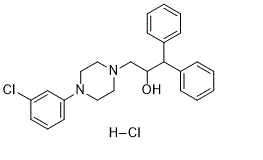
Physicochemical Properties
| Molecular Formula | C25H29CL3N2O |
| Molecular Weight | 479.87 |
| Exact Mass | 478.1345 |
| Elemental Analysis | C, 67.72; H, 6.37; Cl, 15.99; N, 6.32; O, 3.61 |
| CAS # | 1173022-77-9 |
| Related CAS # | 193611-72-2 |
| PubChem CID | 9891303 |
| Appearance | Typically exists as solid at room temperature |
| Boiling Point | 580.7ºC at 760 mmHg |
| Flash Point | 305ºC |
| Vapour Pressure | 2.51E-14mmHg at 25°C |
| LogP | 5.459 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 31 |
| Complexity | 451 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | 0 |
| Synonyms | BRL-15,572; BRL 15572 HCl; 193611-72-2; BRL-15572 dihydrochloride; 3-(4-(3-chlorophenyl)piperazin-1-yl)-1,1-diphenylpropan-2-ol dihydrochloride; BRL-15572 (dihydrochloride); BRL-15572 2HCl; BRL 15572; 3-[4-(3-chlorophenyl)piperazin-1-yl]-1,1-diphenylpropan-2-ol;dihydrochloride; 3-[4-(3-chlorophenyl)piperazin-1-yl]-1,1-diphenylpropan-2-ol dihydrochloride; BRL15572 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 5-HT1D ( pKi = 7.9 ); 5-HT1A ( pKi = 7.7 ); 5-HT2B ( pKi = 7.4 ); 5-HT2A ( pKi = 6.6 ); 5-HT7 ( pKi = 6.3 ) |
| ln Vitro | BRL-15572 has a 60-fold greater affinity than the 5-HT1B receptor for the h5-HT1D receptor produced in CHO cells (pKi=7.9) [1]. [35S]GTPγS binding is stimulated in the membranes of CHO cells expressing h5-HT1B and h5-HT1D receptors by BRL-15572 (0.1 nM–10 μM) [1]. |
| ln Vivo | In the formalin test, BRL-15572 inhibits the analgesic effect caused by (-)-epicatechin [2]. The intraperitoneal injection of BRL-15572 (0.3–100.0 mg/kg) is inert, and BRL-15572 (0.1–10 mg/kg) has no effect on the body temperature of guinea pigs [3]. |
| Enzyme Assay | Despite only modest homology between h5-HT1B and h5-HT1D receptor amino acid sequences, these receptors display a remarkably similar pharmacology. To date there are few compounds which discriminate between these receptor subtypes and those with some degree of selectivity, such as ketanserin, have greater affinity for other 5-HT receptor subtypes. We now report on two compounds, SB-216641 (N-[3-(2-dimethylamino) ethoxy-4-methoxyphenyl]-2'-methyl-4'-(5-methyl-1,2,4-oxadiazol-3-yl)-(1,1'-biphenyl)-4-carboxamide) and BRL-15572 3-[4-(3-chlorophenyl) piperazin-1-yl]-1,1-diphenyl-2-propanol), which display high affinity and selectivity for h5-HT1B and h5-HT1D receptors, respectively. In receptor binding studies on human receptors expressed in CHO cells, SB-216641 has high affinity (pKi = 9.0) for h5-HT1B receptors and has 25-fold lower affinity at h5-HT1D receptors. In contrast, BRL-15572 has 60-fold higher affinity for h5-HT1D (pKi = 7.9) than 5-HT1B receptors. Similar affinities for these compounds were determined on native tissue 5-HT1B receptors in guinea-pig striatum. Functional activities of SB-216641 and BRL-15572 were measured in a [35S]GTPgammaS binding assay and in a cAMP accumulation assay on recombinant h5-HT1B and h5-HT1D receptors. Both compounds were partial agonists in these high receptor expression systems, with potencies and selectivities which correlated with their receptor binding affinities. In the cAMP accumulation assay, results from pK(B) measurements on the compounds again correlated with receptor binding affinities (SB-216641, pK(B) = 9.3 and 7.3; BRL-15572, pK(B) = <6 and 7.1, for h5-HT1B and h5-HT1D receptors respectively). These compounds will be useful pharmacological agents to characterise 5-HT1B and 5-HT1D receptor mediated responses[1]. |
| Animal Protocol |
The aim of this study was to investigate the antinociceptive potential of (-)-epicatechin and the possible mechanisms of action involved in its antinociceptive effect. The carrageenan and formalin tests were used as inflammatory pain models. A plethysmometer was used to measure inflammation and L5/L6 spinal nerve ligation as a neuropathic pain model. Oral (-)-epicatechin reduced carrageenan-induced inflammation and nociception by about 59 and 73%, respectively, and reduced formalin- induced and nerve injury-induced nociception by about 86 and 43%, respectively. (-)-Epicatechin-induced antinociception in the formalin test was prevented by the intraperitoneal administration of antagonists: methiothepin (5-HT1/5 receptor), WAY-100635 (5-HT1A receptor), SB-224289 (5-HT1B receptor), BRL-15572 (5-HT1D receptor), SB-699551 (5-HT5A receptor), naloxone (opioid receptor), CTAP (μ opioid receptor), nor-binaltorphimine (κ opioid receptor), and 7-benzylidenenaltrexone (δ1 opioid receptor). The effect of (-)-epicatechin was also prevented by the intraperitoneal administration of L-NAME [nitric oxide (NO) synthase inhibitor], 7-nitroindazole (neuronal NO synthase inhibitor), ODQ (guanylyl cyclase inhibitor), glibenclamide (ATP-sensitive K channel blocker), 4-aminopyridine (voltage-dependent K channel blocker), and iberiotoxin (large-conductance Ca-activated K channel blocker), but not by amiloride (acid sensing ion channel blocker). The data suggest that (-)-epicatechin exerts its antinociceptive effects by activation of the NO-cyclic GMP-K channels pathway, 5-HT1A/1B/1D/5A serotonergic receptors, and μ/κ/δ opioid receptors.[2] The selective, brain penetrant, 5-HT(1B/D) (formerly 5-HT(1D beta/alpha)) receptor agonist SKF-99101H (3-(2-dimethylaminoethyl)-4-chloro-5-propoxyindole hemifumarate) (30 mg/kg i.p.) causes a dose related fall in rectal temperature in guinea pigs which previous studies have shown to be blocked by the non-selective 5-HT(1B/D) receptor antagonist GR-127935 (N-[4-methoxy-3-(4-methyl-1-piperazinyl) phenyl]-2'-methyl-4'-(5-methyl-1,2,4-oxadiazol-3-yl) [1,1'biphenyl]-4-carboxamide oxalate). The present study shows that the hypothermic response to SKF-99101H is dose-dependently blocked by SB-224289G (1'-methyl-5-(2'-methyl-4'-[(5-methyl-1,2,4-oxadiazol-3-yl)bipheny l-4-yl]carbonyl)-2,3,6,7-tetrahydrospiro[furo[2,3-f]indole-3,4'-pi peridone] hemioxalate) (0.3-10.0 mg/kg p.o.) (ED50 3.62 mg/kg), which is the first compound to be described which is more than 60 fold selective for the 5-HT1B receptor over the 5-HT1D receptor. SB-216641A (N-[3-(2-dimethylamino) ethoxy-4-methoxy-phenyl] 2'-methyl-4'-(5-methyl-1,2,4-oxadiazol-3-yl)-(1,1'-biphenyl)-4-car boxamide hydrochloride) (0.6-20.0 mg/kg i.p.), which is somewhat less selective (30 fold) for the 5-HT1B receptor over the 5-HT1D receptor had a similar effect (ED50 4.43 mg/kg). The brain penetrant 5-HT1D selective receptor antagonist, BRL-15572 (4-(3-chlorophenyl)-alpha-(diphenylmethyl)-1-piperazineethanol+ ++ dihydrochloride) (0.3-100.0 mg/kg i.p.) was inactive. When administered alone neither BRL-15572 (0.1-10 mg/kg i.p.) nor SB-224289G (2.2-22 mg/kg p.o.) had an effect on body temperature. These data demonstrate that 5-HT1B (formerly 5-HT(1D beta)) and not 5-HT1D (formerly 5-HT(1D alpha)) receptors mediate the hypothermic response to SKF-99101H (30 mg/kg i.p.) in guinea pigs. The compounds described are useful pharmacological tools for distinguishing responses to 5-HT1B and 5-HT1D receptors.[3] |
| References |
[1]. SB-216641 and BRL-15572--compounds to pharmacologically discriminate h5-HT1B and h5-HT1D receptors. Naunyn Schmiedebergs Arch Pharmacol. 1997 Sep; 356(3): 312-20. [2]. Antinociceptive effect of (-)-epicatechin in inflammatory and neuropathic pain in rats. Behav Pharmacol. 2018 Apr; 29(2 and 3-Spec Issue): 270-279. [3]. Stimulation of 5-HT1B receptors causes hypothermia in the guinea pig. Eur J Pharmacol. 1997 Jul 23; 331(2-3): 169-74. |
| Additional Infomation |
1. In the present study, we investigated how alloxan-induced diabetes affects the ability of 5-hydroxytryptamine (5-HT) to modulate bradycardia induced in vivo by electrical stimulation of the vagus nerve in pithed rats. We also analysed the type and/or subtype of 5-HT receptors involved. 2. Diabetes was induced in male Wistar rats with a single injection of alloxan (150 mg/kg, s.c.). Four weeks later, rats were anaesthetized, pretreated with atenolol and pithed. Electrical stimulation (3, 6 and 9 Hz) of the vagus nerve resulted in frequency dependent decreases in heart rate (HR). 3. In diabetic rats, intravenous bolus administration of high doses of 5-HT (100 and 200 microg/kg) increased the bradycardia induced by vagal electrical stimulation. Similarly, low doses (10 microg/kg) of the 5-HT(1/7) receptor agonist 5-carboxamidotryptamine (5-CT), increased vagally induced bradycardia. However, at high doses (50, 100 and 150 microg/kg), 5-CT reduced the bradycardia. Attenuation of the vagally induced bradycardia evoked by the higher doses of 5-CT was reproduced by L-694,247 (50 microg/kg), a selective agonist for the non-rodent 5-HT(1B) and 5-HT(1D) receptors. Enhancement of the vagally induced bradycardia elicited by low doses of 5-CT was reproduced by the selective 5-HT(1A) receptor agonist 8-hydroxydipropylaminotretalin hydrobromide (8-OH-DPAT; 50 microg/kg). These stimulatory and inhibitory actions on vagal stimulation-induced bradycardia in diabetic rats were also observed after administration of exogenous acetylcholine. 4. Vagally induced bradycardia in diabetic rats was not affected by administration of the selective 5-HT(2) receptor agonist alpha-methyl-5-HT (150 microg/kg), the selective 5-HT(3) receptor agonist 1-phenylbiguanide (150 microg/kg) or the selective 5-HT(1B) receptor agonist CGS-12066B (50 microg/kg). 5. Enhancement of the electrical stimulation-induced bradycardia in diabetic rats caused by 5-CT (10 microg/kg) or 8-OH-DPAT (50 microg/kg) was abolished by the selective 5-HT(2/7) receptor antagonist mesulergine (1 mg/kg) and the selective 5-HT(1A) receptor antagonist WAY-100,635 (100 microg/kg), respectively. Similarly, pretreatment with the non-selective 5-HT(1) receptor antagonist methiothepin (0.1 mg/kg) blocked the inhibitory effect of 5-CT (50 microg/kg) on the bradycardia induced by vagal electrical stimulation in diabetic rats. BRL-15572 (2 microg/kg), a selective 5-HT(1D) receptor antagonist, inhibited the action of L-694,247 (50 microg/kg), a selective agonist for the non-rodent 5-HT(1B) and 5-HT(1D) receptors, on the vagally induced bradycardia. 6. In conclusion, in the present study, experimental diabetes evoked changes in both the nature and 5-HT receptor types/subtypes involved in vagally induced bradycardia.[https://pubmed.ncbi.nlm.nih.gov/17880377/] It has previously been suggested that ergotamine produces external carotid vasoconstriction in vagosympathectomised dogs via 5-HT1B/1D receptors and alpha2-adrenoceptors. The present study has reanalysed this suggestion by using more selective antagonists alone and in combination. Fifty-two anaesthetised dogs were prepared for ultrasonic measurements of external carotid blood flow. The animals were divided into thirteen groups (n=4 each) receiving an i.v. bolus injection of, either physiological saline (0.3 ml/kg; control), or the antagonists SB224289 (300 microg/kg; 5-HT1B), BRL15572 (300 microg/kg; 5-HT1D), rauwolscine (300 microg/kg; alpha2), SB224289 + BRL15572 (300 microg/kg each), SB224289 + rauwolscine (300 microg/kg each), BRL15572 + rauwolscine (300 microg/kg each), rauwolscine (300 microg/kg) + prazosin (100 microg/kg; alpha1), SB224289 (300 microg/kg) + prazosin (100 microg/kg), SB224289 (300 microg/kg) + rauwolscine (300 microg/kg) + prazosin (100 microg/kg), SB224289 (300 microg/kg) + prazosin (100 microg/kg) + BRL44408 (1,000 microg/kg; alpha2A), SB224289 (300 microg/kg) + prazosin (100 microg/kg)+ imiloxan (1,000 microg/kg; alpha2B), or SB224289 (300 microg/kg) + prazosin (100 microg/kg) + MK912 (300 microg/kg; alpha2C). Each group received consecutive 1-min intracarotid infusions of ergotamine (0.56, 1, 1.8, 3.1, 5.6, 10 and 18 microg/min), following a cumulative schedule. In saline-pretreated animals, ergotamine induced dose-dependent decreases in external carotid blood flow without affecting arterial blood pressure or heart rate. These control responses were: unaffected by SB224289, BRL15572, rauwolscine or the combinations of SB224289 + BRL15572, BRL15572 + rauwolscine, rauwolscine + prazosin, SB224289 + prazosin, or SB224289 + prazosin + imiloxan; slightly blocked by SB224289 + rauwolscine; and markedly blocked by SB224289 + rauwolscine + prazosin, SB224289 + prazosin + BRL44408 or SB224289 + prazosin + MK912. Thus, the cranio-selective vasoconstriction elicited by ergotamine in dogs is predominantly mediated by 5-HT1B receptors as well as alpha2A/2C-adrenoceptor subtypes and, to a lesser extent, by alpha1-adrenoceptors.[https://pubmed.ncbi.nlm.nih.gov/15224175/] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~250 mg/mL (~520.97 mM) H2O : ~2 mg/mL (~4.17 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.33 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.33 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.33 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0839 mL | 10.4195 mL | 20.8390 mL | |
| 5 mM | 0.4168 mL | 2.0839 mL | 4.1678 mL | |
| 10 mM | 0.2084 mL | 1.0419 mL | 2.0839 mL |