1] I2 receptors are abundantly expressed in the brain, particularly on the outer mitochondrial membrane of astrocytes.[1] The study suggests that the cognitive benefits of BU-224 in the 5XFAD mouse model are not due to modulation of amyloid pathology but likely result from its anti-inflammatory effects (reducing microgliosis and pro-inflammatory cytokines), promoting astrocyte activation, and potentially protecting synaptic function (increasing spine size, modulating NMDA receptor activity).[1] The findings propose that targeting I2 receptors with ligands like BU-224 could represent a therapeutic strategy for Alzheimer's disease, particularly at stages where significant pathology and inflammation are present, by targeting pathways secondary to Aβ deposition.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~12.5 mg/mL (~53.49 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (5.35 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (5.35 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.2790 mL | 21.3950 mL | 42.7899 mL | |
| 5 mM | 0.8558 mL | 4.2790 mL | 8.5580 mL | |
| 10 mM | 0.4279 mL | 2.1395 mL | 4.2790 mL |