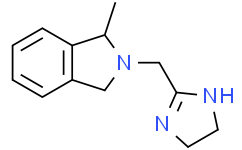
Physicochemical Properties
| Molecular Formula | C13H17N3 |
| Molecular Weight | 215.29418 |
| Exact Mass | 331.153 |
| CAS # | 118343-19-4 |
| Related CAS # | 681806-46-2; 118343-19-4 |
| PubChem CID | 121850 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.22g/cm3 |
| Boiling Point | 365.8ºC at 760mmHg |
| Flash Point | 175ºC |
| Vapour Pressure | 1.53E-05mmHg at 25°C |
| Index of Refraction | 1.657 |
| LogP | 0.979 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 16 |
| Complexity | 287 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC1C2=CC=CC=C2CN1CC3=NCCN3 |
| InChi Key | SGOFAUSEYBZKDQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C13H17N3/c1-10-12-5-3-2-4-11(12)8-16(10)9-13-14-6-7-15-13/h2-5,10H,6-9H2,1H3,(H,14,15) |
| Chemical Name | 2-(4,5-dihydro-1H-imidazol-2-ylmethyl)-1-methyl-1,3-dihydroisoindole |
| Synonyms | 118343-19-4; BRL 44408; Brl-44408; 2-((4,5-Dihydro-1H-imidazol-2-yl)methyl)-2,3-dihydro-1-methyl-1H-isoindole; BRL44408; UNII-ZET7B198W2; 2-(4,5-dihydro-1H-imidazol-2-ylmethyl)-1-methyl-1,3-dihydroisoindole; ZET7B198W2; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | α2A-adrenoceptor antagonist (Ki = 8.5 nM) |
| ln Vitro | BRL 44408 is a potent (Ki=8.5 nM) and selective (>50-fold) alpha2A-adrenoceptor antagonist (K(B)=7.9 nM)[1]. |
| ln Vivo |
In rats, BRL 44408 penetrated the central nervous system resulting in peak brain and plasma concentrations of 586 ng/g and 1124 ng/ml, respectively. In a pharmacodynamic assay, pretreatment with BRL 44408 to rats responding under a fixed-ratio 30 operant response paradigm resulted in a rightward shift of the clonidine dose-response curve, an effect indicative of alpha2-adrenoceptor antagonism in vivo. Consistent with presynaptic autoreceptor antagonism and tonic regulation of neurotransmitter release, acute administration of BRL 44408 elevated extracellular concentrations of norepinephrine and dopamine, but not serotonin, in the medial prefrontal cortex. Additionally, BRL 44408, probably by inhibiting alpha2A heteroceptors, produced a significant increase in cortical levels of acetylcholine. In the forced swim test and schedule-induced polydipsia assay, BRL 44408 produced an antidepressant-like response by dose-dependently decreasing immobility time and adjunctive water intake, respectively, while in a model of visceral pain, BRL 44408 exhibited analgesic activity by decreasing para-phenylquinone (PPQ)-induced abdominal stretching. Finally, BRL 44408 did not produce deficits in overall motor coordination nor alter general locomotor activity. This preclinical characterization of the neurochemical and behavioural profile of BRL 44408 suggests that selective antagonism of alpha2A-adrenoceptors may represent an effective treatment strategy for mood disorders and visceral pain.[1] Acute respiratory distress syndrome (ARDS), characterized by acute hypoxic respiratory dysfunction or failure, is a manifestation of multiple organ failure in the lung, and the most common risk factor is sepsis. Researchers previously showed that blocking α2 -adrenoceptor (α2 -AR) could attenuate lung injury induced by endotoxin in rats. α2A -adrenoceptor (α2A -AR), a subtype of α2 -AR plays a key role in inflammatory diseases, but the mechanism remains unknown. Here, they explored the effect of BRL 44408 maleate (BRL), a specific α2A -AR antagonist, on cecal ligation puncture (CLP)-induced ARDS in rats and the underlying mechanism. Preadministration of BRL-44408 maleate significantly alleviated CLP-induced histological injury, macrophage infiltration, inflammatory response, and wet/dry ratio in lung tissue. However, there was no statistical difference in survival rate between the CLP and CLP+BRL groups. Extracellular regulated protein kinase (ERK1/2), p38MAPK, and p65 were activated in the CLP group, and BRL-44408 maleate inhibited the activation of these signal molecules, c-Jun N-terminal kinase (JNK) and protein kinase A (PKA) showed no changes in activation between these two groups. BRL-44408 maleate decreased lipopolysaccharide (LPS)-induced expression of cytokines in NR8383 rat alveolar macrophages and reduced phosphorylation of ERK1/2, p38MAPK, and p65. JNK and PKA were not influenced by LPS. Together, these findings suggest that antagonism of α2A -AR improves CLP-induced acute lung injury and involves the downregulation of ERK1/2, p38MAPK, and p65 pathway independent of the activation of JNK and PKA[2]. |
| Enzyme Assay |
Radioligand binding and cAMP accumulation assay[1] Membranes for radioligand binding studies were prepared from cultured Chinese Hamster Ovary (CHO) cells expressing the human α2A-adrenoceptor cDNA. Confluent monolayers were harvested, homogenized in 50 mM Tris (HCl), pH 7.4 buffer and centrifuged at 1000 g. The supernatant was then centrifuged at 35000 g and the resulting pellet was re-suspended with Tris buffer and frozen at −80°C. On the day of the assay, Tris buffer, BRL 44408, 1 nM [3H]MK 912 (S.A. 85 Ci/mmol), 2.5 µg thawed membrane protein and 0.5 mg PVT WGA SPA beads were combined together in each well of a 96-well microtitre Packard Optiplate. Regression analysis of % specific bound data from no fewer than 11 concentrations of BRL 44408 was performed using a four-parameter logistic curve fitting model in order to derive IC50 estimates. Subsequent Ki values were calculated by the Cheng–Prusoff equation (Cheng & Prusoff, 1973). |
| Animal Protocol |
Pharmacokinetic studies in the rat[1] The pharmacokinetics of BRL-44408 were characterized in fasted (overnight) male Sprague–Dawley rats (275–350 g) after a single subcutaneous (s.c.) dose of 10 mg/kg. Serial blood and brain samples were collected at intervals up to 6 h after BRL 44408 administration into tubes containing EDTA as the anticoagulant, and the content diluted 1:1 with de-ionized water. Samples were treated with protein precipitation by addition of acetonitrile containing an internal standard. The supernatants were transferred into clean test tubes and evaporated under a stream of N2 at 37°C followed by reconstitution in acetonitrile/de-ionized water [50:50 (v/v)] for LC–MS analysis. LC–MS analysis was performrd on Agilent 1100 HPLC interfaced with API4000. Separation was accomplished on a column of Water C-18 Xterra MS column (2.1×20 mm, 2.5 µm) with 10 mM ammonium acetate in water (A) and acetonitrile (B) at a flow rate of 400 µl/min in linear gradient manner from 5% B to 90% in 5 min. The mass spectrometer was operated in positive electrospray and multiple reaction monitoring (MRM) mode, respectively. Rat fixed-ratio responding (FR-30)[1] Adult male Sprague–Dawley rats were individually housed and restricted to ∼85% of their free-feeding body weights. Rats were trained to respond on the response lever under a FR-30 schedule of food presentation. Daily experimental sessions consisted of three components, with each component consisting of a 10-min timeout period followed by a 10-min response component. During the timeout period, the chamber was dark and there were no programmed consequences. During the response components, the house-light was illuminated and lever pressing was paired with an audible click. Completion of the ratio resulted in the presentation of the food pellet (45-mg dustless pellet). Incremental doses of clonidine (0.01–0.1 mg/kg) or the 5-HT1A agonist 8-OH-DPAT (0.1–1.0 mg/kg) were administered cumulatively [intraperitoneally (i.p.)] at the beginning of each timeout component. For antagonism studies, BRL-44408 (10 mg/kg i.p.) or idazoxan (3.0 mg/kg i.p.) were administered as a 30 min pretreatment prior to the cumulative clonidine dose–response curve. The ED50 value was defined as the dose of clonidine or 8-OH-DPAT that produced a 50% reduction in the response rate in the absence or presence of the antagonist. In-vivo microdialysis[1] Microdialysis techniques were performed as previously described (Beyer et al.2002). Male Sprague–Dawley rats (280–350 g, Charles River Laboratories, USA) were anaesthetized with isoflurane and a microdialysis guide cannula was positioned above the medial prefrontal cortex (mPFC; AP +3.2 mm; ML +0.5 mm; DV −1.8 mm relative to bregma and dura) using stereotaxic surgery. Following 24 h post-operative recovery from stereotaxic surgery, microdialysis probes (CMA/12; 20 kDa cut-off; CMA Microdialysis), with a 4-mm active membrane, were perfused with artificial CSF (aCSF; 125 mM NaCl, 3 mM KCl, 0.75 mM MgSO4, 1.2 mM CaCl2; pH 7.4) at 1 µl/min (or 0.5 µl/min for the ACh experiments) and inserted into the mPFC. Three hours after probe insertion, dialysates were collected in freely moving rats every 30 min (or 40 min for the ACh study). Four baseline samples were collected after which either 0.9% NaCl (saline) or BRL-44408 (10 mg/kg s.c.) was administered. Dialysates were collected in freely moving rats in the presence of drug for at least 3.5 h. Schedule-induced polydipsia (SIP)[1] As previously described (Platt et al.2008), male Sprague–Dawley rats (300–400 g; Charles River Laboratories) were used for the SIP studies. Experimental sessions (120 min) were performed in a ventilated, sound-attenuated chamber fitted with a food pellet dispensing magazine, a food trough and a water bottle. Test sessions occurred on Tuesdays and Fridays, with the remaining days serving as control days. During each session, one food pellet (BioServ 45-mg precision dustless pellet) was dispensed each min for a total of 120 pellets delivered. BRL-44408 (10–30 mg/kg i.p.) or H2O was administered 20 min prior to the start of the test session. At the end of the test session, the volume of water ingested was measured in milliliters. Water consumption (ml) was calculated and expressed as % control water intake and data were analysed using a one-way ANOVA with Dunnett's post-hoc test. Para-phenylquinone (PPQ) model[1] The ability of compounds to attenuate acute visceral (abdominal) pain was assessed in male CD-1 mice (20–25 g) according to previous methods (Siegmund et al.1957) following an i.p. injection of 2 mg/kg PPQ (dissolved in 4% ethanol in dH2O; Sigma-Aldrich). BRL-44408 (1–30 mg/kg i.p.) was administered 60 min prior to PPQ administration. Following PPQ administration, mice were individually placed in a Plexiglas cage, and the total number of abdominal constrictions was recorded for 1-min periods starting at 5 and 10 min after PPQ injection. Data were analysed using a one-way ANOVA with a Dunnett's post-hoc test and are represented as mean±S.E.M. % reversal of pain behaviour. Rotarod assay and locomotor activity[1] To examine the potential effects of BRL-44408 on motor performance, rats (male Sprague–Dawley rats, 125–150 g) were tested on an accelerating rotarod apparatus as previously described (Dunham & Miya, 1957). The rotarod was set to accelerate from 0 to 40 rpm over 300 s, with the maximum time spent on the rotarod set at 300 s. Rats received two training trials on the first day and then received a single dose of vehicle (0.9% NaCl), BRL-44408 (30 mg/kg s.c.) or morphine (10 mg/kg s.c). Data were analysed using a repeated-measures ANOVA and are represented as mean±S.E.M. latency (s) to fall off the rotarod. Evaluation of locomotor activity occurred 4 h after administration of BRL (3–30 mg/kg i.p.). Rats were placed in an open field (90×90 cm Plexiglas box with 30 cm walls) while locomotor activity was recorded for 5 min by a camera suspended over the open field so that the entire box was viewed and recorded. The rats were randomly divided into four groups: sham, BRL-44408/BRL, CLP, and CLP + BRL groups. Each group was divided into two subgroups (6 hr or 24 hr after CLP) and each subgroup included six to eight rats. The rats were anesthetized with 2% sodium pentobarbital in saline (40 mg/kg, intraperitoneally). CLP was performed as described previously (Otero-Antón et al., 2001). Briefly, a 2-cm midline abdominal incision was performed. The cecum was exposed, ligated just distal to the ileocecal valve to avoid intestinal obstruction, punctured once with an 18-gauge needle, squeezed slightly to allow a small amount of fecal matter to flow from the holes, and then returned the cecum to the abdominal cavity, the abdominal incision was then closed in layers. The rats were immediately resuscitated with normal saline 3 ml/100 g BW subcutaneously. The rats in the sham and BRL group underwent the general operation procedure without ligated or punctured of the cecum. The rats were returned to their cages and fed normally. At 5 hr after CLP, the rats in the BRL and CLP + BRL group received an intraperitoneal injection of 5 mg/kg BRL-44408 maleate in 1 ml of normal saline, while the rats in the sham group and CLP group received the same volume of normal saline intraperitoneally.[2] |
| ADME/Pharmacokinetics |
Pharmacokinetic properties of BRL 44408[1] The brain and plasma exposures of BRL 44408 were determined in male Sprague–Dawley rats. Following a single injection of 10 mg/kg (s.c.), this α2A-adrenoceptor antagonist was found to moderately cross the blood–brain barrier with peak brain and plasma concentrations of 586 ng/g and 1124 ng/ml, respectively [brain:plasma ratio=0.51 based on AUC0−inf 2344 (h per ng/g) in brain and 4637 (h per ng/ml) in plasma]. The peak levels in both compartments were measured at 2 h (Table 3). |
| References |
[1]. Preclinical characterization of BRL 44408: antidepressant- and analgesic-like activity through selective alpha2A-adrenoceptor antagonism. Int J Neuropsychopharmacol. 2010 Oct;13(9):1193-205. [2]. α2A -AR antagonism by BRL-44408 maleate attenuates acute lung injury in rats with downregulation of ERK1/2, p38MAPK, and p65 pathway. J Cell Physiol. 2020 Oct;235(10):6905-6914. |
| Additional Infomation |
In summary, the present series of experiments describe the receptor and behavioural pharmacology of the selective α2A-adrenoceptor antagonist, BRL 44408. We have shown that acute treatment with BRL 44408 can produce an array of neurochemical and behavioural effects in preclinical models, including antidepressant-like and analgesic activity. These data suggest that selective antagonism of α2A-adrenoceptors may represent a viable mechanism to consider for treating mood disorders and some types of pain conditions. Furthermore, based on the neurochemical effects on ACh, α2A-adrenoceptor antagonism may help in treating cognitive deficits that often manifest as comorbid pathology in major depressive disorders and chronic pain conditions.[1] Here we demonstrated the lung-protective role of BRL-44408 maleate on sepsis-induced ALI in rats and its antiinflammatory effect in the NR8383 cell line. Our data indicated these effects are associated with inhibiting the activation of ERK1/2, p38, and p65 pathway, with no impact on JNK and PKA. Further efforts are needed to elucidate the relationship between α2A-AR and the ERK1/2, p38MAPK, and NF-κB pathways.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.6449 mL | 23.2245 mL | 46.4490 mL | |
| 5 mM | 0.9290 mL | 4.6449 mL | 9.2898 mL | |
| 10 mM | 0.4645 mL | 2.3224 mL | 4.6449 mL |